WNT Pathway - The Key Players
- Ligand: WNT proteins (secreted glycoproteins).
- Receptors: Frizzled (FZD) family (transmembrane proteins) & LRP5/6 (co-receptor).
- Cytoplasmic Mediators: Dishevelled (Dsh), β-catenin, and the destruction complex (Axin, APC, GSK-3β).
- Nuclear Effectors: TCF/LEF family of transcription factors.
⭐ High-Yield: The Adenomatous Polyposis Coli (APC) gene is a tumor suppressor. Its inactivation is a key step in colorectal cancer, leading to constitutive WNT pathway activation due to β-catenin accumulation.
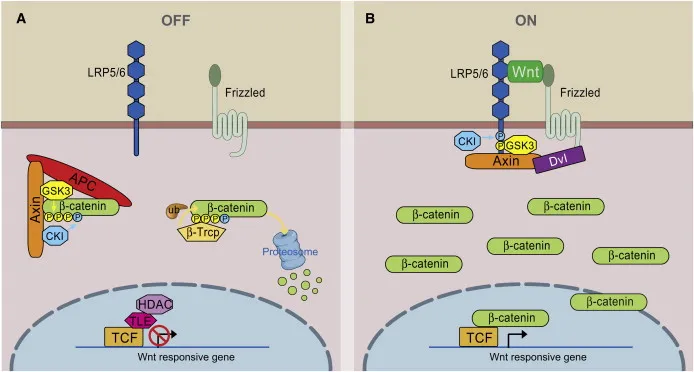
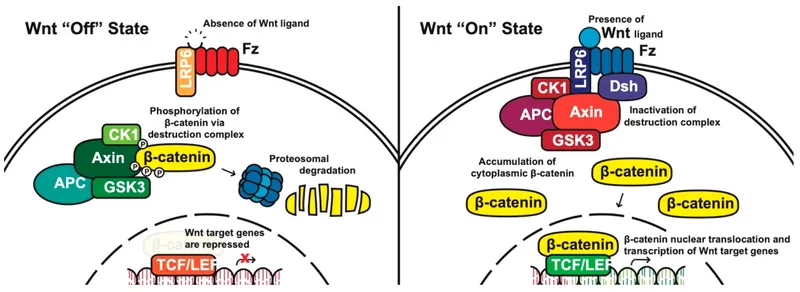
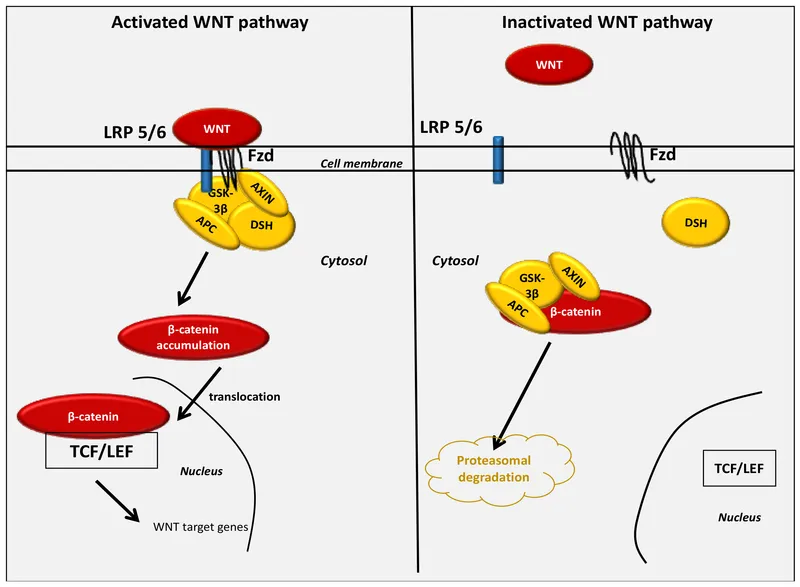
Canonical Pathway - The 'Off' Switch
- No WNT Ligand: In the absence of WNT, the Frizzled (Fz) receptor and its co-receptor LRP5/6 are inactive.
- Active Destruction Complex: A cytoplasmic protein assembly-comprising Axin, Adenomatous Polyposis Coli (APC), Glycogen Synthase Kinase 3β (GSK-3β), and Casein Kinase 1 (CK1)-is active.
- β-Catenin Phosphorylation: This complex sequentially phosphorylates β-catenin, tagging it for destruction.
- Proteasomal Degradation: Tagged β-catenin is ubiquitinated and rapidly degraded by the proteasome, keeping cytosolic levels low.
- Gene Repression: In the nucleus, TCF/LEF transcription factors are bound by the Groucho co-repressor, silencing WNT target genes.
⭐ Germline mutations in the APC gene, a tumor suppressor, cause Familial Adenomatous Polyposis (FAP), leading to a near 100% risk of colorectal cancer if untreated.
Canonical Pathway - The 'On' Switch
- Activation: Wnt ligand binds to the Frizzled (Fz) and LRP5/6 co-receptor complex.
- Signal Transduction:
- Recruits Dishevelled (Dsh).
- Dsh inhibits the β-catenin "destruction complex" (Axin, APC, GSK-3β).
- β-catenin Accumulation:
- Unphosphorylated β-catenin is no longer degraded.
- Cytoplasmic levels ↑, leading to nuclear translocation.
- Gene Transcription:
- In the nucleus, β-catenin displaces the Groucho repressor from TCF/LEF transcription factors.
- Activates target genes for cell proliferation & differentiation.
⭐ Mutations in the APC gene, a core part of the destruction complex, are central to most sporadic colorectal cancers. This causes constant Wnt activation, driving tumor growth.
Clinical Tie-Ins - When WNT Goes Wild
- Oncogenesis: Aberrant WNT activation is a key driver in many cancers. The pathway's failure to switch off leads to unchecked cell proliferation and tumor formation.
- Colorectal Cancer (CRC): The most classic association.
- Familial Adenomatous Polyposis (FAP): An autosomal dominant condition caused by a germline mutation in the APC gene. This leads to thousands of polyps and a near 100% risk of CRC if the colon is not removed.
- Sporadic CRC: Acquired mutations in APC or β-catenin are found in the vast majority of cases.
⭐ The APC gene is a quintessential tumor suppressor. Its inactivation is the gatekeeping event in the adenoma-carcinoma sequence for most colorectal cancers.
- Other Malignancies: WNT dysregulation is also implicated in hepatocellular carcinoma, medulloblastoma, breast, and ovarian cancers.
- Therapeutic Targets: The pathway is a major focus for drug development, with agents targeting various components like Porcupine (WNT secretion) and β-catenin.
High‑Yield Points - ⚡ Biggest Takeaways
- WNT ligands bind to Frizzled (Fz) and LRP coreceptors, initiating the cascade.
- Without WNT, a destruction complex (containing APC, Axin, GSK3β) phosphorylates and degrades β-catenin.
- WNT signaling inhibits the destruction complex, causing β-catenin to accumulate in the cytoplasm.
- Accumulated β-catenin translocates to the nucleus, acting as a transcriptional coactivator.
- It promotes genes involved in cell proliferation, polarity, and fate determination.
- Mutations in APC lead to unregulated β-catenin, causing familial adenomatous polyposis (FAP).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

