Acid-Base and Electrolyte Balance — MCQs

On this page
Diabetes mellitus is associated with which type of lactic acidosis?
Which of the following is NOT a component of Oral Rehydration Salts (ORS)?
Lactic acidosis is not seen in which of the following conditions or substances?
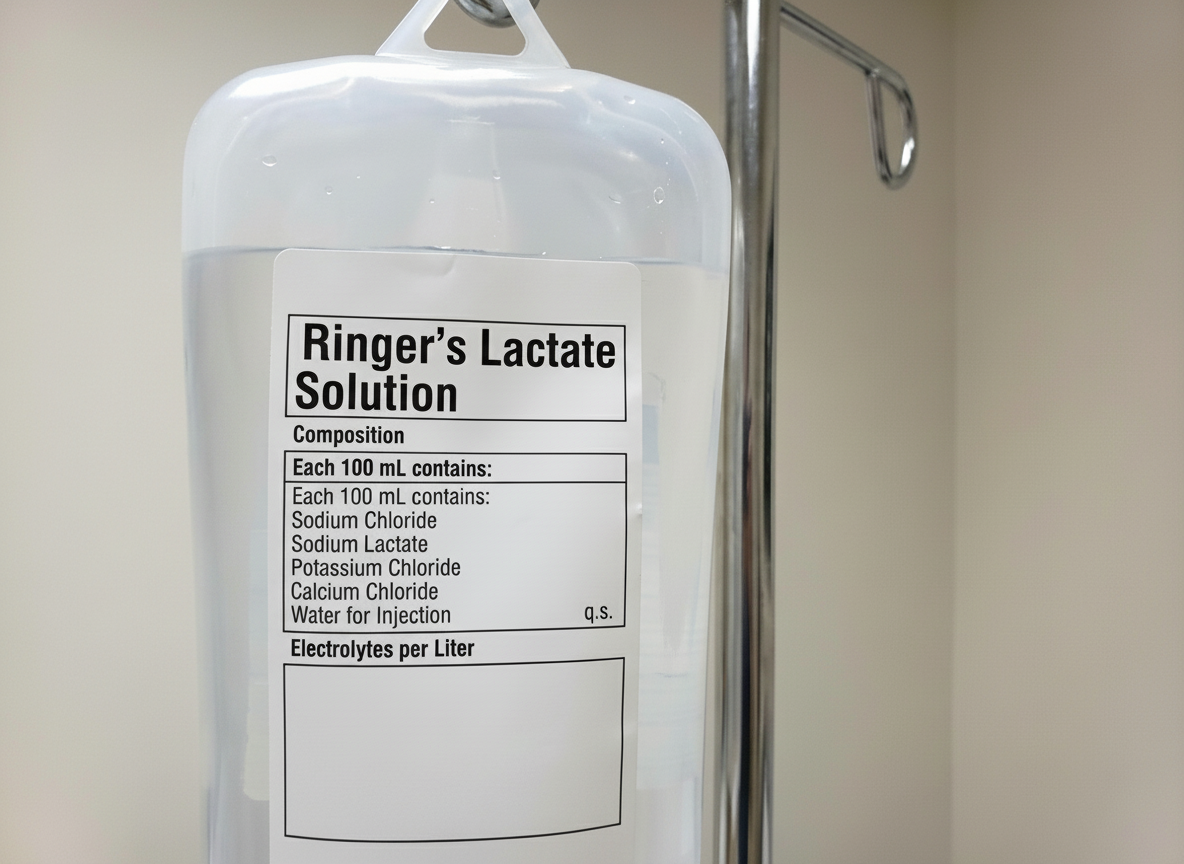
What is the total amount of potassium delivered to the patient after administering one bottle?
Normal anion gap is seen in all of the following conditions except:
What is the commonest cause of metabolic alkalosis?
A 45-year-old woman with Crohn disease and a small intestinal fistula develops tetany during the second week of parenteral nutrition. The laboratory findings include Na: 135 mEq/L K: 3.2 mEq/L Cl: 103 mEq/L HCO3 : 25 mEq/L Ca: 8.2 mEq/L Mg: 1.2 mEq/L PO4 : 2.4 mEq/L Albumin: 2.4. An arterial blood gas sample reveals a pH of 7.42, PCO2 of 38 mm Hg, and PO2 of 84 mm Hg. Which of the following is the most likely cause of the patient's tetany?
Persistent vomiting most likely causes which of the following?
Hypokalemia is not present in which of the following conditions?
Which of the following statements regarding insulin is true?
Practice by Chapter
Acid-Base Chemistry and Buffers
Practice Questions
pH Regulation in Body Fluids
Practice Questions
Respiratory Regulation of Acid-Base Balance
Practice Questions
Renal Regulation of Acid-Base Balance
Practice Questions
Respiratory and Metabolic Acidosis
Practice Questions
Respiratory and Metabolic Alkalosis
Practice Questions
Mixed Acid-Base Disorders
Practice Questions
Interpretation of Arterial Blood Gases
Practice Questions
Electrolyte Homeostasis
Practice Questions
Sodium and Water Balance
Practice Questions
Potassium Balance
Practice Questions
Calcium and Phosphate Metabolism
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
