Electrolyte Homeostasis — MCQs

In hypoparathyroidism:
Osmolality of plasma in a normal adult:
Which of the following is an early symptom of hypermagnesemia?
The second most abundant intracellular cation is
Given the following electrolyte values: Sodium (Na+) = 140 mmol/L, Potassium (K+) = 3 mmol/L, Chloride (Cl-) = 112 mmol/L, and Bicarbonate (HCO3-) = 16 mmol/L, what is the plasma anion gap?
Rapid infusion of insulin causes
Which of the following statements about normal saline is false?
In a patient with severe dehydration, which of the following compensatory mechanisms work together to restore blood volume and maintain hemodynamic stability?
Most clinically significant characteristic of Ringer's Lactate is -
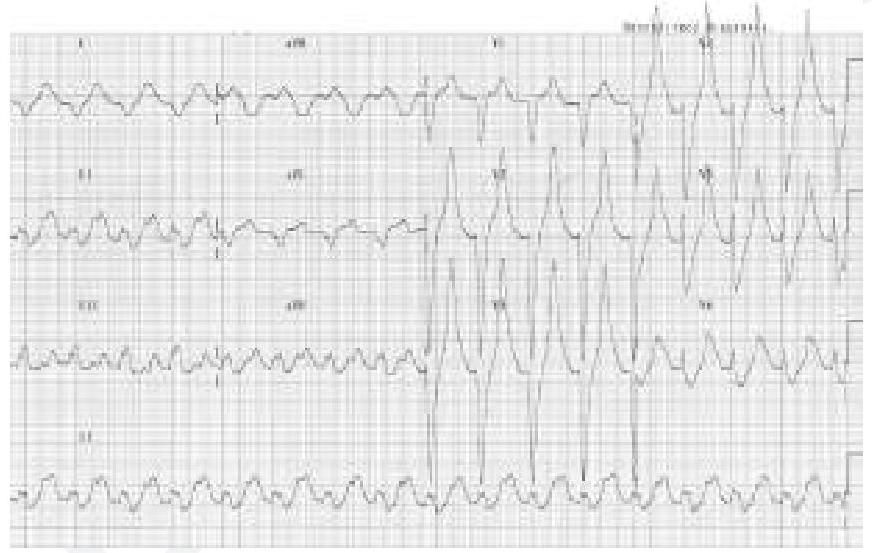
A patient who is a known case of hypertension on multiple anti-hypertensive medications came to OPD. His ECG finding is given below. Which of the following drugs is responsible for the ECG finding? (Image of ECG finding)
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
