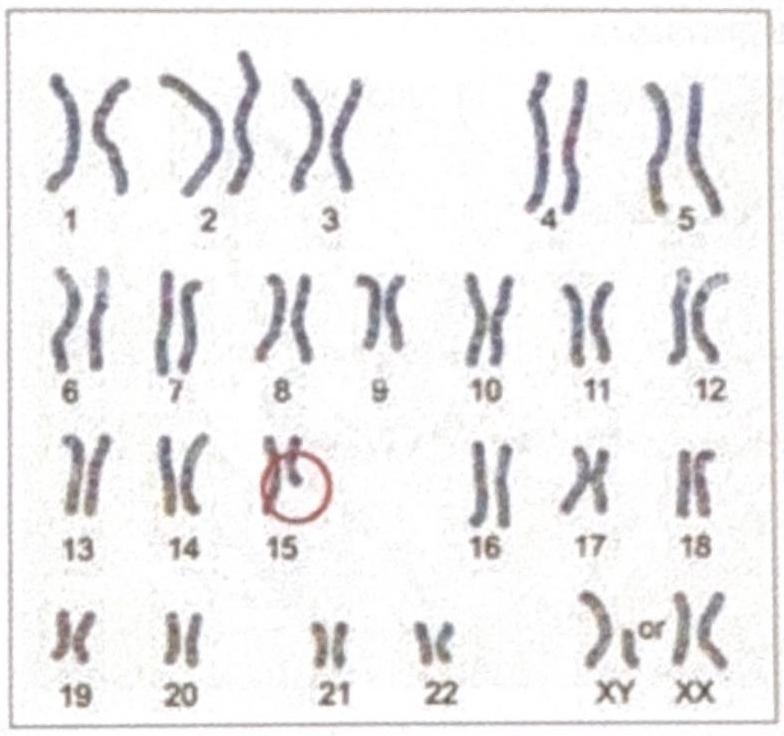
The diagrammatic representation of the karyotype of an individual indicates a specific genetic abnormality. What is the diagnosis?
Enter your email to get your 85% OFF code and unlock the full NEET PG question bank on the app.
A patient's relatives sent a message on social media to the consulting doctor, mentioning that the patient's urinary coproporphyrin test is positive. What is the probable cause?
A neonate was brought to the hospital with chief complaints of poor feeding, vomiting, acidosis, and cataract. Benedict's test on urine was positive, but urinary glucose was negative. What is the defective enzyme in the above-mentioned disorder?
A patient presents with a skin rash that is exaggerated on sun exposure. What is the repair mechanism involved in this condition?
A 45-year-old patient presents with joint pain and weakness and is known to have homocystinuria. Which vitamin is required in the treatment?
A patient came to the hospital with severe abdominal pain, and lipase levels were elevated. On imaging, a stone is found in the common bile duct (CBD). Which enzyme is most likely elevated in this condition?
A patient who has always neglected his nutrition presented with follicular hyperkeratosis on the extensor aspect of the forearm. What is the diagnosis?
The diagrammatic representation of the karyotype of an individual indicates a specific genetic abnormality. What is the diagnosis?

A 13-year-old boy presents with jaundice, fatigue, muscle stiffness, tremors, and behavioral changes. Examination reveals an enlarged liver and spleen. A Kayser-Fleischer ring was noted. What is the definitive diagnostic test?
A patient with a history of binge alcohol intake presented to the emergency department with convulsions, altered sensorium, and a plasma glucose level of $45 \mathrm{mg} / \mathrm{dL}$. Which of the following treatments is needed?
Methotrexate use causes reduced synthesis of which of the following?