Contrast and Radiological Procedures

On this page
🗺️ Contrast Agents Decoded: Your Radiology Arsenal Foundation
Master contrast agents, and you unlock the diagnostic power behind every advanced imaging procedure. These pharmacological tools transform invisible anatomy into clinical gold-but only when you understand their chemistry, physiology, and risk profiles. This lesson builds your expertise from molecular mechanisms through emergency management, integrating 12+ specific thresholds that separate safe practice from catastrophic complications. You'll develop pattern recognition for reactions, master prevention protocols, and synthesize multi-system considerations that define modern radiological practice.
Contrast media represent the chemical bridge between invisible pathology and diagnostic certainty. Understanding their behavior across organ systems-particularly renal clearance kinetics, osmolality thresholds, and allergic cascade triggers-forms the foundation for every interventional decision. The difference between ionic vs non-ionic agents affects adverse reaction rates by 5-8 fold, while proper hydration protocols reduce nephropathy risk by >50%. These aren't abstract concepts-they're the quantitative principles that guide daily clinical decisions.
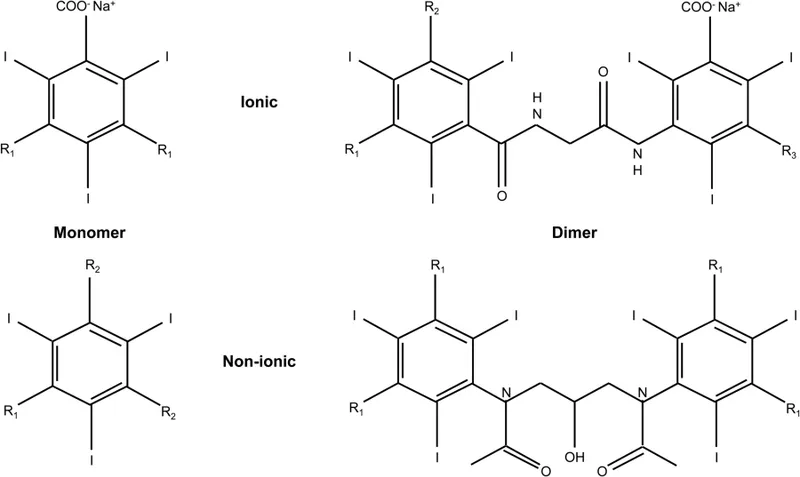
The Chemical Architecture of Visibility
Contrast agents function through differential X-ray attenuation based on atomic number-iodine (Z=53) and barium (Z=56) absorb significantly more photons than soft tissue (effective Z=7.4). This creates the radiographic density differences that reveal vascular anatomy, organ perfusion, and pathological processes.
- Iodinated Contrast Media (ICM)
- Tri-iodinated benzene ring core with 3 iodine atoms per molecule
- Concentration measured in mg iodine/mL (typically 300-400 mg I/mL)
- Osmolality determines physiological impact and adverse reaction profile
- High-osmolar (HOCM): >1400 mOsm/kg (5-8× plasma osmolality)
- Low-osmolar (LOCM): 600-900 mOsm/kg (2-3× plasma)
- Iso-osmolar (IOCM): 290 mOsm/kg (equal to plasma)
📌 Remember: "3-6-9 Rule" for osmolality classification-HOCM >1400, LOCM 600-900, IOCM ~290 mOsm/kg. The lower the osmolality, the lower the adverse reaction rate: HOCM causes reactions in 12-15%, LOCM in 3-5%, IOCM in 1-3% of patients.
- Barium Sulfate Suspensions
- Insoluble inorganic salt providing high atomic number contrast (Z=56)
- Concentration: 30-250% weight/volume depending on examination
- Particle size 0.5-2 micrometers for optimal mucosal coating
- Cannot be absorbed-purely intraluminal contrast agent

Pharmacokinetic Profiles: The Journey Through Body Compartments
Understanding contrast distribution and elimination predicts both diagnostic timing and complication risk. ICM follows a two-compartment model with rapid vascular distribution followed by renal clearance.
-
Distribution Phase (first 5-10 minutes)
- Rapid equilibration with extracellular fluid (ECF) space
- Volume of distribution: 0.2-0.3 L/kg (approximates ECF volume)
- Peak tissue enhancement: 30-70 seconds for arterial phase, 70-90 seconds for portal venous phase
- Does NOT cross intact blood-brain barrier (lipophobic molecules)
-
Elimination Phase (half-life 1.5-2 hours)
- Glomerular filtration without tubular secretion or reabsorption
- >95% excreted unchanged in urine within 24 hours
- Clearance rate approximates GFR: 120 mL/min in normal kidneys
- Delayed clearance in renal impairment: half-life extends to >10 hours when eGFR <30 mL/min/1.73m²
⭐ Clinical Pearl: Contrast enhancement timing is critical for diagnostic accuracy. Arterial phase imaging at 25-35 seconds post-injection captures hypervascular tumors (hepatocellular carcinoma shows 85% sensitivity), while portal venous phase at 65-75 seconds optimizes liver metastasis detection (sensitivity 90%). Delayed phases at 3-5 minutes reveal transitional cell carcinomas in the collecting system.
| Contrast Property | HOCM | LOCM | IOCM | Clinical Impact |
|---|---|---|---|---|
| Osmolality (mOsm/kg) | 1400-2100 | 600-900 | 290 | Pain, heat sensation, hemodynamic effects |
| Viscosity (cP at 37°C) | 4-8 | 5-10 | 12-20 | Injection rate, catheter size requirements |
| Adverse Reaction Rate | 12-15% | 3-5% | 1-3% | Overall safety profile |
| Severe Reaction Rate | 0.2-0.4% | 0.04-0.1% | 0.02-0.05% | Life-threatening event risk |
| Nephrotoxicity Risk | Highest | Moderate | Lowest | CIN incidence at eGFR <60 |
| Cost Ratio | 1× | 3-5× | 6-8× | Economic considerations |
💡 Master This: The shift from HOCM to LOCM reduced severe adverse reactions by 75%, but LOCM still carries 3-5% overall reaction rates. IOCM (iodixanol) offers the lowest nephrotoxicity in high-risk patients (eGFR 30-60 mL/min/1.73m²), reducing CIN by approximately 40% compared to LOCM in meta-analyses. However, its higher viscosity (12-20 cP) requires larger bore catheters and slower injection rates, affecting procedural efficiency.
Gadolinium-Based Contrast Agents: The MRI Revolution
Gadolinium chelates exploit paramagnetic properties to shorten T1 relaxation times, creating hyperintensity on T1-weighted sequences. Free gadolinium (Gd³⁺) is highly toxic-chelation with organic ligands ensures safe biodistribution.
-
Linear vs Macrocyclic Chelates
- Linear agents: Higher dissociation rates, greater gadolinium deposition risk
- Macrocyclic agents: Cage-like structure, thermodynamically stable, preferred in high-risk patients
- Gadolinium retention in brain (dentate nucleus, globus pallidus) demonstrated after ≥4 doses of linear agents
-
Pharmacokinetics
- Extracellular distribution (like ICM): volume 0.2-0.3 L/kg
- Renal elimination: >95% within 24 hours at normal GFR
- Standard dose: 0.1 mmol/kg (approximately 0.2 mL/kg for 0.5 M solutions)
- Half-life: 1.5-2 hours (normal renal function), >30 hours in severe renal impairment
⚠️ Warning: Nephrogenic systemic fibrosis (NSF) risk emerges when GBCAs are administered to patients with eGFR <30 mL/min/1.73m². This devastating fibrosing disorder shows >95% association with linear gadolinium agents in dialysis patients. Current guidelines recommend macrocyclic agents only, at lowest possible doses, with ≥7 days between administrations in Stage 4-5 CKD.

Connect these foundational principles through and to understand how molecular structure dictates clinical behavior and complication profiles.
🗺️ Contrast Agents Decoded: Your Radiology Arsenal Foundation
📚 The Osmolality Paradox: Chemistry Meets Physiology
Osmolality represents the single most important physicochemical property determining contrast media tolerability and adverse effects. Understanding the relationship between molecular structure, osmolality, and physiological responses unlocks the logic behind agent selection and complication prediction. The 5-8 fold difference in adverse reaction rates between HOCM and LOCM stems directly from osmotic and ionic properties-master this relationship, and you predict patient responses before injection.
The transition from ionic HOCM to non-ionic LOCM revolutionized interventional radiology in the 1980s-1990s, reducing overall adverse reactions from 12-15% to 3-5%. This wasn't merely incremental improvement-it represented fundamental redesign of molecular architecture to minimize physiological disruption while maintaining diagnostic efficacy.
Ionic vs Non-Ionic: The Molecular Decision
Traditional ionic contrast media dissociate in solution, creating two particles per molecule (anion + cation), doubling osmotic load. Non-ionic agents remain intact, generating one particle per molecule-this simple change halves osmolality while maintaining iodine concentration.
-
Ionic High-Osmolar Contrast Media (HOCM)
- Diatrizoate (Hypaque, Renografin): ionic monomer, ratio 1.5:1 (iodine atoms:particles)
- Osmolality 1400-2100 mOsm/kg (5-7× plasma)
- Dissociation produces free ions that directly stimulate nociceptors
- Cost advantage: 3-8× cheaper than LOCM/IOCM
- Still used in some barium studies and resource-limited settings
-
Non-Ionic Low-Osmolar Contrast Media (LOCM)
- Iopamidol (Isovue), iohexol (Omnipaque), iopromide (Ultravist): non-ionic monomers, ratio 3:1
- Osmolality 600-900 mOsm/kg (2-3× plasma)
- Reduced pain, heat sensation, and hemodynamic effects
- Current standard of care in most developed healthcare systems
- Adverse reactions 3-5% overall, severe reactions 0.04-0.1%
📌 Remember: "IONIC = IRONIC" because ionic agents are cheaper but cause MORE reactions. Think "3-6-9" for ratios-ionic gives 1.5:1 (round to 2), non-ionic monomer 3:1, non-ionic dimer 6:1. The higher the iodine-to-particle ratio, the lower the osmolality for equivalent iodine concentration.
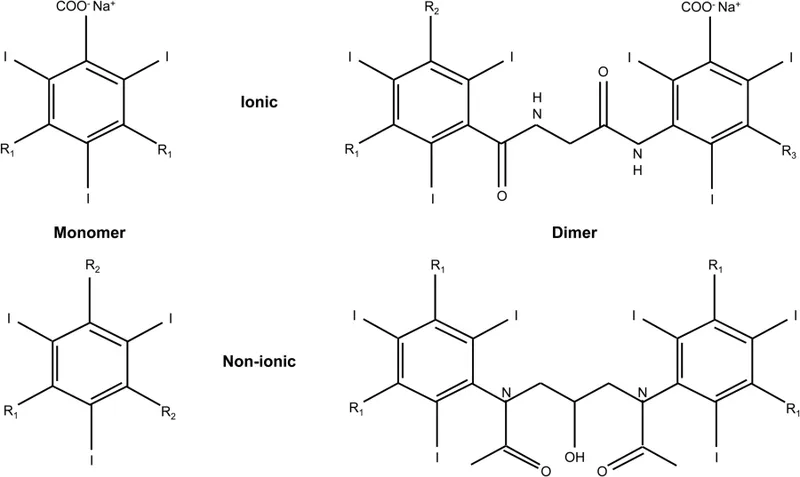
The Iso-Osmolar Innovation: Dimeric Architecture
Iso-osmolar contrast media (IOCM) achieve plasma-equivalent osmolality through dimeric structure-two tri-iodinated benzene rings linked together, providing six iodine atoms per particle (ratio 6:1).
-
Iodixanol (Visipaque) Characteristics
- Only commercially available IOCM: osmolality 290 mOsm/kg (matches plasma)
- Iodine concentration 270-320 mg I/mL with minimal osmotic gradient
- Viscosity 12-20 cP at 37°C (2-4× higher than LOCM)
- Requires 18-20 gauge catheters for adequate flow rates
- 40% reduction in CIN compared to LOCM in high-risk patients (meta-analysis data)
-
Ioxaglate (Hexabrix) - Ionic Dimer
- Ionic low-osmolar agent: 600 mOsm/kg (intermediate category)
- Ratio 3:1 due to ionic dissociation despite dimeric structure
- Historical bridge between HOCM and true LOCM/IOCM
- Largely superseded by non-ionic alternatives
⭐ Clinical Pearl: Viscosity creates a clinical trade-off with IOCM. While iodixanol's iso-osmolality reduces pain (65% decrease vs LOCM) and potentially nephrotoxicity, its high viscosity (12-20 cP) slows injection rates, requiring larger catheters and potentially limiting use in peripheral IV access <20 gauge. For rapid bolus CT angiography requiring 5-7 mL/sec injection rates, LOCM may be preferable despite slightly higher osmolality.
Physiological Consequences of Hyperosmolality
Osmotic gradients drive water movement, electrolyte shifts, and cellular stress responses. HOCM's 5-7× plasma osmolality creates dramatic physiological perturbations during intravascular injection.
-
Cardiovascular Effects
- Transient ↑ cardiac output (20-30% increase) from volume expansion
- ↓ Peripheral vascular resistance (15-25% decrease) from vasodilation
- Negative inotropic effect on myocardium (direct calcium channel interference)
- ↓ Blood pressure (10-20 mmHg systolic) in severe reactions
- QT prolongation and arrhythmia risk in susceptible patients
-
Renal Hemodynamic Changes
- Initial ↑ renal blood flow (10-15%) followed by prolonged ↓ 20-40%
- Increased medullary hypoxia from viscosity and osmotic diuresis
- Tubular vacuolization and epithelial cell injury
- Contrast-induced nephropathy (CIN) risk: 2% baseline, 20-30% in high-risk patients with HOCM
-
Neurological Effects
- Blood-brain barrier disruption at high doses (>300 mL total volume)
- Seizure risk in patients with CNS pathology or renal failure
- Transient cortical blindness (rare, <0.1%)
💡 Master This: The "osmotic load" concept explains multiple adverse effects simultaneously. HOCM's 1400-2100 mOsm/kg creates osmotic gradients that: (1) draw water into vessels causing transient volume expansion and hemodilution (↓ hematocrit 5-10%), (2) dehydrate endothelial cells causing pain and heat sensation, (3) increase blood viscosity through RBC crenation, and (4) trigger osmoreceptor-mediated nausea and vasovagal responses. LOCM's 600-900 mOsm/kg reduces these effects by 60-70%, while IOCM's 290 mOsm/kg virtually eliminates osmotic gradients.
| Physiological Parameter | Baseline | HOCM Effect | LOCM Effect | IOCM Effect | Clinical Significance |
|---|---|---|---|---|---|
| Plasma Osmolality (mOsm/kg) | 285-295 | +200-400 | +50-100 | +5-15 | Pain, nausea, hemodynamic changes |
| Cardiac Output Change | 0% | +20-30% | +5-10% | +2-5% | Heart failure decompensation risk |
| Renal Blood Flow Change | 0% | -30-40% | -10-15% | -5-10% | CIN risk correlation |
| Adverse Reaction Rate | 0% | 12-15% | 3-5% | 1-3% | Overall safety profile |
| Pain/Heat Sensation | 0% | 70-80% | 20-30% | 5-10% | Patient tolerance |
| Injection Site Extravasation Injury | Baseline | Severe | Moderate | Mild | Tissue damage potential |
Viscosity: The Flow Resistance Factor
Viscosity represents resistance to flow-higher viscosity requires greater pressure for equivalent injection rates. This becomes critical in power injector protocols and small-caliber catheter procedures.
-
Temperature Dependence
- Viscosity decreases exponentially with temperature increase
- LOCM at 20°C: 10-15 cP; at 37°C: 5-8 cP (40-50% reduction)
- IOCM at 20°C: 20-30 cP; at 37°C: 12-20 cP
- Warming contrast to 37°C before injection reduces viscosity and improves patient comfort
-
Clinical Implications
- Flow rate limitations: IOCM maximum 4-5 mL/sec through 20G catheter, vs 6-7 mL/sec for LOCM
- Catheter size requirements: IOCM requires minimum 18-20G for adequate flow
- Injection pressure: Higher viscosity increases risk of catheter/tubing rupture at >300 psi
- Peripheral IV considerations: 22G IV adequate for LOCM at 3-4 mL/sec, marginal for IOCM
⚠️ Warning: Never inject cold contrast at high flow rates. Room temperature (20-25°C) contrast has 2-3× higher viscosity than body temperature, dramatically increasing injection pressures and extravasation risk. Studies show warming contrast to 37°C reduces pain scores by 40% and improves vascular opacification by 15-20% through better flow dynamics.
📚 The Osmolality Paradox: Chemistry Meets Physiology
🔬 Reaction Recognition: The Allergic-Like Cascade Demystified
Contrast reactions mimic allergic responses but operate through non-IgE-mediated mechanisms in >95% of cases-understanding this distinction transforms prevention and management strategies. Traditional terminology of "allergic reactions" misleads clinicians into expecting predictable immunological patterns, when contrast reactions actually represent direct cellular activation, complement activation, and osmotic/chemotoxic effects. The 3-5% overall reaction rate with LOCM breaks down into 85% mild, 10% moderate, and 5% severe reactions-pattern recognition at each severity level enables rapid, appropriate intervention.
True IgE-mediated contrast allergy accounts for only <5% of reactions, yet the clinical presentation of urticaria, bronchospasm, and anaphylaxis-like symptoms creates diagnostic confusion. The key insight: contrast reactions are idiosyncratic and unpredictable, with poor correlation between prior reactions and future events (17-35% recurrence rate even without premedication).

Immediate Reactions: The First 60 Minutes
Immediate reactions occur within 1 hour of injection, representing 85-90% of all contrast adverse events. These stem from direct mast cell and basophil degranulation, complement activation, or direct chemotoxic effects-not classic Type I hypersensitivity in most cases.
- Mild Reactions (70-80% of immediate reactions)
- Limited urticaria (<5 scattered hives): 2-3% incidence
- Pruritus without skin changes: 1-2% incidence
- Transient nausea: 3-5% incidence with LOCM, 8-12% with HOCM
- Flushing and warmth sensation: 10-15% incidence (osmotic effect)
- Self-limited, no treatment required in most cases
- Observation for 20-30 minutes sufficient
📌 Remember: "ITCH-HIVE-FLUSH-PUKE" for mild reactions-Itching, Hives (limited), Flushing, Nausea (puke). These require observation only unless progression occurs. The "5-20-30 Rule": <5 hives = mild, 20 minutes observation minimum, 30 minutes ideal before discharge.
-
Moderate Reactions (15-20% of immediate reactions)
- Diffuse urticaria or angioedema: 0.5-1% incidence
- Bronchospasm with wheezing: 0.3-0.5% incidence
- Facial/laryngeal edema without airway compromise: 0.2-0.4% incidence
- Tachycardia >100 bpm or bradycardia <50 bpm
- Hypertension >180/110 mmHg or hypotension <90 mmHg systolic
- Persistent vomiting (risk of aspiration)
- Requires treatment but not immediately life-threatening
-
Severe Reactions (5-10% of immediate reactions, 0.04-0.1% overall)
- Hypotensive shock: systolic BP <80 mmHg or >30% decrease from baseline
- Respiratory arrest or severe bronchospasm with hypoxia (SpO2 <90%)
- Laryngeal edema with stridor or airway compromise
- Seizures or loss of consciousness
- Cardiac arrest or life-threatening arrhythmias
- Immediate resuscitation required, call code team
⭐ Clinical Pearl: The "3-Minute Rule" for severe reactions-if symptoms don't respond to initial interventions within 3 minutes, escalate immediately. Hypotension not responding to 500 mL bolus + epinephrine 0.1 mg IM within 3 minutes requires IV epinephrine infusion and ICU-level care. Bronchospasm not improving with albuterol + epinephrine within 3 minutes needs IV corticosteroids and possible intubation preparation.
Delayed Reactions: The 1-Hour to 7-Day Window
Delayed reactions occur >1 hour after contrast administration, representing 10-15% of all adverse events. These typically manifest as skin reactions (70-80% of delayed reactions) and follow a T-cell mediated mechanism similar to drug hypersensitivity.
-
Cutaneous Delayed Reactions (onset 3-48 hours, peak 7-10 days)
- Maculopapular exanthem: 1-3% incidence, most common delayed reaction
- Delayed urticaria: 0.5-1% incidence
- Fixed drug eruption: 0.1-0.3% incidence
- Erythema multiforme: <0.1% incidence
- Management: antihistamines, topical corticosteroids, observation
-
Severe Delayed Reactions (rare, <0.01%)
- Stevens-Johnson syndrome (SJS): <0.001% incidence
- Toxic epidermal necrolysis (TEN): <0.0001% incidence
- Acute generalized exanthematous pustulosis (AGEP): <0.01% incidence
- Requires immediate dermatology consultation and hospitalization
💡 Master This: Delayed reactions show poor correlation with immediate reaction history-patients can have immediate reactions without delayed reactions and vice versa. The T-cell mediated pathophysiology means premedication with antihistamines and corticosteroids (designed for mast cell stabilization) provides minimal protection against delayed reactions. Alternative contrast agents or imaging modalities should be considered for patients with prior severe delayed reactions.
Physiologic Reactions vs Allergic-Like Reactions
Distinguishing physiologic responses from true allergic-like reactions guides management intensity and future contrast use decisions.
-
Physiologic Reactions (predictable, dose-dependent)
- Warmth and flushing: osmotic vasodilation, expected with HOCM
- Nausea from chemoreceptor trigger zone stimulation
- Transient hypotension from vasodilation (10-20 mmHg drop)
- Pain at injection site from osmotic endothelial irritation
- Management: Reassurance, observation, slow injection rates
-
Allergic-Like Reactions (idiosyncratic, unpredictable)
- Urticaria, angioedema, bronchospasm
- Anaphylactoid shock (clinically indistinguishable from anaphylaxis)
- Not dose-dependent: can occur with small test doses
- Management: Treat as allergic emergency regardless of mechanism
| Feature | Physiologic Reaction | Allergic-Like Reaction | True IgE-Mediated Allergy |
|---|---|---|---|
| Mechanism | Osmotic, chemotoxic | Direct mast cell activation | IgE antibody-mediated |
| Predictability | Dose-dependent, expected | Idiosyncratic, unpredictable | Predictable on re-exposure |
| Prior Exposure Required | No | No | Yes (sensitization phase) |
| Recurrence Rate | Consistent with same agent | 17-35% with same agent | >80% without premedication |
| Premedication Efficacy | Not needed | 40-60% risk reduction | 60-90% risk reduction |
| Skin Testing Value | Not applicable | Limited value (<30% sensitivity) | Potentially useful (70-80% sensitivity) |
| Cross-Reactivity | Not applicable | 10-15% between agents | 40-50% between similar structures |
Risk Stratification: Identifying High-Risk Patients
Certain patient factors increase reaction risk 3-6 fold-systematic risk assessment before contrast administration enables targeted prevention strategies.
-
Major Risk Factors (increase risk 5-6 fold)
- Prior contrast reaction (most significant risk factor)
- Mild prior reaction: 10-15% recurrence risk
- Moderate/severe prior reaction: 20-35% recurrence risk
- Without premedication, risk approaches 35-40%
- Asthma requiring medication: 5-6 fold increased risk
- Multiple drug allergies (≥3 documented allergies): 4-5 fold increased risk
- Prior contrast reaction (most significant risk factor)
-
Moderate Risk Factors (increase risk 2-3 fold)
- History of any allergy (food, environmental): 2-3 fold risk
- Cardiac disease (especially heart failure, arrhythmias)
- Dehydration or volume depletion
- Renal insufficiency (eGFR <60 mL/min/1.73m²)
- Extremes of age (<1 year or >70 years)
-
Minor Risk Factors (increase risk <2 fold)
- Anxiety or high stress level
- Female gender (slightly higher reaction rates)
- Thyroid disease (relevant for iodine uptake considerations)
⚠️ Warning: "Shellfish allergy" is a myth as a contrast reaction predictor. Shellfish allergy stems from tropomyosin protein, not iodine. Patients with shellfish allergy have no increased risk of contrast reactions compared to general population (3-5% baseline). However, patients with multiple food allergies (including shellfish as one of several) do have increased risk due to general atopic tendency, not the iodine content.
📌 Remember: "PAPA-DREAD" for high-risk patients-Prior reaction, Asthma, Polypharmacy allergies, Anxiety, Dehydration, Renal disease, Elderly, Atopy, Drugs (beta-blockers, ACE inhibitors). These patients need premedication protocols and enhanced monitoring during and after contrast administration.
🔬 Reaction Recognition: The Allergic-Like Cascade Demystified
🏥 Contrast-Induced Nephropathy: The Renal Vulnerability Window
Contrast-induced nephropathy (CIN) represents the third most common cause of hospital-acquired acute kidney injury, accounting for 10-12% of cases. This iatrogenic complication transforms a diagnostic procedure into a clinical crisis-20-30% of high-risk patients develop CIN, and those who do face 30-50% increased mortality risk within 1 year. Understanding the 72-hour vulnerability window, recognizing high-risk profiles, and implementing evidence-based prevention protocols separates safe contrast practice from preventable renal disaster.
The pathophysiology centers on renal medullary hypoxia-contrast agents create a perfect storm of vasoconstriction, increased oxygen demand, and direct tubular toxicity that overwhelms the kidney's limited oxygen reserve. The outer medulla operates at baseline pO2 10-20 mmHg (compared to 40-50 mmHg in cortex), making it exquisitely vulnerable to any additional hypoxic stress.

Defining and Diagnosing CIN: The 48-72 Hour Window
CIN diagnosis requires temporal relationship to contrast exposure and exclusion of alternative causes-the serum creatinine rise within 48-72 hours serves as the diagnostic cornerstone.
-
Standard CIN Definition (most widely used)
- Absolute increase in serum creatinine ≥0.5 mg/dL from baseline
- OR relative increase ≥25% from baseline
- Occurring within 48-72 hours of contrast administration
- In absence of alternative explanations for AKI
- Peak creatinine typically at 3-5 days, return to baseline by 7-14 days
-
Alternative CIN Definitions (more sensitive but less specific)
- Creatinine increase ≥0.3 mg/dL within 48 hours (KDIGO AKI criteria)
- Urine output <0.5 mL/kg/hr for >6 hours (rarely used for CIN)
- eGFR decrease ≥25% from baseline
📌 Remember: "3-5-7 Rule" for CIN timeline-creatinine rises within 3 days, peaks at 3-5 days, returns to baseline by 7-14 days. If creatinine doesn't peak by day 5 or doesn't improve by day 14, consider alternative diagnoses (atheroembolic disease, acute tubular necrosis from other causes, acute interstitial nephritis).
- CIN Severity Stratification (RIFLE criteria adaptation)
- Risk: Creatinine increase 1.5-2× baseline or eGFR decrease 25-50%
- Injury: Creatinine increase 2-3× baseline or eGFR decrease 50-75%
- Failure: Creatinine increase >3× baseline or eGFR <30 mL/min with acute rise
- Dialysis requirement: <1% in low-risk patients, 5-10% in high-risk patients
Risk Stratification: The Mehran Score and Beyond
Quantitative risk prediction enables personalized prevention strategies. The Mehran Risk Score (developed from >8000 patients) predicts CIN with C-statistic 0.67, providing structured risk assessment.
-
Mehran Risk Score Components (maximum 16 points)
- Hypotension requiring inotropes: 5 points
- Intra-aortic balloon pump: 5 points
- Congestive heart failure (NYHA III-IV or pulmonary edema): 5 points
- Age >75 years: 4 points
- Anemia (hematocrit <39% men, <36% women): 3 points
- Diabetes mellitus: 3 points
- Contrast volume (per 100 mL): 1 point
- Serum creatinine >1.5 mg/dL: 4 points
- OR eGFR 40-60: 2 points, 20-40: 4 points, <20: 6 points
-
Mehran Score Risk Categories
- Low risk (0-5 points): CIN risk 7.5%
- Moderate risk (6-10 points): CIN risk 14%
- High risk (11-16 points): CIN risk 26.1%
- Very high risk (>16 points): CIN risk 57.3%
⭐ Clinical Pearl: The "3-Risk-Factor Rule" provides rapid bedside assessment without calculation-patients with ≥3 of the following face >20% CIN risk: (1) eGFR <60, (2) diabetes, (3) age >70, (4) heart failure, (5) anemia, (6) contrast volume >200 mL. These patients mandate aggressive prevention protocols including IV hydration, minimum contrast volume, and consideration of alternative imaging.
Pathophysiologic Mechanisms: The Triple Threat
CIN results from three synergistic mechanisms-understanding each guides targeted prevention strategies.
-
Renal Vasoconstriction (primary mechanism)
- Contrast triggers adenosine release → afferent arteriole vasoconstriction
- ↓ Renal blood flow 20-40% lasting 4-6 hours post-contrast
- Preferential medullary hypoperfusion (baseline pO2 10-20 mmHg drops to <10 mmHg)
- Endothelin and reactive oxygen species amplify vasoconstriction
- Prevention target: Volume expansion to counteract vasoconstriction
-
Direct Tubular Toxicity (secondary mechanism)
- Osmotic injury to proximal tubule epithelial cells
- Mitochondrial dysfunction and ATP depletion
- Tubular obstruction from Tamm-Horsfall protein precipitation
- Apoptosis and necrosis of tubular cells
- Prevention target: Minimize contrast volume and osmolality
-
Oxidative Stress (amplifying mechanism)
- Reactive oxygen species (ROS) generation in tubular cells
- Lipid peroxidation and membrane damage
- Inflammation and cytokine release
- Prevention target: Antioxidant strategies (limited evidence)
💡 Master This: The "contrast volume-to-creatinine clearance ratio" predicts CIN risk-when contrast volume (mL) ÷ creatinine clearance (mL/min) >3.7, CIN risk exceeds 50%. For example, a patient with eGFR 40 mL/min receiving 150 mL contrast has ratio 3.75 (high risk). This simple calculation guides volume limitation strategies: keep ratio <2.0 for low risk, <3.0 for moderate risk.
| Risk Factor | Baseline CIN Risk | With Risk Factor | Relative Risk Increase | Prevention Strategy |
|---|---|---|---|---|
| eGFR 30-60 mL/min | 2% | 10-15% | 5-7 fold | IV hydration, LOCM/IOCM |
| eGFR <30 mL/min | 2% | 20-30% | 10-15 fold | Aggressive hydration, IOCM, minimize volume |
| Diabetes + CKD | 2% | 20-50% | 10-25 fold | All prevention measures |
| Heart Failure | 2% | 15-20% | 7-10 fold | Careful hydration, hemodynamic monitoring |
| Contrast Volume >200 mL | 2% | 8-12% | 4-6 fold | Minimize volume, staged procedures |
| HOCM vs LOCM | 2% | 6-8% vs 3-4% | 2-3 fold | Use LOCM minimum |
Evidence-Based Prevention: The Hydration-Contrast-Monitoring Triad
Prevention strategies focus on three pill
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app


















