Acid-Base and Electrolyte Balance — MCQs

On this page
Which of the following phospholipids serves as a marker of apoptosis?
In metabolic alkalosis, which statement is true about urinary excretion?
Which of the following amino acids primarily acts as a buffer in blood due to its ability to accept and donate protons at physiological pH?
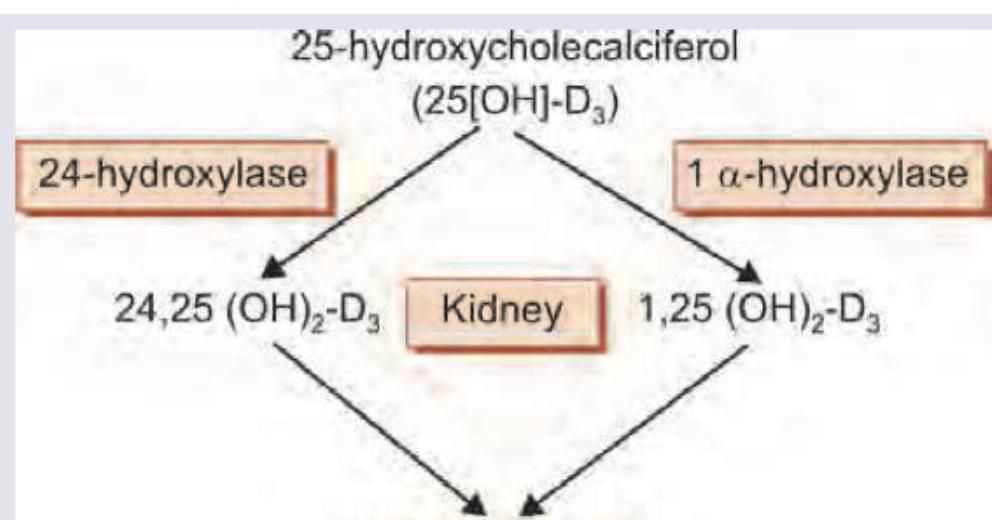
The following reaction occurs in which part of kidney?
Which one of the following biochemical abnormalities can be produced by repeated vomiting?
Maximum buffering capacity at physiological pH is for:
What is the daily requirement of potassium in a healthy adult?
Concentration of H+ ion is 10-9. What is the pH of the solution?
Osmolarity of 4.2% solution of sodium bicarbonate is ?
What is the normal serum magnesium level?
Practice by Chapter
Acid-Base Chemistry and Buffers
Practice Questions
pH Regulation in Body Fluids
Practice Questions
Respiratory Regulation of Acid-Base Balance
Practice Questions
Renal Regulation of Acid-Base Balance
Practice Questions
Respiratory and Metabolic Acidosis
Practice Questions
Respiratory and Metabolic Alkalosis
Practice Questions
Mixed Acid-Base Disorders
Practice Questions
Interpretation of Arterial Blood Gases
Practice Questions
Electrolyte Homeostasis
Practice Questions
Sodium and Water Balance
Practice Questions
Potassium Balance
Practice Questions
Calcium and Phosphate Metabolism
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
