Mechanism of Action - The Nerve Blockers
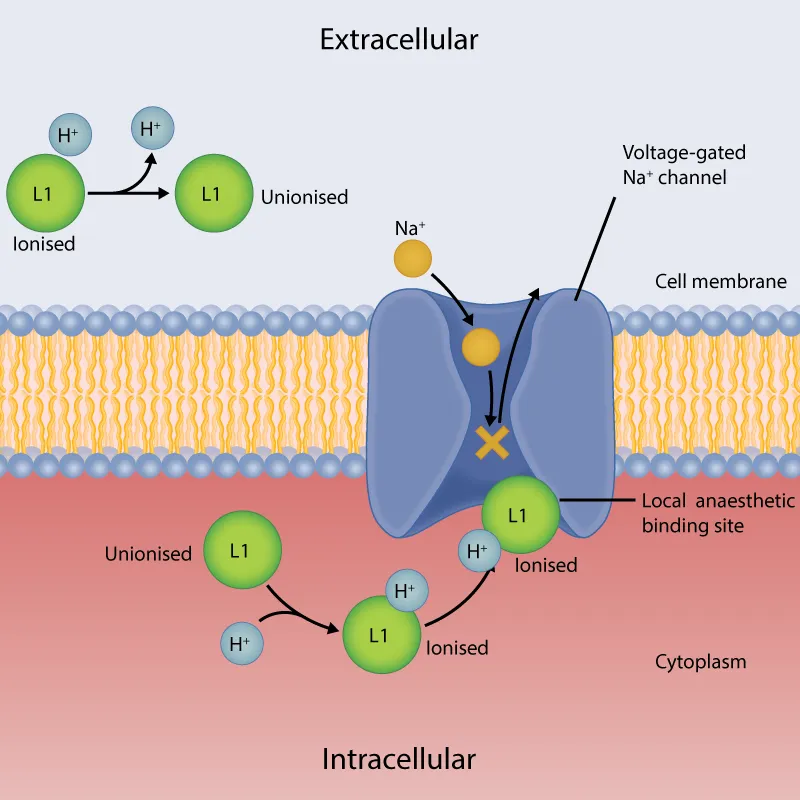
- Reversibly block nerve conduction by binding to voltage-gated sodium ($Na^+$) channels from inside the axon, preventing depolarization.
- State-Dependent Blockade: Higher affinity for open or inactivated channels than for resting channels.
- pH Influence: ↓ efficacy in acidic/infected tissue; the ionized, charged form cannot easily cross the nerve membrane to reach its target.
⭐ Use-Dependent Blockade: Nerves that are more active (higher frequency of depolarization) are blocked more rapidly. The more the channel opens, the more access the drug has.
Amides vs. Esters - A Tale of Two 'i's
- Amides: Have two 'i's in their name (e.g., Lidocaine, Bupivacaine, Ropivacaine).
- Metabolized by the liver (CYP450).
- Longer acting and more stable.
- Allergic reactions are rare.
- Esters: Have only one 'i' in their name (e.g., Procaine, Cocaine, Benzocaine).
- Metabolized in plasma by pseudocholinesterases.
- Shorter acting and less stable.
- Higher allergy potential due to para-aminobenzoic acid (PABA) metabolite.
📌 Mnemonic: Amides have two 'i's; Esters have one.
⭐ Allergic reactions to one ester anesthetic indicate likely sensitivity to other esters, but cross-reactivity with amides is rare.
Clinical Use & Potency - The Numbing Game
- Applications: Used for topical anesthesia, infiltration (e.g., suturing), peripheral nerve blocks, and spinal/epidural anesthesia.
- Physicochemical Properties:
- Potency ∝ Lipid Solubility (more lipid-soluble drugs cross membranes easier).
- Duration ∝ Protein Binding (more binding keeps drug at Na+ channel).
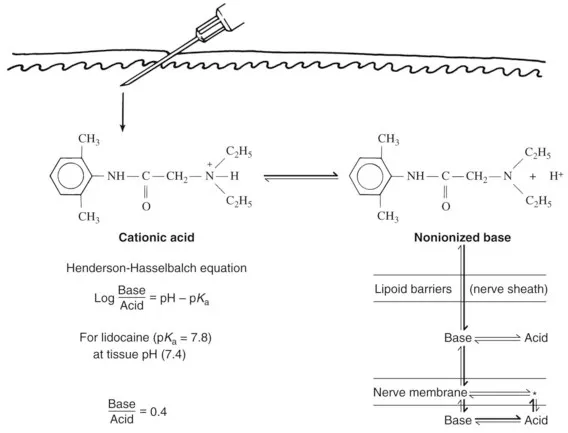
- pH & Onset:
- Anesthetics are weak bases. The un-ionized form penetrates the nerve sheath.
- Infection → acidic tissue (↓pH) → traps anesthetic in ionized form → ↓efficacy.
- Vasoconstrictors (e.g., Epinephrine): Added to prolong duration & reduce systemic toxicity.
⭐ Order of Blockade: Small, myelinated fibers (pain, temp) are blocked first, followed by touch/pressure, and finally motor function.
Systemic Toxicity - When Good Drugs Go Bad
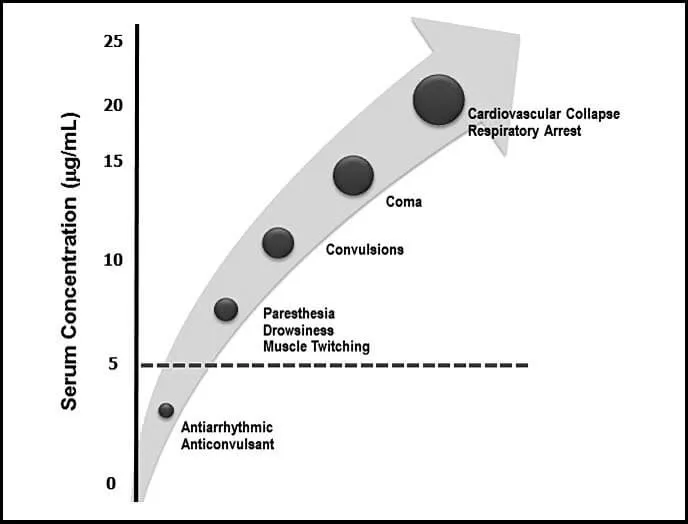
- Results from excessive plasma concentration, often via accidental intravascular injection or rapid absorption. CNS effects typically precede cardiovascular toxicity.
- CNS Manifestations (Biphasic):
- Early (Excitation): Tinnitus, perioral numbness, metallic taste, agitation, muscle twitching, leading to seizures.
- Late (Depression): Drowsiness, coma, respiratory arrest.
- Cardiovascular Effects:
- Hypotension, arrhythmias (VT/VF), bradycardia.
- Myocardial depression (↓ contractility).
- Management: Airway support, benzodiazepines for seizures, and IV lipid emulsion therapy.
⭐ Bupivacaine is the most cardiotoxic local anesthetic due to its high potency and strong binding to cardiac Na+ channels, making resuscitation challenging.
High‑Yield Points - ⚡ Biggest Takeaways
- Local anesthetics reversibly block voltage-gated Na+ channels, with increased affinity for activated/inactivated states.
- Blockade is most effective on small, myelinated fibers, leading to loss of pain and temperature first.
- Amides (e.g., lidocaine) are metabolized by the liver; esters (e.g., procaine) risk allergic reactions via PABA.
- Co-administration with epinephrine causes vasoconstriction, prolonging anesthesia and reducing systemic absorption.
- Bupivacaine carries a high risk of severe cardiotoxicity.
- Treat systemic toxicity with intravenous lipid emulsion.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

