Aminoglycoside MOA - Protein Factory Sabotage
- Bactericidal inhibitors of protein synthesis that target the 30S ribosomal subunit.
- Entry into Gram-negative bacteria is an oxygen-dependent process.
- Mechanism of Disruption:
- Interferes with the initiation complex of peptide formation.
- Induces misreading of mRNA, leading to toxic, nonfunctional proteins.
- Causes breakdown of polysomes into nonfunctional monosomes.
⭐ Because aminoglycoside uptake requires oxygen, they are characteristically ineffective against anaerobic bacteria.
📌 Mnemonic: "Buy AT 30" (Aminoglycosides & Tetracyclines act on the 30S subunit).
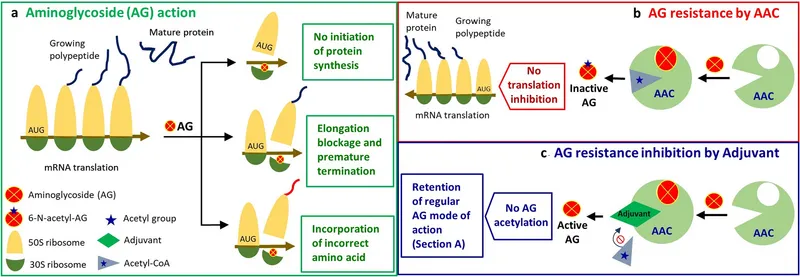
Enzymatic Modification - The Main Defense
- Most common mechanism of aminoglycoside resistance, mediated by Aminoglycoside-Modifying Enzymes (AMEs).
- These enzymes, encoded by bacterial genes, transfer chemical groups to the drug, inactivating it.
- Acetylation via Aminoglycoside Acetyltransferases (AAC)
- Phosphorylation via Aminoglycoside Phosphotransferases (APH)
- Adenylation via Aminoglycoside Nucleotidyltransferases (ANT)
- This structural change prevents the aminoglycoside from binding effectively to its target on the 30S ribosomal subunit.
- As a result, protein synthesis is no longer inhibited, and the bacterium survives.
- 📌 Mnemonic for enzymes: Always Add Phosphate or Nucleotides (Acetyl, Phospho, Nucleotidyl).
⭐ The genes encoding AMEs are often carried on plasmids and transposons, allowing for rapid dissemination of resistance through horizontal gene transfer between bacteria.
Altered Target & Transport - Keeping the Drug Out
-
Altered Ribosomal Target:
- Mechanism: Methylation of 16S rRNA on the 30S ribosomal subunit.
- Enzyme: Plasmid-encoded rRNA methyltransferases.
- Result: Steric hindrance ↓ prevents aminoglycoside binding at the A-site. This confers broad, high-level resistance to most aminoglycosides.
-
Impaired Drug Transport:
- Uptake: Requires an oxygen-dependent active transport system linked to the electron transport chain.
- Resistance Mechanisms:
- Mutation or loss of porin channels (Gram-negatives) limits entry.
- Disruption of the electron transport chain ↓ eliminates the electrical gradient needed for inner membrane transport.
⭐ Intrinsic Resistance: Anaerobes are intrinsically resistant to aminoglycosides because they lack the necessary oxygen-dependent transport system to move the drug across the cell membrane.

High‑Yield Points - ⚡ Biggest Takeaways
- The primary mechanism of resistance is enzymatic modification (e.g., acetylation, phosphorylation) by plasmid-encoded transferase enzymes.
- Resistance genes are readily transferred between bacteria on plasmids and transposons, facilitating rapid spread.
- Another key mechanism is the alteration of the ribosomal target, specifically through mutations in the 16S rRNA of the 30S subunit.
- Reduced uptake or active efflux of the drug out of the cell also confers resistance.
- This is a major clinical problem for serious aerobic Gram-negative infections, particularly Pseudomonas aeruginosa.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

