Hormonal Milieu - The Hunger Hormones
- Primary Metabolic Switch: The defining change is the ↓ Insulin : Glucagon ratio.
- Key Hormonal Shifts:
- ↓ Insulin: Due to low blood glucose.
- ↑ Glucagon: Secreted by pancreatic α-cells.
- ↑ Epinephrine: Released from adrenal medulla.
- ↑ Cortisol: Secreted to promote gluconeogenesis and lipolysis.
⭐ The Insulin:Glucagon ratio, not the absolute level of either hormone alone, is the most critical factor governing the metabolic transition from the fed to the fasting state.
Hepatic Glycogenolysis - Glycogen's Last Stand
- Timeframe: Primary glucose source for the first 12-24 hours of fasting.
- Process: Liver glycogenolysis bridges the gap until gluconeogenesis fully activates.
- Key Enzyme: Glycogen phosphorylase (activated by ↑ glucagon & epinephrine).
- Liver vs. Muscle Glycogen:
- Liver: Has Glucose-6-phosphatase ($G6Pase$) to release free glucose into blood for brain/RBCs.
- Muscle: Lacks $G6Pase$; glycogen provides fuel for muscle contraction only. 📌 Muscle is "selfish."
⭐ The liver's expression of Glucose-6-phosphatase is the key distinction allowing it to buffer blood glucose, a function muscle cannot perform.
Gluconeogenesis - Desperate Glucose Measures
Active from ~24 hours to ~3-5 days. The liver, and later kidneys, must synthesize new glucose as glycogen depletes to fuel the brain and RBCs.
- Primary Substrates:
- Alanine: From muscle protein breakdown (Glucose-Alanine Cycle).
- Lactate: From RBCs & muscle (Cori Cycle).
- Glycerol: From triglyceride breakdown in adipose tissue.
- Key Regulation:
- PEPCK (Phosphoenolpyruvate carboxykinase) is the key regulatory enzyme; its synthesis is induced by glucagon and cortisol. ↑
⭐ Even-chain fatty acids cannot yield net glucose because they produce only Acetyl-CoA, and the pyruvate dehydrogenase complex reaction is irreversible. Odd-chain fatty acids are an exception, yielding propionyl-CoA.
Ketosis & Sparing - The Ketone Switch
- Timeline: Kicks in after >3-5 days of starvation, marking the shift to prolonged fasting metabolism.
- Central Process: Liver shifts from gluconeogenesis to robust ketogenesis.
- Massive fatty acid oxidation from adipose tissue generates excess acetyl-CoA.
- The liver converts this acetyl-CoA into ketone bodies: acetoacetate and β-hydroxybutyrate.
- Primary Goal: Protein Sparing
- Reduces the need for gluconeogenesis from amino acid skeletons, thus preserving vital muscle tissue.
- Fuel Source Adaptation:
- Brain: Adapts to derive up to ~70% of its energy from ketone bodies.
- RBCs: Lack mitochondria and always depend exclusively on glucose.
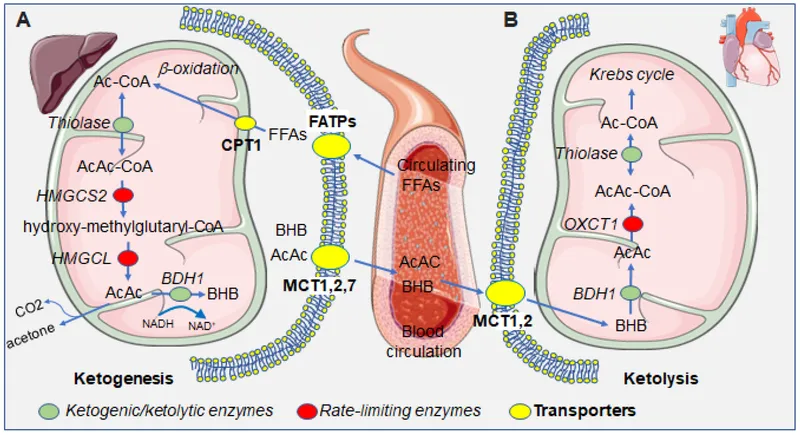
⭐ High-Yield Fact: The liver can produce ketone bodies but cannot use them for energy because it lacks the enzyme thiophorase (Succinyl-CoA-Acetoacetate CoA Transferase). This enzyme is present in extrahepatic tissues like the brain, allowing them to convert ketones back to acetyl-CoA.
- In early starvation, hepatic glycogenolysis is the primary source of glucose, depleting within ~24 hours.
- The body then shifts to gluconeogenesis, using substrates like lactate, alanine, and glycerol.
- Fatty acid oxidation becomes the main energy source for most tissues.
- The liver produces ketone bodies, which the brain starts using after 2-3 days to spare glucose.
- Muscle protein catabolism decreases significantly to preserve lean body mass.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

