Pain Basics & Nociceptors - Ouch Origins!
- Pain (IASP): Unpleasant sensory & emotional experience linked to actual/potential tissue damage.
- Nociception: Neural encoding of noxious stimuli.
- Nociceptors: Specialized free nerve endings that detect tissue damage.
- Aδ fibers: Myelinated, fast conduction (5-30 m/s), transmit sharp, well-localized "first pain" (mechanical, thermal stimuli).
- C fibers: Unmyelinated, slow conduction (0.5-2 m/s), transmit dull, burning, poorly-localized "second pain" (polymodal: thermal, mechanical, chemical stimuli).
- Transduction: Conversion of noxious stimuli into electrical signals (action potentials) by nociceptors.
- Key ion channels involved: TRPV1 (activated by capsaicin, heat >43°C, H+), TRPA1 (irritants, cold), ASICs (acid).
- Inflammatory mediators ("Sensitizing Soup") enhance nociceptor sensitivity: Bradykinin, Prostaglandins, Serotonin (5-HT), Histamine, K+, H+, ATP, Substance P.
⭐ C fibers are polymodal and primarily responsible for the persistent, dull, aching, and poorly localized component of pain, often referred to as "second pain."
oka
Transmission & Spinal Modulation - The Relay Race
- Primary Afferents:
- Aδ fibers: Myelinated, fast, sharp, localized pain. NT: Glutamate.
- C fibers: Unmyelinated, slow, dull, diffuse pain. NTs: Glutamate, Substance P, CGRP.
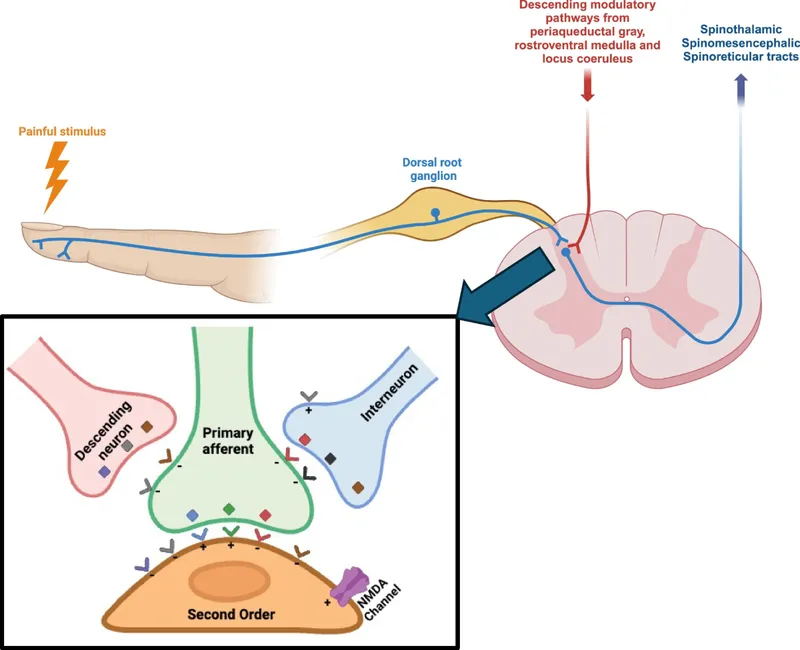
- Spinal Cord Entry & Synapse:
- Fibers enter via Dorsal Root Ganglion (DRG).
- Synapse in Dorsal Horn (Rexed Laminae I, II, V).
- Lamina II (Substantia Gelatinosa - SG): Key modulation site.
- Spinal Modulation:
- Gate Control Theory: Aβ fibers (touch) inhibit Aδ/C transmission in SG via inhibitory interneurons (GABA, enkephalins).
- Descending Inhibition: Brainstem (PAG, RVM) pathways release 5-HT & NE, inhibiting nociceptive neurons.
⭐ Substantia Gelatinosa (Rexed Lamina II) is a crucial site for pain modulation and a primary target for opioid analgesia.
Ascending Tracts & Perception - Brain's Pain Map
Pain signals ascend via spinal tracts to brain.
- Major Ascending Tracts:
- Spinothalamic Tract (STT): Primary pain pathway.
- Lateral (Neospinothalamic): Fast, sharp, localized pain (Aδ fibers). 📌 "L" for Localized. To VPL thalamus → S1/S2.
- Anterior (Paleospinothalamic): Slow, dull, diffuse pain (C fibers). 📌 "A" for Affective/Awful. To medial thalamus, RF, limbic.
- Spinoreticular Tract (SRT): Arousal, emotional. To RF → thalamus, limbic.
- Spinomesencephalic Tract (SMT): Pain modulation. To PAG.
- Spinothalamic Tract (STT): Primary pain pathway.
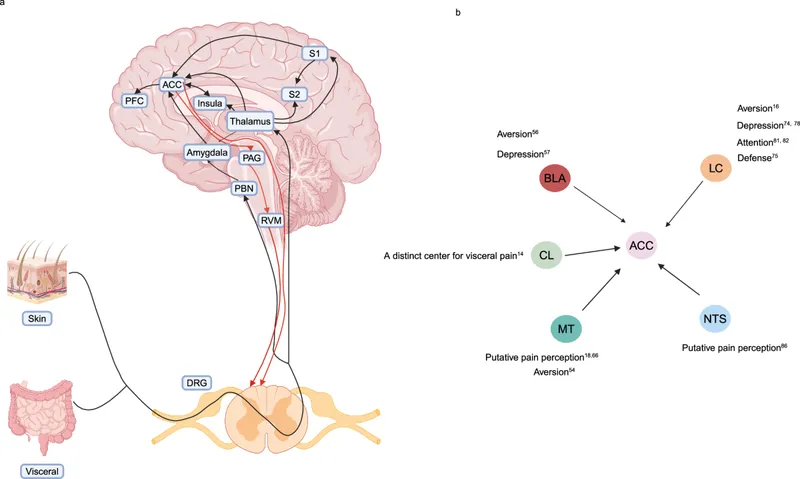
- Brain's Pain Processing Centers:
- Thalamus: Relay & processing (VPL, medial nuclei).
- Somatosensory Cortex (S1, S2): Discriminates location, intensity.
- Limbic System (ACC, Amygdala, Insula): Emotional & affective response.
⭐ The Anterior Cingulate Cortex (ACC) is key for the unpleasantness of pain.
- Prefrontal Cortex (PFC): Cognitive interpretation.
Descending Control & Sensitization - Pain's Volume Control
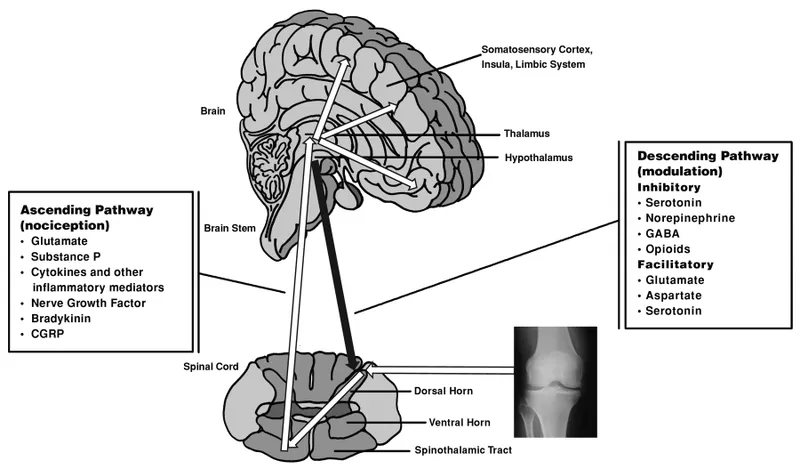
- Descending Modulation: Brain's analgesia.
- Centers: Periaqueductal Gray (PAG), Rostral Ventromedial Medulla (RVM), Locus Coeruleus (LC).
- Pathways: PAG → RVM (5-HT); LC (NE) → Dorsal Horn.
- Neurotransmitters: Serotonin, Norepinephrine, Endogenous Opioids (endorphins, enkephalins).
- Action: Inhibit nociceptive signals in dorsal horn.
- Sensitization: Pain system amplification.
- Peripheral: At nociceptor. Inflammatory mediators (PGs, bradykinin) → ↓ threshold, primary hyperalgesia.
- Central: In CNS (dorsal horn). "Wind-up"; NMDA receptor (glutamate) activation, ↑ intracellular $Ca^{2+}$. Results in secondary hyperalgesia, allodynia.
⭐ Central sensitization, driven by NMDA receptor activation and ↑ intracellular $Ca^{2+}$, is key in chronic pain, causing widespread hypersensitivity like allodynia.
High‑Yield Points - ⚡ Biggest Takeaways
- Aδ fibers mediate fast, sharp pain; C fibers mediate slow, dull, burning pain.
- The spinothalamic tract is the primary ascending pathway for pain and temperature.
- Key excitatory neurotransmitters for pain include Glutamate (acting on NMDA/AMPA receptors) and Substance P.
- Descending pain modulation involves the PAG and RVM, releasing serotonin (5-HT), norepinephrine (NE), and endogenous opioids.
- The Gate Control Theory proposes that non-noxious stimuli via Aβ fibers can inhibit pain signals in the dorsal horn.
- Central sensitization, or wind-up, is an NMDA receptor-dependent process leading to hyperalgesia and allodynia.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

