Receptor-Ligand Interactions: Basics - Keys & Locks
- Receptor: Cellular macromolecule (protein/glycoprotein) that binds specific signaling molecules (ligands).
- Locations: Cell membrane, cytoplasm, nucleus.
- Ligand: Molecule that binds to a receptor (e.g., hormone, neurotransmitter, drug).
- Endogenous: Produced by the body.
- Exogenous: Originates outside the body (e.g., drugs).
- Interaction Specificity: "Lock & Key" model (rigid fit) or "Induced Fit" model (flexible, conformational change).
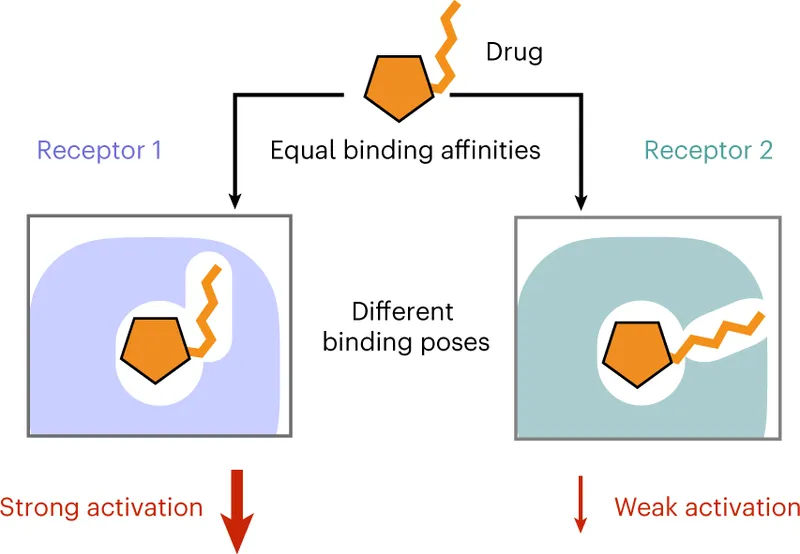
- Affinity: Strength of binding between ligand and receptor. High affinity = strong attraction.
- Intrinsic Activity (Efficacy): Ability of a ligand-receptor complex to produce a functional response.
- Agonist: Binds receptor, activates it, produces a response (has efficacy).
- Antagonist: Binds receptor, no activation, blocks agonist action (no efficacy).
⭐ Most therapeutic drugs exert their effects by interacting with specific receptors, acting as agonists or antagonists.
Receptor-Ligand Interactions: Kinetics - The Dating Game
- Affinity: Strength of ligand-receptor bond.
- $K_d$ (dissociation constant): Ligand concentration for 50% receptor occupancy. $K_d = [R][L]/[RL]$.
- ↓$K_d$ = ↑Affinity (tight binding).
- ↑$K_d$ = ↓Affinity (loose binding).
⭐ $K_d$ (dissociation constant) is inversely proportional to binding affinity; a lower $K_d$ indicates higher affinity and tighter binding.
- Specificity: Receptor's ability to bind specific ligands.
- Saturation: Finite receptors; max effect when all bound.
- Scatchard Plot: Analyzes $K_d$ & $B_{max}$ (total receptors).
- Slope = $-1/K_d$. X-intercept = $B_{max}$.

- Slope = $-1/K_d$. X-intercept = $B_{max}$.
- Dose-Response Curves:
- Potency: Drug conc. for 50% max effect ($EC_{50}$). Left shift = ↑Potency.
- Efficacy: Max possible effect ($E_{max}$).
- Antagonism:
- Competitive: Reversible; agonist site. ↓Potency, $E_{max}$ same. Overcome by ↑agonist.
- Non-competitive: Allosteric/irreversible. ↓$E_{max}$ (efficacy).
Receptor-Ligand Interactions: Types - Cellular Switchboards
Receptors act as cellular switchboards, translating extracellular signals into intracellular responses. Major types differ in structure, location, and mechanism:
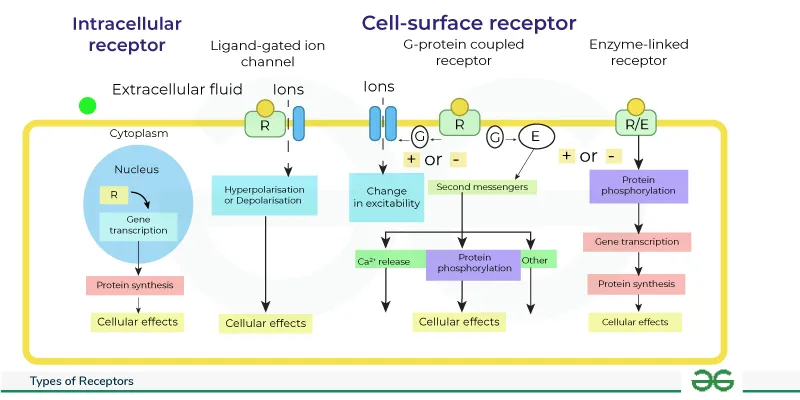
| Receptor Superfamily | Location | Coupling | Effector Pathway | Speed | Examples |
|---|---|---|---|---|---|
| Ligand-Gated Ion Channels (Ionotropic) | Cell Membrane | Direct | Ion flux ($Na^+$, $K^+$, $Ca^{2+}$, $Cl^-$) | Very Fast (ms) | Nicotinic AChR, GABA-A, Glutamate (NMDA, AMPA) |
| G-Protein Coupled Receptors (GPCRs) | Cell Membrane | G-Protein | 2nd messengers ($cAMP$, $IP_3$/$DAG$, $Ca^{2+}$) | Fast (s-min) | Adrenergic, Muscarinic AChR, Opioid, Glucagon |
| Enzyme-Linked Receptors | Cell Membrane | Direct/Associated | Enzyme activity (Tyr Kinase, Ser/Thr Kinase, Guanylyl Cyclase) | Slow (min-hr) | Insulin, Growth Factors (EGF, PDGF), ANP |
| Nuclear Receptors (Intracellular) | Cytosol/Nucleus | Direct | Gene transcription modulation | Very Slow (hr-days) | Steroids, Thyroid Hormones, Vit D, Retinoids |
- Gs: Stimulates adenylyl cyclase → ↑$cAMP$.
- Gi: Inhibits adenylyl cyclase → ↓$cAMP$; opens $K^+$ channels.
- Gq: Activates phospholipase C → ↑$IP_3$ & $DAG$ → ↑$Ca^{2+}$ & PKC activation.
⭐ G-Protein Coupled Receptors (GPCRs) constitute the largest family of cell surface receptors and are targets for approximately 30-40% of all modern medicinal drugs.
Receptor-Ligand Interactions: Regulation - Cellular Mood Swings
- Receptors dynamic: number & sensitivity modulate.
- Upregulation: ↑ receptor number/sensitivity (e.g., prolonged antagonist).
- Downregulation: ↓ receptor number/sensitivity (e.g., prolonged agonist); internalization.
- Desensitization: ↓ receptor responsiveness (e.g., phosphorylation).
- Tolerance: Gradual ↓ drug effect with chronic use.

⭐ Tachyphylaxis refers to the rapid decrease in response to a drug after repeated doses over a short period, often due to receptor desensitization or depletion of mediators.
High‑Yield Points - ⚡ Biggest Takeaways
- Affinity (Kd): Strength of ligand-receptor binding; low Kd = high affinity.
- Specificity: Receptors bind specific ligands, ensuring precise cellular responses.
- Saturation: Finite receptor numbers limit the maximal effect of a ligand.
- Agonists (full, partial, inverse) activate receptors; antagonists (competitive, non-competitive) block them.
- Competitive antagonists increase ED50; non-competitive antagonists decrease Emax.
- Spare receptors enable maximal response without 100% receptor occupancy.
- Receptor downregulation/desensitization occurs with prolonged agonist exposure.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

