Acid-Base and Electrolyte Balance — MCQs

On this page
Histidine is the most important amino acid for buffering under normal physiological conditions because?
Which of the following is associated with elevated alkaline phosphatase, low calcium, and low phosphate?
Which of the following conditions is NOT associated with an increased anion-gap metabolic acidosis?
Which of the following buffer systems is most effective for maintaining a physiological pH of 7.4?
All are causes of increased anion gap except?
Which of the following is a precursor of the molecule shown in the illustration?
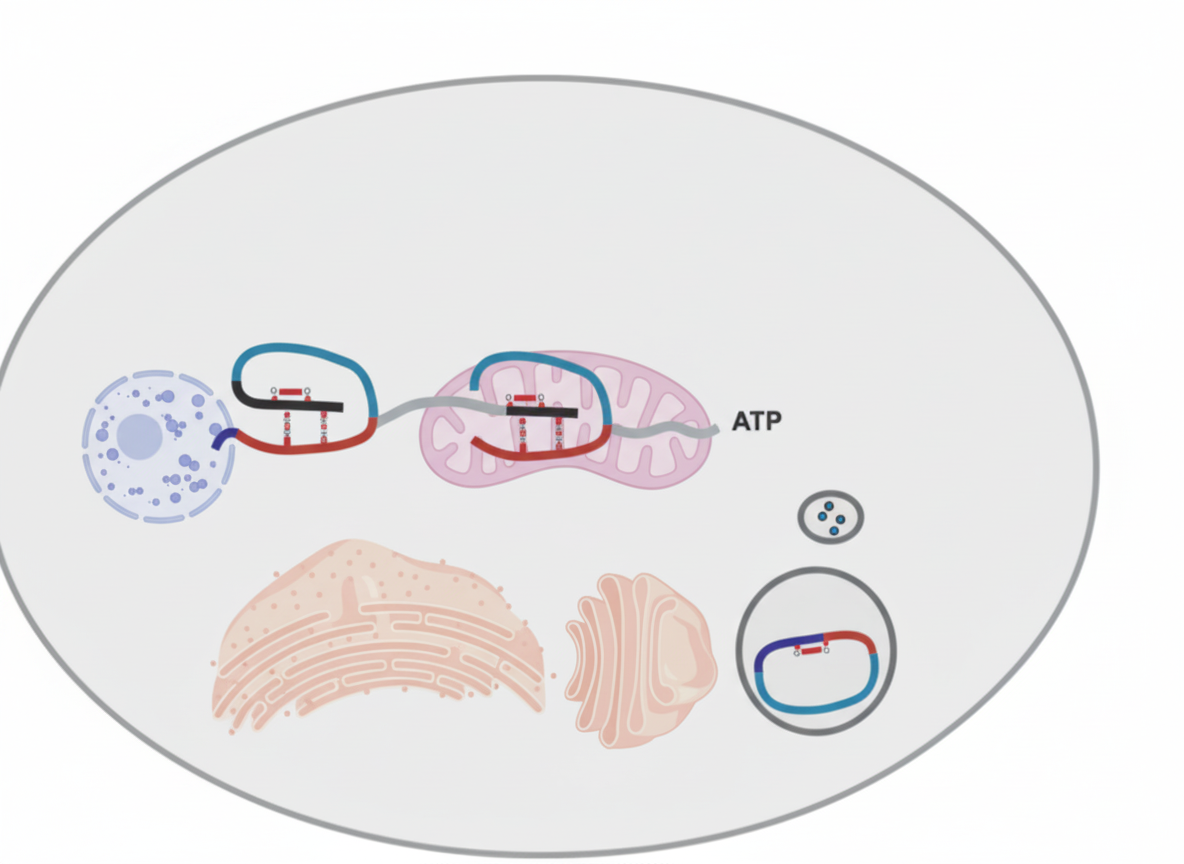
Which of the following conditions is associated with normal anion gap acidosis?
Increased anion gap is not seen in which of the following conditions?
A 40-year-old male presents with recurrent bouts of vomiting for 9 months because of pyloric obstruction. What is the compensatory biochemical change?
Hypercalcemia is seen in all of the following conditions except?
Practice by Chapter
Acid-Base Chemistry and Buffers
Practice Questions
pH Regulation in Body Fluids
Practice Questions
Respiratory Regulation of Acid-Base Balance
Practice Questions
Renal Regulation of Acid-Base Balance
Practice Questions
Respiratory and Metabolic Acidosis
Practice Questions
Respiratory and Metabolic Alkalosis
Practice Questions
Mixed Acid-Base Disorders
Practice Questions
Interpretation of Arterial Blood Gases
Practice Questions
Electrolyte Homeostasis
Practice Questions
Sodium and Water Balance
Practice Questions
Potassium Balance
Practice Questions
Calcium and Phosphate Metabolism
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
