Hemoglobin & O₂ Binding - The Oxygen Taxi
- Structure: Tetrameric protein (2 α, 2 β chains), each with a heme group containing iron ($Fe^{2+}$).
- States & Affinity:
- T (taut) state: Low O₂ affinity (deoxyhemoglobin).
- R (relaxed) state: High O₂ affinity (oxyhemoglobin).
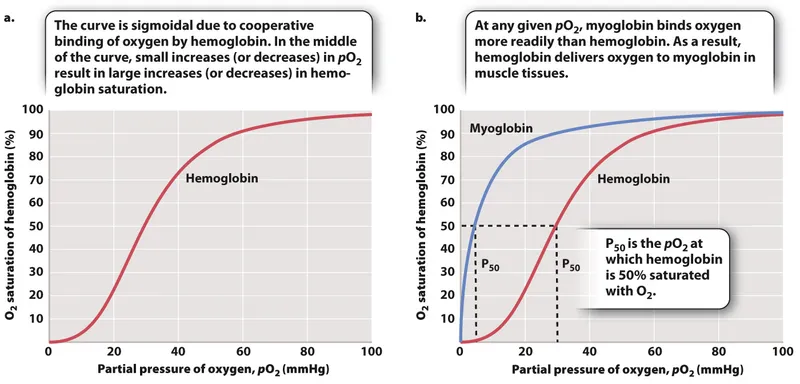
- Cooperative Binding: O₂ binding to one heme group increases the O₂ affinity of the other hemes. This positive cooperativity results in a sigmoidal-shaped oxygen-hemoglobin dissociation curve.
- Key Equation: $O₂ \text{ content} = (1.34 \times [Hb] \times SaO₂) + (0.003 \times PaO₂)$.
⭐ In the lungs, high $PO₂$ favors the R state (loading O₂). In peripheral tissues, lower $PO₂$ and allosteric regulators (like 2,3-BPG) favor the T state, facilitating O₂ unloading.
O₂-Hb Dissociation Curve - The Great Let-Go
-
Shape: Sigmoidal curve reflects cooperative binding; Hb's affinity for $O₂$ increases as each $O₂$ molecule binds.
-
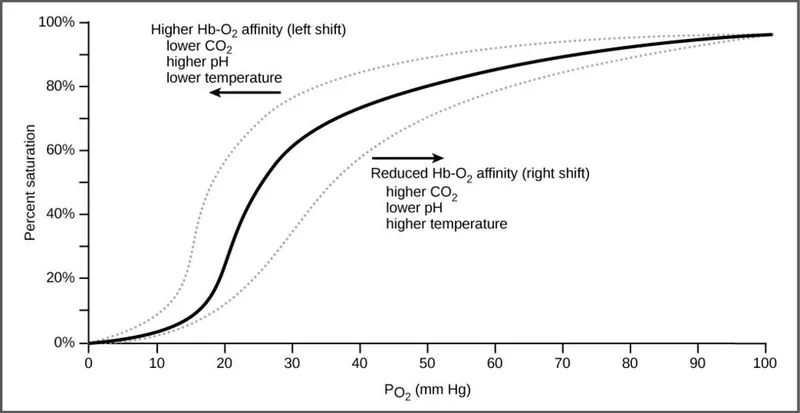
$P_{50}$: The $PO₂$ at which hemoglobin is 50% saturated. Normal value is ~27 mmHg. An ↑ $P_{50}$ indicates ↓ affinity.
-
Right Shift (↓ Affinity → $O₂$ Release)
- Favors the "unloaded" taut (T) form of Hb, releasing $O₂$ to tissues.
- 📌 CADET, face Right!
- ↑ CO₂
- ↑ Acid (↓pH) - Bohr Effect
- ↑ 2,3-DPG (BPG)
- ↑ Exercise
- ↑ Temperature
-
Left Shift (↑ Affinity → $O₂$ Hoarding)
- Favors the "loaded" relaxed (R) form of Hb.
- Caused by: ↓ Temp, ↓ 2,3-BPG, ↓ $CO₂$, ↑ pH.
- Also seen with Fetal Hb (HbF), Methemoglobin, and CO-Hb.
⭐ In CO poisoning, the curve shifts far to the left, drastically increasing Hb's affinity for $O₂$ and preventing its release to tissues. It also reduces the number of available binding sites, lowering the maximal $O₂$ saturation.
Curve Shifters - Right & Left Turns
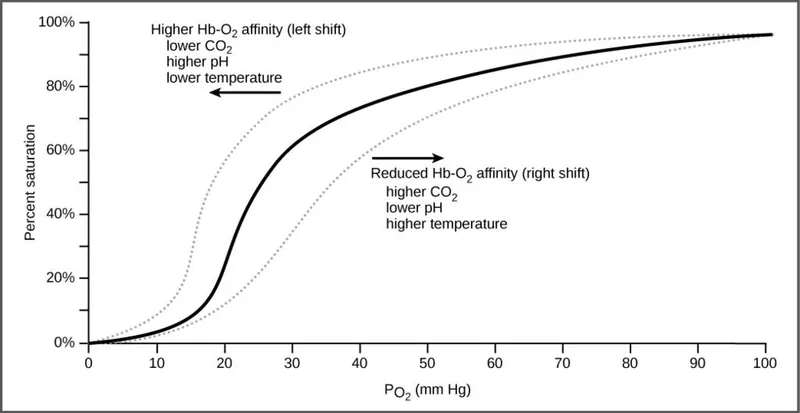
Right Shift (↓ Affinity, ↑ O₂ Unload)
- Causes:
- ↑ $CO₂$ (Bohr effect)
- ↑ Acid (↓ pH)
- ↑ 2,3-DPG
- ↑ Temperature
- Exercise
- 📌 Mnemonic: CADET, face Right! (CO₂, Acid, 2,3-DPG, Exercise, Temperature)
Left Shift (↑ Affinity, ↓ O₂ Unload)
- Causes:
- ↓ $CO₂$
- ↓ Acid (↑ pH)
- ↓ 2,3-DPG
- ↓ Temperature
- Fetal Hemoglobin (HbF)
- Carbon Monoxide (CO)
⭐ High-Yield: Fetal hemoglobin (HbF) causes a left shift because it binds 2,3-DPG poorly. This increases HbF's oxygen affinity, facilitating O₂ transport across the placenta from mother to fetus.
Clinical Correlations - Transport Under Fire
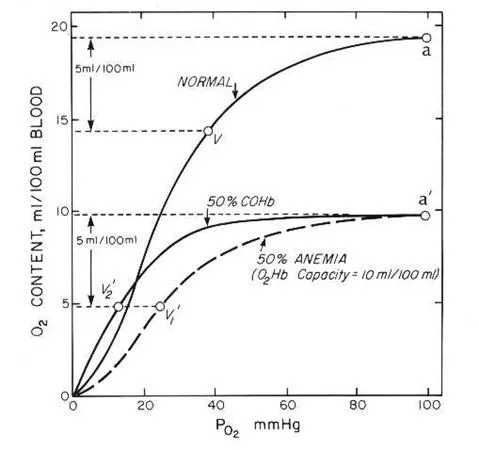
- Carbon Monoxide (CO) Poisoning
- CO has >200x the affinity for Hb than $O_2$, forming carboxyhemoglobin.
- Causes a left shift in the O2-Hb curve, impairing $O_2$ unloading.
- Presents with headaches, dizziness; classic cherry-red skin is rare.
- Treatment: 100% $O_2$; hyperbaric $O_2$.
- Methemoglobinemia
- Iron in heme is oxidized from $Fe^{2+}$ to $Fe^{3+}$.
- Causes functional anemia, cyanosis; blood appears chocolate-brown.
- Treatment: Methylene blue.
⭐ In CO poisoning, dissolved $O_2$ ($PaO_2$) remains normal, but $O_2$ saturation ($SaO_2$) is severely decreased.
High‑Yield Points - ⚡ Biggest Takeaways
- Hemoglobin (Hb) binds up to 4 O₂ molecules cooperatively, resulting in a sigmoidal O₂-dissociation curve.
- A right shift reflects decreased O₂ affinity, enhancing O₂ unloading in tissues. Key factors are ↑CO₂, ↓pH, ↑2,3-DPG, and ↑temperature.
- A left shift indicates increased O₂ affinity, impairing O₂ release. This is seen with fetal Hb and CO poisoning.
- The Bohr effect: H⁺ and CO₂ binding to Hb facilitates O₂ release.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

