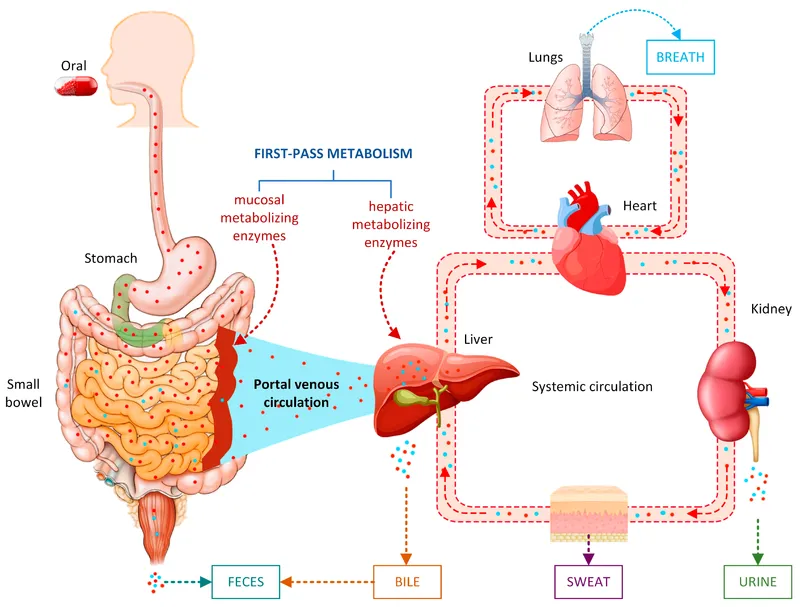
Drug Excretion Fundamentals - The Great Escape!
- Excretion: Irreversible removal of drug/metabolites, primarily terminating drug action and preventing toxicity.
- Major Organs:
- Kidney: Main route for water-soluble drugs & metabolites.
- Liver (Biliary Excretion): For larger, lipophilic drugs; may lead to enterohepatic circulation.
- Lungs: For volatile compounds (e.g., gaseous anesthetics, ethanol).
- Minor Routes: Saliva, sweat, tears, breast milk (important for drug transfer to infants).
⭐ First-order kinetics is the most common mechanism for drug elimination: a constant fraction of drug is eliminated per unit time ($C_t = C_0 e^{-kt}$).
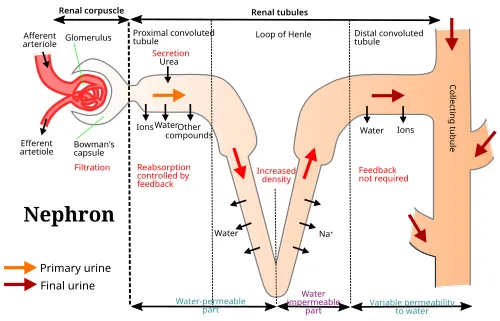
Renal Excretion - Kidney's Clean Sweep
Kidneys are vital for eliminating water-soluble drugs/metabolites. Renal clearance ($Cl_R$) involves three key processes: $Cl_R = (Rate \ of \ filtration + Rate \ of \ secretion - Rate \ of \ reabsorption) / C_p$.
- Glomerular Filtration (GF):
- Passive; rate depends on GFR & fraction unbound (fu) of drug.
- Only unbound, low MW (< 60,000 Da) drugs are filtered. Protein-bound drugs are not.
- Active Tubular Secretion (ATS):
- Proximal tubule; carrier-mediated, saturable, requires energy.
- Transporters: OATs (for acidic drugs like penicillin; probenecid-sensitive), OCTs (for basic drugs like metformin), P-gp, MRPs.
- 📌 Probenecid + Penicillin: Probenecid inhibits OATs, ↓ penicillin secretion, ↑ its $t_{1/2}$ ("PROlongs PENicillin").
- Tubular Reabsorption (TR):
- Mainly passive diffusion in proximal & distal tubules.
- Favors lipid-soluble, non-ionized drugs.
- Ion trapping (pH-dependent excretion, based on drug pKa):
- Acidic drugs (e.g., aspirin): Alkalinize urine (NaHCO₃) → ↑ ionization → ↑ excretion.
- Basic drugs (e.g., amphetamine): Acidify urine (NH₄Cl) → ↑ ionization → ↑ excretion.
⭐ For drugs eliminated solely by glomerular filtration (e.g., inulin), renal clearance equals GFR multiplied by the unbound fraction ($Cl_R = GFR \times fu$).
Non-Renal Excretion - Beyond the Bladder
Drugs can also be eliminated through routes other than the kidneys. Key non-renal pathways include:
| Route | Drug Properties | Significance & Examples |
|---|---|---|
| Biliary | High MW (>500 Da), conjugated, polar | Enterohepatic circulation (EHC) prolongs drug action (e.g., digoxin, OCPs, morphine) |
| Pulmonary | Volatile liquids, gases | Rapid elimination via exhalation (e.g., general anesthetics like halothane) |
| Salivary | Lipid-soluble, unionized; pH-dependent | Minor route; drug taste, compliance issues (e.g., lithium, phenytoin, metronidazole) |
| Sweat | Lipid-soluble, unionized | Minor, variable; can cause skin reactions (e.g., rifampicin - red sweat, metals) |
| Milk | Basic drugs (milk pH $~7.0-7.2$ vs plasma $~7.4$), lipid-soluble | Ion trapping of basic drugs; risk to breastfeeding infant (e.g., morphine, diazepam) |
⭐ Enterohepatic circulation significantly prolongs drug half-life (e.g., OCPs, digoxin), affecting dosing schedules and potentially leading to accumulation if not considered.
Clinical Excretion Kinetics - Dosing Decisions
- Clearance ($Cl$): Plasma volume cleared of drug/time.
- $Cl_{Total} = Cl_{Renal} + Cl_{Hepatic} + Cl_{Other}$.
- $Cl_{Renal}$ vital for kidney-excreted drugs.
- Elimination Half-life ($t_{1/2}$): Time for drug conc. to halve.
- $t_{1/2} = (0.693 \times V_d) / Cl$.
- Steady state in 4-5 $t_{1/2}$; guides dosing interval.
- Renal Impairment: ↓GFR → ↓$Cl_R$ → drug accumulation/toxicity.
- Dose adjustment crucial for renally cleared drugs.
- Dose Adjustment: Based on Creatinine Clearance (CrCl).
- Cockcroft-Gault: $CrCl = [(140-age) \times Wt (kg) \times (0.85 \ if \ female)] / (72 \times SCr)$.
- Methods: ↓dose, ↑interval, or both.
- Dialysis: Removes drugs: Low MW, Low $V_d$, Low protein binding, Water-soluble.
- Supplemental dose post-dialysis may be needed.
⭐ For narrow therapeutic index drugs mainly kidney-excreted (e.g., aminoglycosides, digoxin), CrCl-based dose adjustment (e.g., if GFR < 50 mL/min) is vital to avoid toxicity.
High‑Yield Points - ⚡ Biggest Takeaways
- Glomerular filtration is passive, affected by plasma protein binding & renal blood flow.
- Tubular secretion is an active, saturable process (e.g., penicillin with probenecid).
- Tubular reabsorption is pH-dependent; ion trapping (e.g., aspirin with NaHCO₃) aids excretion.
- Clearance (CL) = Rate of elimination / Plasma concentration; Total CL sums organ clearances.
- Enterohepatic circulation (e.g., digitoxin, rifampicin) prolongs drug action.
- Pulmonary excretion is key for volatile anesthetics and alcohol.
- Biliary excretion is important for high molecular weight drugs and their conjugates.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

