Nomenclature & MoA - What's in a 'mab'?
A monoclonal antibody's suffix reveals its non-human protein content, which correlates with immunogenicity.
-
Nomenclature by Source:
- -omab: Murine (~0% human)
- -ximab: Chimeric (~65% human)
- -zumab: Humanized (~90% human)
- -umab: Human (100% human)
-
Mechanisms of Action:
- Direct Blockade: Bind to cell surface receptors (e.g., Trastuzumab → HER2) or ligands (e.g., Bevacizumab → VEGF).
- Immune-Mediated Killing:
- ADCC: Fc portion binds NK cells.
- CDC: Activates complement cascade.
- Payload Delivery: Deliver conjugated toxins or radioisotopes.
⭐ Higher non-human protein content (e.g., -omab, -ximab) increases the risk of infusion reactions and anti-drug antibody formation, reducing efficacy.
Key Targets & Drugs - Cancer's Off Switches
Monoclonal antibodies (-mabs) are engineered proteins that target specific antigens on cancer cells or immune cells, effectively "switching off" growth signals or "switching on" an anti-tumor immune response.
- Vascular Endothelial Growth Factor (VEGF)
- Bevacizumab: Inhibits angiogenesis. Used for colorectal, lung, and kidney cancers.
- Epidermal Growth Factor Receptor (EGFR)
- Cetuximab: Blocks signaling in colorectal, head & neck cancers.
- Human Epidermal Growth Factor Receptor 2 (HER2)
- Trastuzumab: For HER2+ breast and gastric cancers.
- CD20 Antigen
- Rituximab: Depletes B-cells in lymphomas and CLL.
- Immune Checkpoints (PD-1, CTLA-4)
- Pembrolizumab (PD-1), Ipilimumab (CTLA-4): Block immune suppression, unleashing T-cells against tumors like melanoma and lung cancer.
⭐ Trastuzumab carries a significant risk of cardiotoxicity (dilated cardiomyopathy). Always check cardiac function (ECHO) before and during treatment.
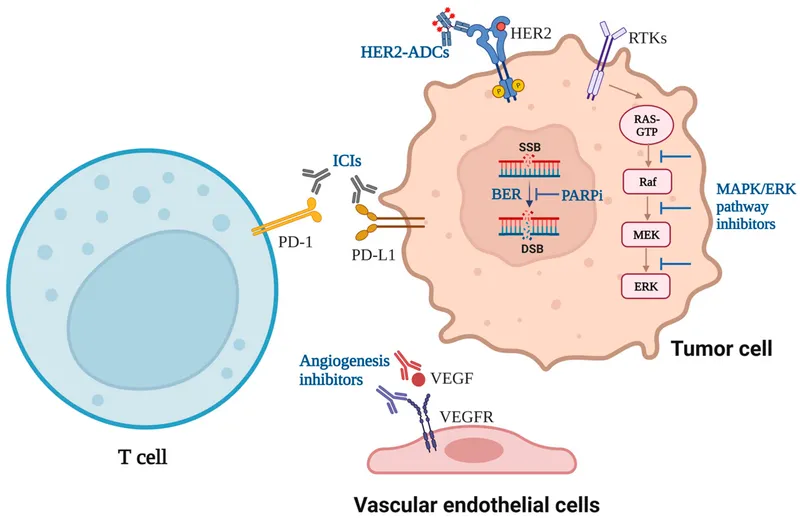
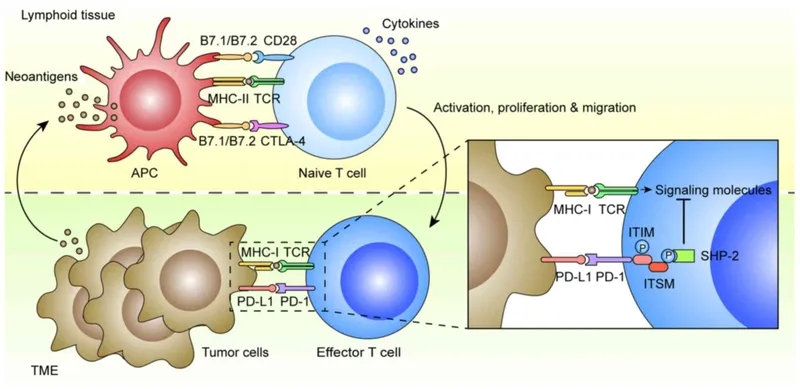
Checkpoint Inhibitors - Releasing the Brakes
- Mechanism: Blocks inhibitory signals on T-cells, "releasing the brakes" to enhance the body's anti-tumor immune response. Cancer cells exploit checkpoints like PD-1 and CTLA-4 to evade immune surveillance.
- Key Targets & Drugs:
- CTLA-4 Inhibitor: Ipilimumab
- PD-1 Inhibitors: Nivolumab, Pembrolizumab
- PD-L1 Inhibitors: Atezolizumab, Durvalumab, Avelumab
- Side Effects (Immune-Related Adverse Events - irAEs):
- Widespread inflammation: colitis, pneumonitis, hepatitis, endocrinopathies (e.g., hypophysitis), dermatitis.
- Treat with corticosteroids; high-dose for severe cases.
⭐ The unique side effects reflect the mechanism of action (autoimmunity). They can occur weeks to months after starting treatment, requiring a high index of suspicion for any new "-itis".
High‑Yield Points - ⚡ Biggest Takeaways
- Monoclonal antibodies (mAbs) use the "-mab" suffix and target specific cell surface proteins like HER2 (Trastuzumab) or CD20 (Rituximab).
- Trastuzumab's major toxicity is dose-independent cardiotoxicity.
- Bevacizumab targets VEGF, inhibiting angiogenesis, but can cause hemorrhage and impaired wound healing.
- Immune checkpoint inhibitors like Nivolumab (anti-PD-1) and Ipilimumab (anti-CTLA-4) unleash the immune system against cancer.
- Watch for immune-related adverse events (irAEs) with checkpoint inhibitors, such as colitis, hepatitis, and endocrinopathies.
- All mAbs carry a risk of infusion reactions.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

