Proteomics and Metabolomics — MCQs

Techniques used for protein expression proteomics study include:
Who was awarded the Nobel Prize for determining the amino acid sequence of insulin?
Methionine can enter the TCA cycle at which level?
Which of the following is a feature of Phenylketonuria?
Separation of proteins based on size is done by
What is the primary purpose of xenobiotic metabolism?
Final common pathway of metabolism of carbohydrate, lipids, and protein metabolism is?
A 6-year-old presents with developmental delay, musty body odor, and fair skin. Lab tests show high phenylalanine levels. What is the most appropriate management?
The technique shown in the image is:
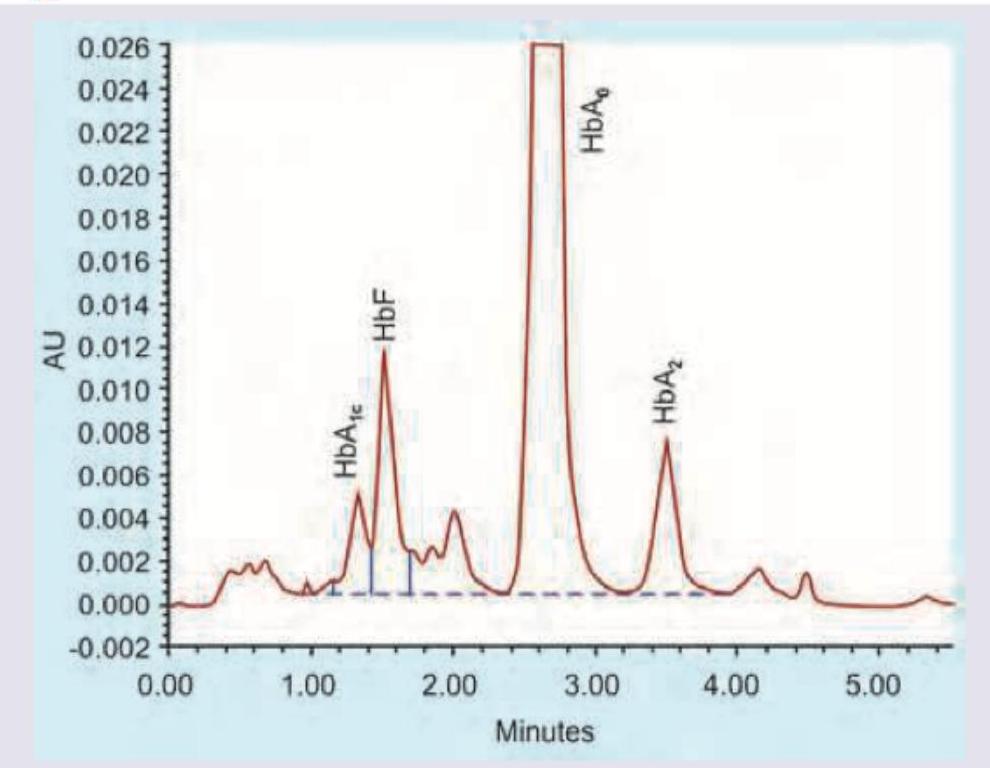
Which of the following separates proteins solely on the basis of their molecular size?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
