A patient came to the emergency room with severe abdominal pain. The serum triglyceride level was $1500 \mathrm{mg} / \mathrm{dL}$. What is the most likely defect?
A patient's relatives sent a message on social media to the consulting doctor, mentioning that the patient's urinary coproporphyrin test is positive. What is the probable cause?
A neonate was brought to the hospital with chief complaints of poor feeding, vomiting, acidosis, and cataract. Benedict's test on urine was positive, but urinary glucose was negative. What is the defective enzyme in the above-mentioned disorder?
A 45-year-old patient presents with joint pain and weakness and is known to have homocystinuria. Which vitamin is required in the treatment?
A patient came to the hospital with severe abdominal pain, and lipase levels were elevated. On imaging, a stone is found in the common bile duct (CBD). Which enzyme is most likely elevated in this condition?
What is the primary function of IL-8?
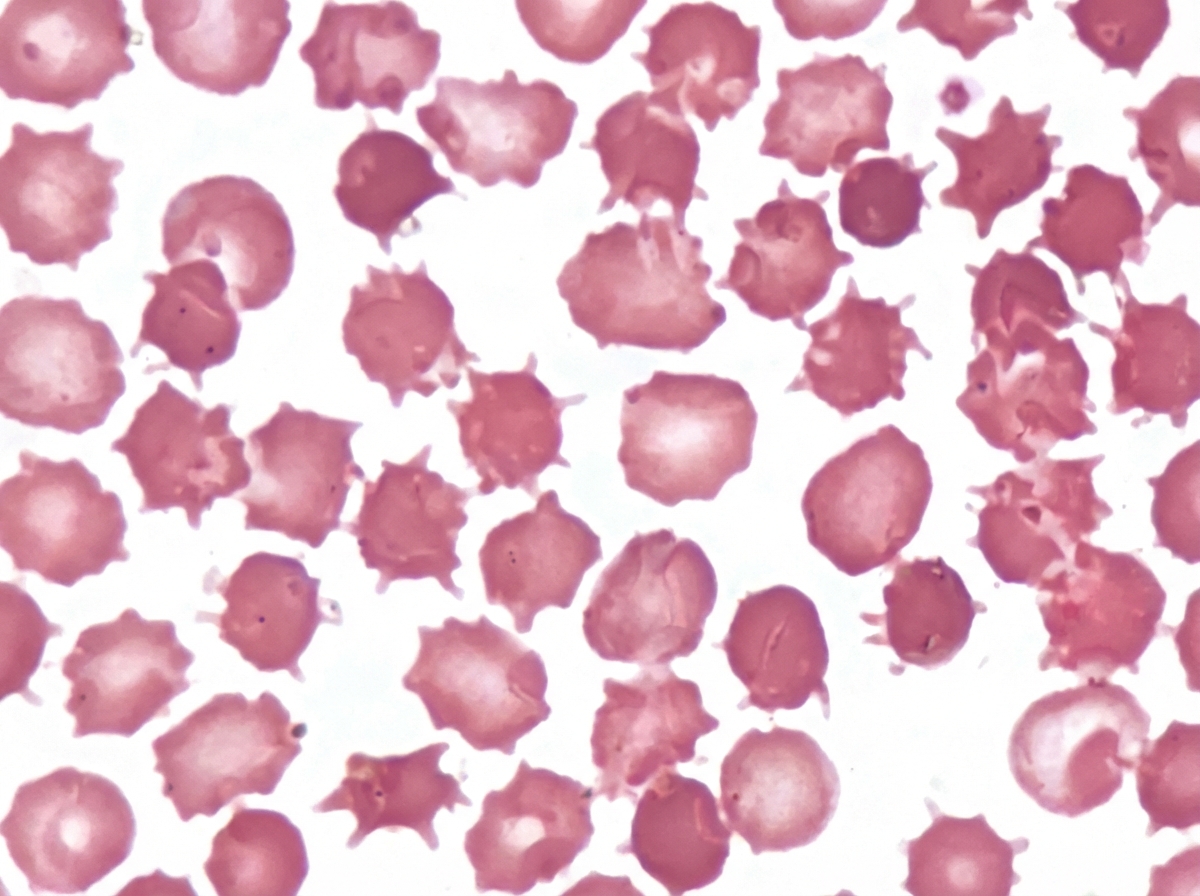
An adult male presented with a protruding abdomen, diarrhea, visual symptoms, and neurological manifestations. His LDL is low. Based on the peripheral smear finding shown in the image, what is the likely diagnosis?
An adult tall male presents with a long arm span, pectus excavatum, and cardiac abnormalities. What is the most likely defective protein?
A patient with homocystinuria presents with ectopia lentis (dislocation of the lens). Which vitamin should be supplemented?
Which of the following foods should be consumed to prevent thiamine deficiency?