INI-CET 2024 — Pathology
8 Previous Year Questions with Answers & Explanations
HBsAg is based on which principle
In which type of lung carcinoma is the p53 mutation most commonly observed?
ER positivity is used as which of the following in the context of breast carcinoma?
Which is an intranuclear immunohistochemistry marker for neuroendocrine tumors?
A patient presents with neck swelling causing compression of the trachea and esophagus. Histopathological assessment reveals cell nests and pink extracellular amyloid stroma. What is the cell of origin of the tumor associated with these findings?
Molecular genetic testing is used to detect all of the following except?
To replenish the inventory, blood banks routinely issue blood packets which are close to the expiry date. Which of the following will be the closest to expiry date, according to the anticoagulant used and the method of storage of the packet:
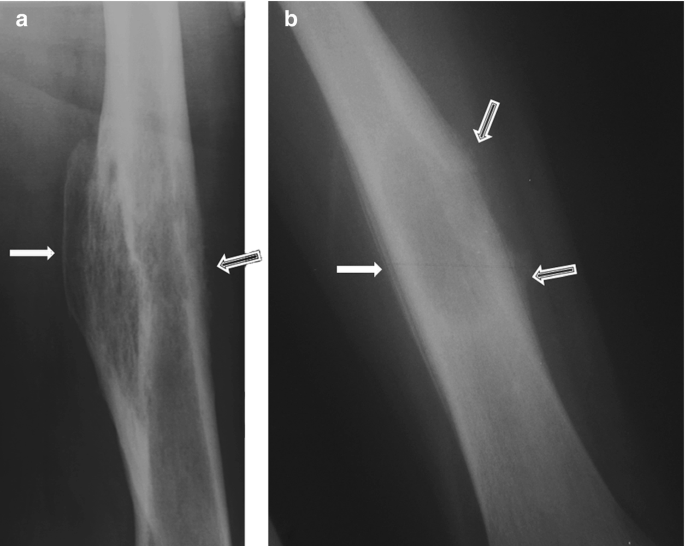
A 12-year-old Caucasian male presents with his mother to the pediatrician’s office complaining of right thigh pain. He reports that he has noticed slowly progressive pain and swelling over the distal aspect of his right thigh over the past two months. He denies any recent trauma to the area and his temperature is 100.9°F (38.3°C). On exam, there is swelling and tenderness overlying the distal right femoral diaphysis. Laboratory evaluation is notable for an elevated white blood cell (WBC) count and elevated erythrocyte sedimentation rate (ESR). A radiograph of the patient’s right leg is shown. Biopsy of the lesion demonstrates sheets of monotonous small round blue cells with minimal cytoplasm. Which of the following genetic mutations is most likely associated with this patient’s condition?
INI-CET 2024 - Pathology INI-CET Practice Questions and MCQs
Question 1: HBsAg is based on which principle
- A. Chemiluminescence
- B. Immunofluorescence
- C. Immunochromatography assays
- D. ELISA (Correct Answer)
Explanation: ***ELISA*** - **Enzyme-linked immunosorbent assay (ELISA)** is a widely used laboratory test to detect and quantify antigens (like HBsAg) or antibodies in a sample. - It involves an enzyme-linked antibody that reacts with a substrate to produce a detectable signal, making it highly sensitive and specific for **HBsAg detection**. *Immunochromatography assays* - These are typically **rapid diagnostic tests (RDTs)** that provide quick qualitative results, often used for point-of-care testing. - While they can detect HBsAg, they generally have lower sensitivity and specificity compared to ELISA. *Chemiluminescence* - This is a detection method used in some immunoassays where a chemical reaction emits light, often providing higher sensitivity than colorimetric detection. - While it can be incorporated into HBsAg testing platforms, it is a *detection principle* rather than the primary assay principle like ELISA itself. *Immunofluorescence* - This technique uses **fluorescently labeled antibodies** to visualize antigens in cells or tissues under a fluorescence microscope [1]. - It is used for localization and identification of antigens, but not typically the primary method for routine quantitative HBsAg serology [1]. **References:** [1] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. (Basic Pathology) introduces the student to key general principles of pathology, both as a medical science and as a clinical activity with a vital role in patient care. Part 2 (Disease Mechanisms) provides fundamental knowledge about the cellular and molecular processes involved in diseases, providing the rationale for their treatment. Part 3 (Systematic Pathology) deals in detail with specific diseases, with emphasis on the clinically important aspects., pp. 259-260.
Question 2: In which type of lung carcinoma is the p53 mutation most commonly observed?
- A. Adenocarcinoma
- B. Squamous cell carcinoma (SCC) (Correct Answer)
- C. Large cell carcinoma
- D. Small cell carcinoma
Explanation: ***Small cell carcinoma*** - **Small cell lung carcinoma (SCLC)** has the highest frequency of **p53 mutations**, occurring in approximately **90-95%** of cases. - These mutations are associated with the **aggressive nature** and **poor prognosis** of SCLC, contributing to its rapid growth and early metastasis. *Adenocarcinoma* - **Adenocarcinoma** has p53 mutations in approximately **50-60%** of cases, which is less frequent than SCLC. - This subtype is more commonly associated with **EGFR mutations** and **ALK rearrangements**, particularly in non-smokers. *Squamous cell carcinoma (SCC)* - **Squamous cell carcinoma** shows p53 mutations in about **70-80%** of cases, but still lower than SCLC. - It is more strongly associated with **smoking** and often displays mutations in **CDKN2A** and **PIK3CA** pathways. *Large cell carcinoma* - **Large cell carcinoma** has variable p53 mutation rates, typically **40-60%** of cases. - This subtype is less well-characterized molecularly and represents a **diagnosis of exclusion** among lung cancers.
Question 3: ER positivity is used as which of the following in the context of breast carcinoma?
- A. Treatment option
- B. Prognostic marker (Correct Answer)
- C. Molecular marker
- D. Diagnostic marker
Explanation: ***Prognostic marker*** - **ER positivity** in **breast carcinoma** is primarily used as a **prognostic marker** indicating more favorable disease outcome [1]. - ER-positive tumors generally **grow more slowly**, are **less aggressive**, and have **better overall survival** compared to ER-negative tumors [2]. - While ER status also has **predictive value** for endocrine therapy response, its classification as a prognostic indicator reflects its association with inherently better tumor biology and patient outcomes [1]. - ER positivity correlates with **well-differentiated tumors** and **lower grade** malignancies. *Treatment option* - ER positivity is not a treatment itself, but rather a **biomarker** that guides treatment selection. - It identifies patients who may benefit from **endocrine therapy** (tamoxifen, aromatase inhibitors) [1]. *Molecular marker* - While ER is indeed a molecular marker (receptor protein detected by immunohistochemistry), this term is too **broad and non-specific**. - The question asks for the **specific clinical utility** of ER positivity, not its general classification. *Diagnostic marker* - ER status is **not used for initial diagnosis** of breast carcinoma. - Diagnosis requires **histopathological examination** of tissue biopsy. - ER testing is performed **after diagnosis** to characterize the tumor and guide management. **References:** [1] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. The Breast, pp. 1059-1060. [2] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. The Breast, pp. 1064-1066.
Question 4: Which is an intranuclear immunohistochemistry marker for neuroendocrine tumors?
- A. NCAM1/CD56
- B. Chromogranin
- C. Synaptophysin
- D. INSM (Correct Answer)
Explanation: ***INSM*** - **INSM1 (Insulinoma-associated protein 1)** is a highly sensitive and specific **transcription factor** expressed in neuroendocrine cells. - It exhibits **intranuclear staining** in immunohistochemistry, making it a reliable marker for neuroendocrine differentiation in tumors. *NCAM1/CD56* - **NCAM1/CD56** is a **cell surface adhesion molecule** - It shows **membranous or cytoplasmic staining** in immunohistochemistry, not intranuclear. *Chromogranin* - **Chromogranin A** is a **storage protein** found in dense core granules of neuroendocrine cells. [1] - It demonstrates **cytoplasmic staining** in immunohistochemistry and is a general neuroendocrine marker. [1] *Synaptophysin* - **Synaptophysin** is a **transmembrane glycoprotein** associated with synaptic vesicles. - It exhibits **cytoplasmic or membranous staining** in immunohistochemistry and is also a general neuroendocrine marker. [2] **References:** [1] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. Common Clinical Problems From Liver And Biliary System Disease, pp. 419-420. [2] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. The Gastrointestinal Tract, pp. 780-781.
Question 5: A patient presents with neck swelling causing compression of the trachea and esophagus. Histopathological assessment reveals cell nests and pink extracellular amyloid stroma. What is the cell of origin of the tumor associated with these findings?
- A. Parafollicular C cells (Correct Answer)
- B. Hurthle cells
- C. Follicular cells
- D. Chief cells
Explanation: ***Parafollicular C cells*** - The presence of **cell nests** and **pink extracellular amyloid stroma** are classic histopathological findings for **medullary thyroid carcinoma (MTC)**, which originates from the parafollicular C cells [2], [3]. - Parafollicular C cells are responsible for producing **calcitonin**, and the amyloid in these tumors is derived from calcitonin [3]. - MTC accounts for 5-10% of thyroid cancers and can be sporadic or familial (associated with MEN 2A and 2B syndromes) [1], [4]. *Chief cells* - Chief cells are **parathyroid gland cells** that produce parathyroid hormone (PTH), not thyroid tumor cells. - While parathyroid adenomas can cause neck masses, they do not produce the characteristic amyloid stroma seen in medullary carcinoma. *Hürthle cells* - Hürthle cells (also known as Askanazy cells or oncocytes) are a type of **follicular cell** characterized by abundant **eosinophilic, granular cytoplasm** due to numerous mitochondria. - While they can form tumors (**Hürthle cell adenoma or carcinoma**), these tumors do not typically feature cell nests or amyloid stroma. *Follicular cells* - Follicular cells are the most common cell type in the thyroid and are the origin of most thyroid cancers, including **papillary** and **follicular carcinomas** [4]. - These tumors generally do not present with the characteristic **amyloid stroma** and cell nests described in the question. **References:** [1] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. The Endocrine System, pp. 1102-1103. [2] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. Common Clinical Problems From Liver And Biliary System Disease, pp. 430-431. [3] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. Common Clinical Problems From Liver And Biliary System Disease, pp. 428-429. [4] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. Common Clinical Problems From Liver And Biliary System Disease, pp. 429-430.
Question 6: Molecular genetic testing is used to detect all of the following except?
- A. Deletion
- B. Translocation (Correct Answer)
- C. Amplification
- D. Point mutation
Explanation: ***Translocation*** - **Translocations** are chromosomal rearrangements that were historically detected primarily by **cytogenetic methods** (karyotyping, conventional FISH), rather than by traditional molecular genetic testing methods focused on DNA sequencing [3]. - While modern molecular techniques like **RT-PCR for fusion transcripts** (e.g., BCR-ABL), **NGS-based fusion detection**, and **targeted breakpoint sequencing** can now detect translocations, the classic distinction is that translocations involve large-scale structural chromosomal changes better visualized by cytogenetics [2], [3]. - In the traditional classification, molecular genetic testing referred primarily to **sequence-based methods** (PCR, Sanger sequencing) that detect smaller-scale DNA changes rather than gross chromosomal rearrangements. *Deletion* - **Deletions** are readily detected by molecular genetic testing using PCR, Sanger sequencing, MLPA (Multiplex Ligation-dependent Probe Amplification), and NGS [5]. - These techniques identify missing DNA sequences by analyzing changes in fragment size, read depth, or absence of expected amplification products [2], [5]. *Amplification* - **Amplification** (increased gene copy number) is detected by molecular methods including **quantitative PCR (qPCR)**, **digital PCR**, and **NGS-based copy number analysis** [4]. - These techniques quantify gene copy numbers to identify amplifications like HER2 amplification in breast cancer. *Point mutation* - **Point mutations** are the primary target of classic molecular genetic testing [1]. - Detected by **Sanger sequencing**, **allele-specific PCR**, **NGS panels**, and other sequence-based methods that identify single nucleotide changes in DNA [1], [2]. **References:** [1] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Genetic Disorders, p. 185. [2] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Genetic Disorders, pp. 185-186. [3] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Neoplasia, pp. 342-343. [4] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Neoplasia, p. 344. [5] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Genetic Disorders, pp. 183-184.
Question 7: To replenish the inventory, blood banks routinely issue blood packets which are close to the expiry date. Which of the following will be the closest to expiry date, according to the anticoagulant used and the method of storage of the packet:
- A. ACD, 14 days
- B. SAGM, 25 days with irradiation (Correct Answer)
- C. CPDA, 27 days
- D. SAGM, 35 days
Explanation: ***SAGM, 25 days with irradiation*** - **SAGM** (Saline-Adenine-Glucose-Mannitol) is an additive solution that extends the storage life of red blood cells significantly. - While SAGM usually allows for storage up to **42 days**, if the blood is **irradiated**, the shelf life is reduced to **28 days from collection or 14 days from irradiation, whichever is sooner**. Given the options, 25 days with irradiation falls within this reduced shelf life, making it the closest to expiry among the provided choices (implying the Irradiation was done earlier). *ACD, 14 days* - **ACD** (Acid Citrate Dextrose) is an older anticoagulant primarily used for apheresis products. - Red blood cells collected with ACD typically have a maximum storage duration of **21 days**. While 14 days is within this, other anticoagulant-additive combinations offer longer storage, and this option is not the closest to expiry when considering the maximum allowed. *CPDA, 27 days* - **CPDA** (Citrate Phosphate Dextrose Adenine) provides a standard shelf life of **35 days** for red blood cells. - While 27 days represents blood that has been stored for a significant period, it still has 8 more days until its maximum expiry, making it less "close to expiry" than the irradiated SAGM option which would expire sooner. *SAGM, 35 days* - **SAGM** (Saline-Adenine-Glucose-Mannitol) allows for the storage of red blood cells for up to **42 days** from collection. - At 35 days, a unit stored in SAGM still has 7 days until its maximum expiry date (if not irradiated), making it less "close to expiry" than a unit that had been irradiated.
Question 8: A 12-year-old Caucasian male presents with his mother to the pediatrician’s office complaining of right thigh pain. He reports that he has noticed slowly progressive pain and swelling over the distal aspect of his right thigh over the past two months. He denies any recent trauma to the area and his temperature is 100.9°F (38.3°C). On exam, there is swelling and tenderness overlying the distal right femoral diaphysis. Laboratory evaluation is notable for an elevated white blood cell (WBC) count and elevated erythrocyte sedimentation rate (ESR). A radiograph of the patient’s right leg is shown. Biopsy of the lesion demonstrates sheets of monotonous small round blue cells with minimal cytoplasm. Which of the following genetic mutations is most likely associated with this patient’s condition?
- A. t(11;22) (Correct Answer)
- B. RB1 inactivation
- C. TP53 inactivation
- D. t(8;14)
- E. t(X;18)
Explanation: ***t(11;22)*** - The clinical presentation of a **12-year-old male** with progressive **thigh pain and swelling**, fever, elevated WBC and ESR, a radiograph showing a bone lesion [1], and a biopsy revealing **small round blue cells with minimal cytoplasm**, is highly suggestive of **Ewing sarcoma** [2]. - **Ewing sarcoma** is characterized by the **t(11;22)(q24;q12) chromosomal translocation**, which fuses the *EWSR1* gene with the *FLI1* gene, leading to the formation of a chimeric transcription factor. *t(X;18)* - The **t(X;18) translocation** is the characteristic genetic abnormality of **synovial sarcoma**, another soft tissue malignancy. - While synovial sarcoma can also present in young patients, it typically affects older adolescents and young adults, and the histology differs from the small round blue cell pattern seen in Ewing sarcoma [2]. *RB1 inactivation* - **RB1 gene inactivation** is centrally involved in the pathogenesis of **retinoblastoma**, a childhood eye cancer. - It also plays a role in various other cancers, such as **osteosarcoma** [3] and small cell lung cancer, but its primary association is not with Ewing sarcoma. *TP53 inactivation* - **TP53 gene inactivation** is a common event in a wide range of human cancers, as *TP53* is a critical **tumor suppressor gene**. - While *TP53* mutations can be found in some sarcomas, it is not the defining or most likely specific genetic mutation for **Ewing sarcoma**. *t(8;14)* - The **t(8;14)(q24;q32) chromosomal translocation** is the characteristic genetic abnormality found in **Burkitt lymphoma**. - This translocation leads to the **c-MYC proto-oncogene** being placed near the immunoglobulin heavy chain locus, promoting its overexpression, which is unrelated to Ewing sarcoma. **References:** [1] Cross SS. Underwood's Pathology: A Clinical Approach. 6th ed. Common Clinical Problems From Osteoarticular And Connective Tissue Disease, pp. 671-672. [2] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Bones, Joints, and Soft Tissue Tumors, pp. 1204-1205. [3] Kumar V, Abbas AK, et al.. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Bones, Joints, and Soft Tissue Tumors, pp. 1200-1202.