Glucose Homeostasis - The Sweet Balance
-
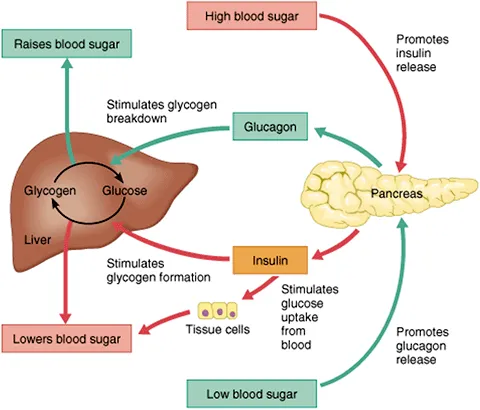
Insulin (Pancreatic β-cells): The "storage" hormone. Released with ↑ blood glucose. It drives glucose into muscle and fat cells (via GLUT4), promotes glycogen synthesis in the liver, and stimulates fat storage. Its net effect is to lower blood glucose.
-
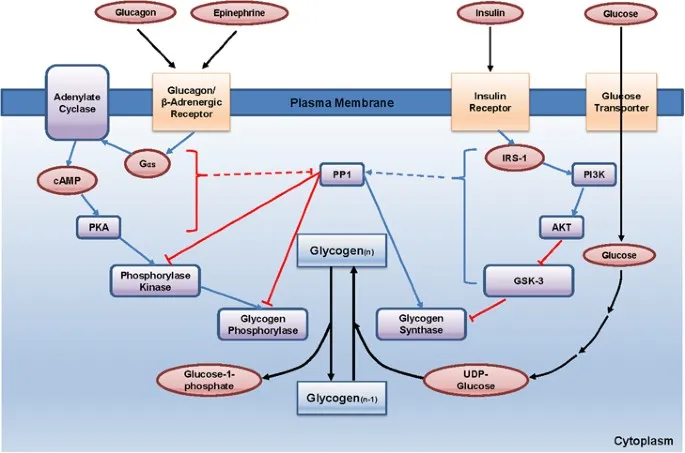
Glucagon (Pancreatic α-cells): The "release" hormone. Secreted during fasting or with ↓ blood glucose. It primarily targets the liver, stimulating glycogenolysis and gluconeogenesis to raise blood glucose.
⭐ In Type 1 Diabetes, the absence of insulin leads to unopposed glucagon action, a key driver of diabetic ketoacidosis (DKA).
Fed State - Insulin's In Charge
Following a meal, elevated blood glucose stimulates pancreatic β-cells to release insulin, the primary anabolic hormone.
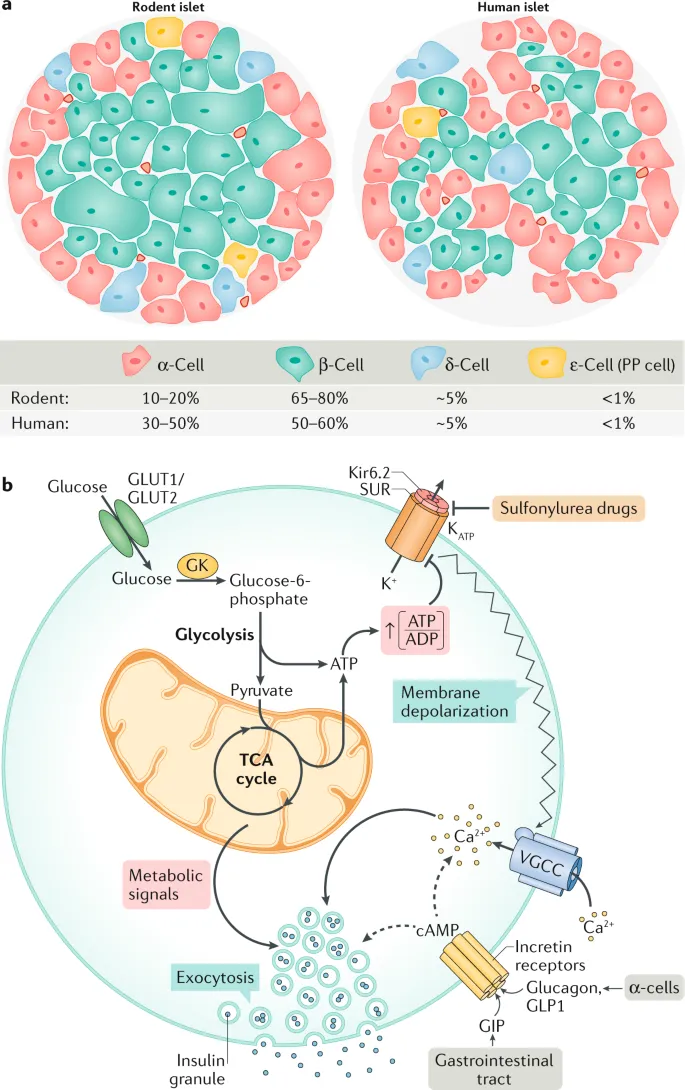
- β-Cell Insulin Secretion Mechanism:
- Glucose enters β-cells via insulin-independent GLUT2 transporters.
- Metabolism ↑ ATP, increasing the cellular ATP/ADP ratio.
- This closes ATP-sensitive K⁺ (KATP) channels, preventing K⁺ efflux.
- The membrane depolarizes, opening voltage-gated Ca²⁺ channels.
- Ca²⁺ influx triggers exocytosis of insulin storage vesicles.
- Key Anabolic Actions:
- Muscle/Adipose: Stimulates GLUT4 translocation to the membrane, ↑ glucose uptake.
- Liver/Muscle: ↑ Glycogen synthesis.
- Adipose Tissue: ↑ Lipogenesis and triglyceride storage.
⭐ C-peptide is secreted in equimolar amounts with endogenous insulin; its level can differentiate type 1 diabetes from type 2 and factitious hypoglycemia.
Fasting State - Glucagon's Gambit
- Trigger: Hypoglycemia (↓ blood glucose < 70 mg/dL) stimulates pancreatic α-cells to release glucagon.
- Primary Action (Liver): Glucagon acts on hepatocytes to ↑ blood glucose via:
- Glycogenolysis (Rapid): The immediate breakdown of stored glycogen.
- Gluconeogenesis (Sustained): Synthesis of new glucose from amino acids (e.g., alanine), lactate, and glycerol. This process is vital in prolonged fasting.
- Other Counter-Regulatory Hormones: Epinephrine, cortisol, and growth hormone provide synergistic or backup effects to maintain glucose levels during stress or extended fasting.
⭐ High-Yield: Glucagon inhibits glycolysis by decreasing fructose-2,6-bisphosphate, which simultaneously stimulates gluconeogenesis.
Glucose Gatekeepers - The GLUT Family
| Transporter | Key Locations | Insulin-Dependence | Key Fact / $K_m$ |
|---|---|---|---|
| GLUT1 | Erythrocytes (RBCs), brain (BBB), cornea, placenta. | No | Low $K_m$ (~1 mM). Constant, basal glucose uptake for tissues with high, continuous need. |
| GLUT2 | Liver, pancreatic β-cells, renal tubules, small intestine. | No | High $K_m$ (~15 mM). Bidirectional transporter; acts as a glucose sensor in pancreas. |
| GLUT3 | Neurons, placenta, testes. | No | Very low $K_m$ (<1 mM). Highest affinity; ensures glucose supply to critical areas during hypoglycemia. |
| GLUT4 | Skeletal/cardiac muscle, adipose tissue. | Yes | Medium $K_m$ (~5 mM). Insulin stimulates translocation from intracellular vesicles to the cell surface. 📌 GLUT 4 is in 4-letter tissues. |
High‑Yield Points - ⚡ Biggest Takeaways
- Insulin is the primary anabolic hormone; it drives glucose into muscle and adipose tissue via GLUT4.
- Glucagon is the main catabolic hormone; it stimulates hepatic glycogenolysis and gluconeogenesis.
- Pancreatic β-cells release insulin when ↑ glucose leads to ↑ ATP, closing K+ATP channels.
- GLUT2 is insulin-independent and facilitates glucose transport in the liver and pancreas.
- Fasting state is dominated by glucagon; the fed state is dominated by insulin.
- Cortisol and epinephrine are key counter-regulatory hormones that also raise blood glucose.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

