Histology — MCQs

On this page
Paneth cells are mainly found in the bases of the crypts in the small intestine. All of the following are true about Paneth cells, EXCEPT:
Gomori's aldehyde fuchsin specifically stains which of the following?
Which cells in the stomach are responsible for secreting acid?
A newborn baby presented with profuse bleeding from the umbilical stump. Rest of the examination and PT, APTT are within normal limits. Most likely diagnosis is which of the following?
Which of the following is the weakest type of cartilage?
A 50-year-old male with a positive family history of prostate cancer presents for screening. What is the most sensitive screening test to detect prostate cancer?
All of the following are macrophages except:
The type of collagen depicted in the staining is:
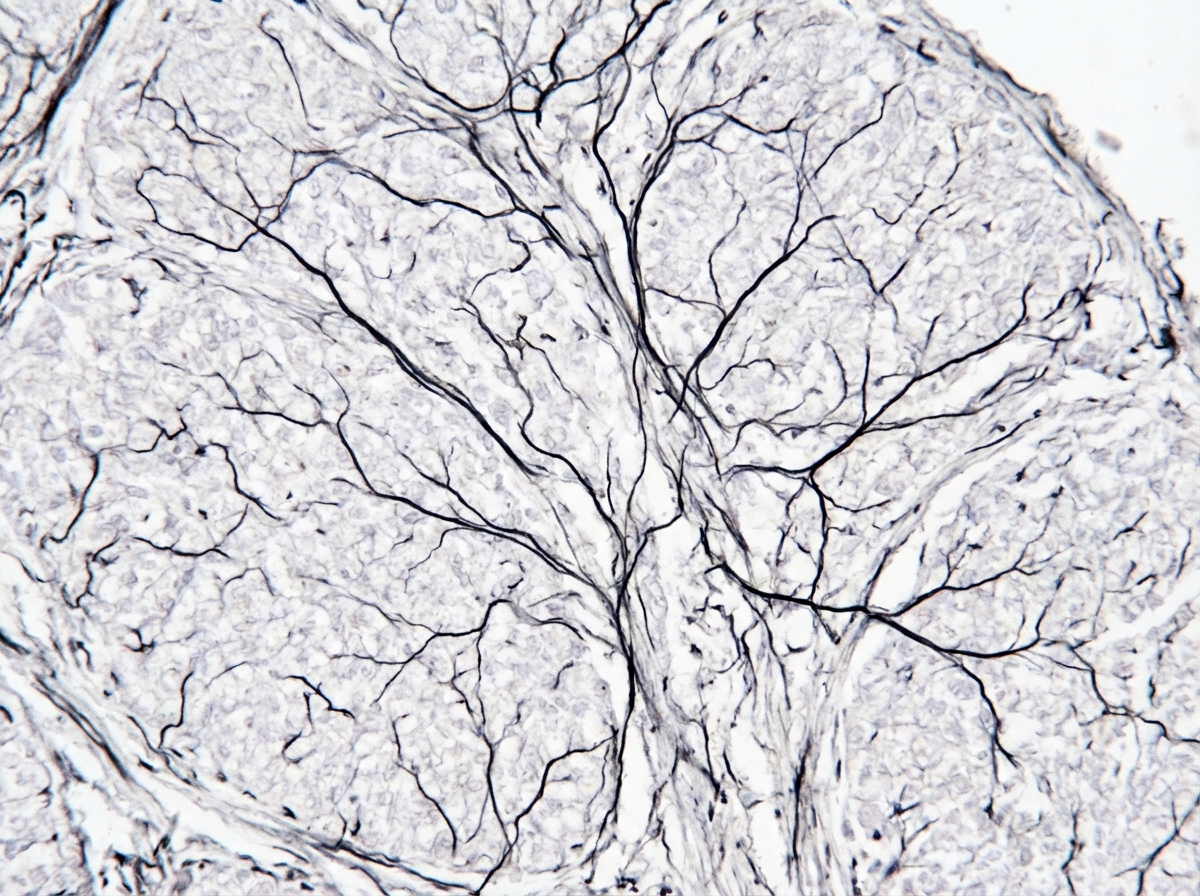
Which of the following is NOT true about hepatocytes?
What is the origin and characteristic of osteoclasts?
Practice by Chapter
Basic Tissue Types
Practice Questions
Cell Biology and Organelles
Practice Questions
Epithelial Tissue
Practice Questions
Connective Tissue
Practice Questions
Muscular Tissue
Practice Questions
Nervous Tissue
Practice Questions
Cardiovascular System Histology
Practice Questions
Lymphoid Organs and Immune System
Practice Questions
Endocrine System Histology
Practice Questions
Respiratory System Histology
Practice Questions
Digestive System Histology
Practice Questions
Urinary and Reproductive System Histology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
