HPV Overview - The Cancer Culprit
- Virus: Double-stranded, non-enveloped DNA virus; over 150 types.
- Transmission: Primarily sexual contact (including skin-to-skin).
- Oncogenic (High-Risk) Strains: 16, 18 cause ~70% of cervical cancers & most other HPV-related cancers (anal, oropharyngeal).
- Non-Oncogenic (Low-Risk) Strains: 6, 11 cause ~90% of anogenital warts (condylomata acuminata).
- Pathogenesis: Integration into host genome. Viral proteins inactivate tumor suppressors:
- E6 → degrades p53
- E7 → inhibits Rb (Retinoblastoma protein)
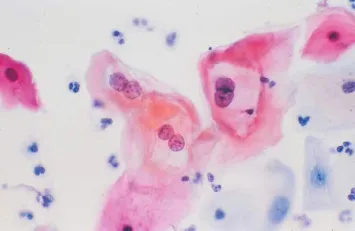
⭐ High-Yield: Koilocytes on Pap smear are pathognomonic; these are squamous epithelial cells with a wrinkled, "raisinoid" nucleus and a perinuclear halo.
HPV Vaccine - Gardasil's Guard
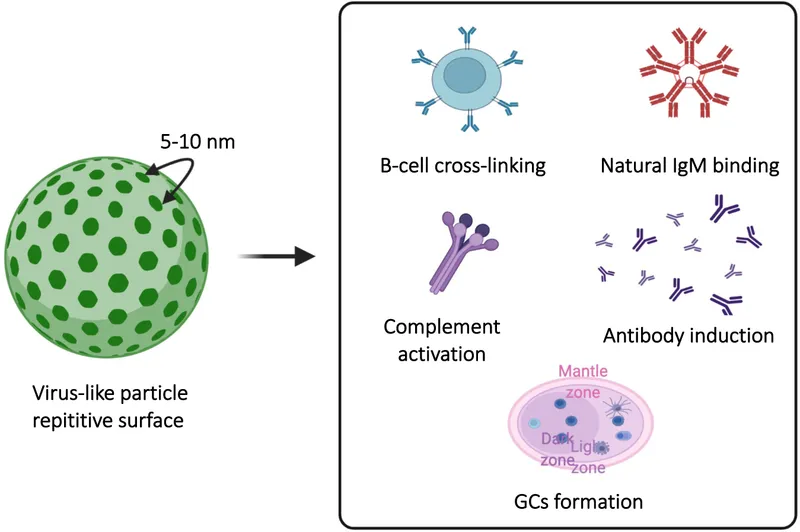
- Type: Recombinant vaccine with L1 major capsid protein, forming non-infectious virus-like particles (VLPs).
- Coverage (Gardasil-9): Protects against 9 HPV types.
- Low-risk (Genital Warts): 6, 11
- High-risk (Cancers - cervical, anal, oropharyngeal): 16, 18, 31, 33, 45, 52, 58
- Administration Schedule:
- Ages 9-14: 2 doses at 0 and 6-12 months.
- Ages 15-26 or immunocompromised: 3 doses at 0, 1-2, and 6 months.
⭐ The vaccine is prophylactic, not therapeutic. It prevents initial infection but does not clear existing DNA or treat HPV-related disease.
📌 HPV types 16 and 18 are implicated in ~70% of cervical cancers.
Dosing & Schedule - The Jab Plan
- Routine Vaccination: Age 11-12 years (can start at age 9).
- Catch-up Vaccination: Recommended for all through age 26.
- Shared Clinical Decision-Making: For adults age 27-45 not adequately vaccinated.
⭐ For adults aged 27-45, vaccination is not routinely recommended but based on shared clinical decision-making, as new HPV infections are less common and the vaccine has lower efficacy in this group.
Efficacy & Safety - Worth a Shot?
- Efficacy: Near 100% effective at preventing cervical precancer and cancer caused by vaccine-targeted HPV types (e.g., HPV 16, 18).
- Also prevents genital warts (HPV 6, 11).
- Reduces rates of anogenital and oropharyngeal cancers.
- Safety Profile: Very safe.
- Common side effects: mild injection site reactions (pain, redness), headache.
- 📌 Post-vaccine syncope is common in adolescents; observe for 15 minutes.
⭐ Large-scale studies show a >85% reduction in cervical cancer prevalence among women vaccinated before age 17.

High‑Yield Points - ⚡ Biggest Takeaways
- The Gardasil 9 vaccine targets HPV types 6, 11, 16, 18, and five other high-risk strains.
- It prevents cervical, anogenital, and oropharyngeal cancers (linked to HPV 16, 18) and genital warts (from HPV 6, 11).
- It is a recombinant, virus-like particle (VLP) vaccine and is not infectious.
- Recommended for all adolescents at age 11-12, with catch-up vaccination up to age 26.
- The vaccine is prophylactic and does not treat existing HPV infections or associated diseases.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

