Embryology and Development — MCQs

On this page
Which blood vessel carries deoxygenated blood back to the placenta?
Fingerprint first develops in how many weeks of intrauterine life?
A child lacks thymus and inferior parathyroid glands. Defective development of which of the following structures is likely to be the cause?
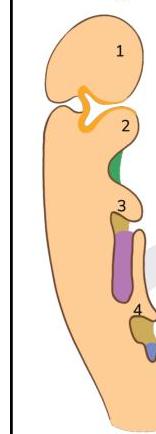
What is the incorrect statement?
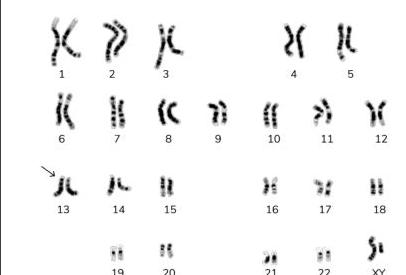
Identify the type of marked chromosome in the given karyotype.
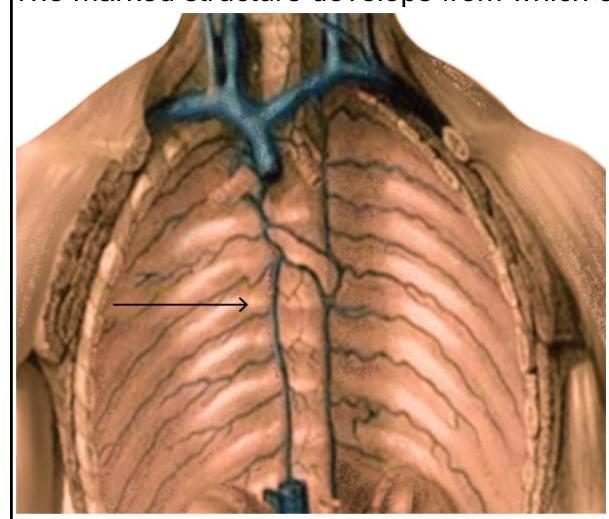
The marked structure develops from which of the following structures?
During embryological development, failure of the lateral palatine processes to fuse with the primary palate results in which of the following conditions?
During development, a fetus is found to have incomplete fusion of the neural tube. Which of the following structures would most likely be affected by this developmental defect?
During embryological development, failure of the urorectal septum to completely separate the cloaca results in which of the following congenital anomalies?
Secondary ossification centre appears before birth at
Practice by Chapter
Gametogenesis and Fertilization
Practice Questions
Early Embryonic Development
Practice Questions
Placentation
Practice Questions
Development of Nervous System
Practice Questions
Development of Cardiovascular System
Practice Questions
Development of Gastrointestinal System
Practice Questions
Development of Urogenital System
Practice Questions
Development of Musculoskeletal System
Practice Questions
Development of Head and Neck
Practice Questions
Congenital Anomalies
Practice Questions
Teratology
Practice Questions
Molecular Mechanisms in Development
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
