Gross Pathology - Shrinking Brain Blues
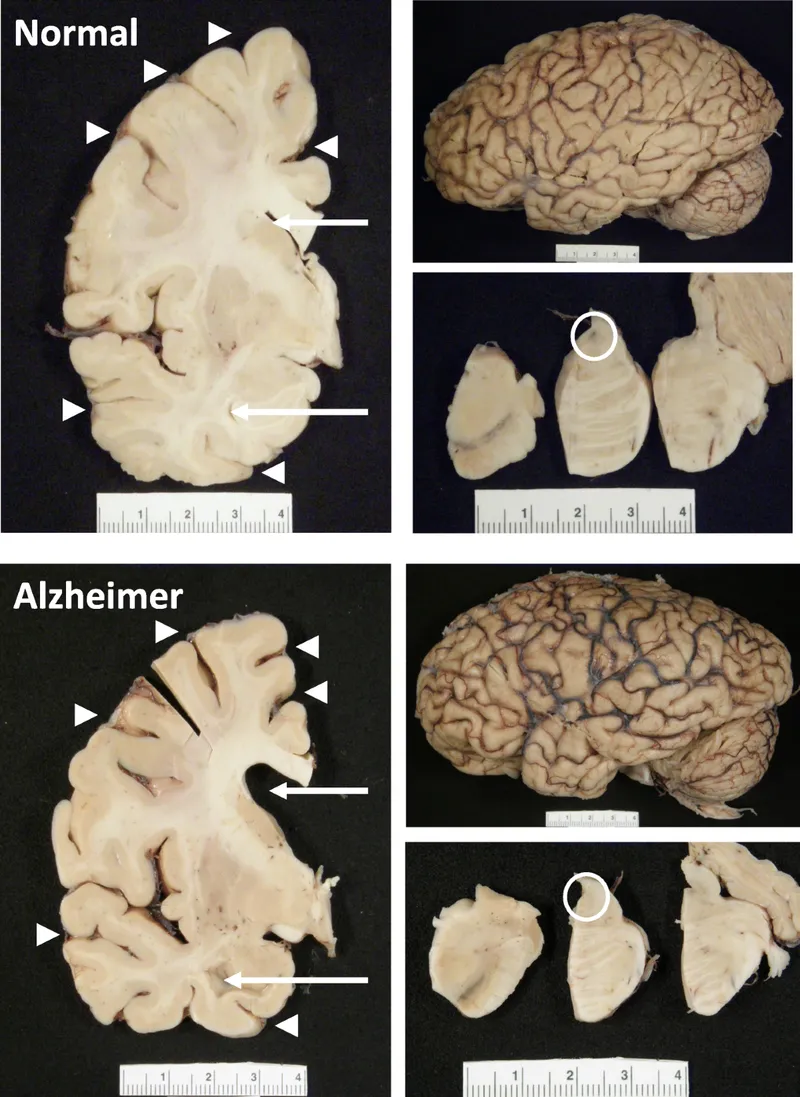
- Generalized cortical atrophy: The brain shrinks, leading to:
- Widened sulci: The grooves on the brain's surface become broader.
- Narrowed gyri: The folds of the brain become thinner.
- Hydrocephalus ex vacuo: Compensatory enlargement of ventricles as brain parenchyma is lost. CSF pressure remains normal.
- Hippocampal atrophy: Severe shrinkage of the hippocampus is a key feature.
⭐ Hippocampal atrophy is a key early finding on MRI, correlating with the severity of memory loss.
Extracellular Pathology - Plaque Attack
- Senile (neuritic) plaques are extracellular aggregates of Amyloid-β (Aβ) peptides, primarily the toxic Aβ42 fragment, formed by enzymatic cleavage of Amyloid Precursor Protein (APP).
- Staining: Visualized with Congo Red (apple-green birefringence) and Thioflavin-S.
- Cerebral Amyloid Angiopathy (CAA): Aβ deposition in vessel walls, increasing hemorrhage risk.
⭐ Aβ42's hydrophobic nature makes it highly prone to aggregation, initiating plaque formation.
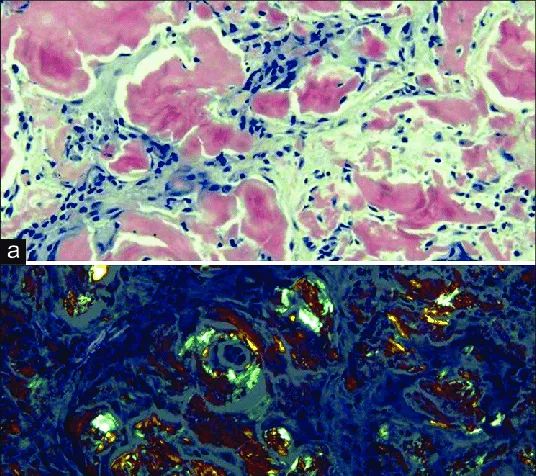
Intracellular Pathology - Tangled Tau
- Normal Function: Tau protein is an intracellular microtubule-associated protein (MAP) that provides structural stability to neuronal axons, acting like railway ties for axonal transport.
- Pathology: In Alzheimer's disease, Tau becomes pathologically hyperphosphorylated.
- This causes Tau to detach from microtubules, leading to their destabilization and disintegration.
- The abnormal Tau protein misfolds and aggregates into insoluble Paired Helical Filaments (PHFs).
- Result - NFTs: These PHFs accumulate within the neuron to form Neurofibrillary Tangles (NFTs).
- Appearance: NFTs have a classic intracytoplasmic, "flame-shaped" appearance in pyramidal neurons.
- Staining: Visualized with silver stains (Bielschowsky, Gallyas) and immunohistochemistry for Tau.

⭐ High-Yield: Tangle density and distribution (measured by Braak staging) correlate much more strongly with the severity of cognitive decline in Alzheimer's disease than amyloid plaque load.
Genetic Factors - The Gene Scene
| Onset Type | Key Genes & Loci | Notes |
|---|---|---|
| Late-Onset (>95%) | ApoE4 allele on Chr 19 | ↑ Risk for sporadic AD |
| (Sporadic) | ApoE2 allele on Chr 19 | ↓ Protective |
| Early-Onset (<5%) | APP on Chr 21 | Amyloid Precursor Protein |
| (Familial) | PSEN1 on Chr 14 | Presenilin 1 (most common) |
| PSEN2 on Chr 1 | Presenilin 2 |
⭐ The ApoE4 allele is the strongest genetic risk factor for late-onset AD.
High‑Yield Points - ⚡ Biggest Takeaways
- Core pathology involves extracellular Aβ-amyloid plaques (derived from APP) and intracellular neurofibrillary tangles of hyperphosphorylated tau.
- Gross anatomy shows diffuse cortical atrophy, most prominent in the hippocampus and temporoparietal lobes.
- Cerebral amyloid angiopathy (CAA) is a frequent co-pathology, elevating the risk of lobar hemorrhage.
- ApoE4 allele is the most significant genetic risk factor for late-onset AD.
- Early-onset familial AD is linked to mutations in APP, PSEN1, and PSEN2.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

