Biotransformation: Overview - Chemical Makeovers
- Definition: Enzymatic chemical alteration of drugs, primarily in the liver.
- Purpose: Convert lipophilic drugs to more polar (water-soluble) metabolites for easier renal excretion.
- Key Sites:
- Liver (dominant)
- GIT, Lungs, Kidneys, Skin, Plasma
- Reaction Phases:
- Phase I (Functionalization): Oxidation, reduction, hydrolysis. Introduces/unmasks a functional group.
- Phase II (Conjugation): Covalent attachment of an endogenous molecule (e.g., glucuronide).

⭐ The cytochrome P450 (CYP450) enzyme system, predominantly in the liver, is crucial for Phase I metabolism of many drugs.
Phase I Metabolism: CYP Enzymes - CYP Showdown
- Reactions: Oxidation (main), Reduction, Hydrolysis, Cyclization, Decyclization. Goal: ↑polarity for excretion.
- Location: Primarily liver (smooth endoplasmic reticulum), also GI tract, lungs, kidneys.
- Key Enzymes & Significance:
- CYP3A4/5: Metabolizes ~50% of clinically used drugs.
- CYP2D6: Marked genetic polymorphism (e.g., codeine efficacy, tamoxifen activation).
- CYP2C9: Metabolizes warfarin (narrow therapeutic index).
- CYP2C19: Activates clopidogrel; polymorphism affects efficacy.
- CYP Inducers: ↑Enzyme synthesis → ↑Metabolism → ↓Drug plasma concentration & effect (or ↑active metabolite).
- 📌 CRAP GPS induces my rage!: Carbamazepine, Rifampicin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, Smoking/St. John's Wort.
- CYP Inhibitors: ↓Enzyme activity → ↓Metabolism → ↑Drug plasma concentration & risk of toxicity.
- 📌 SICKFACES.COM Group: Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole, Grapefruit juice.

- 📌 SICKFACES.COM Group: Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole, Grapefruit juice.
⭐ Grapefruit juice is a potent inhibitor of intestinal CYP3A4, significantly increasing the bioavailability and risk of toxicity of many drugs like statins and calcium channel blockers.
Phase II Metabolism: Conjugation - Conjugation Crew
- Synthetic reactions: drug + endogenous substrate → conjugate.
- Purpose: ↑ water solubility, ↑ excretion.
- Key Reactions:
- Glucuronidation: UGT; UDP-glucuronic acid. Most common.
- Acetylation: NAT; Acetyl-CoA. (📌 Polymorphism: Slow/Fast acetylators - Isoniazid, Hydralazine, Procainamide - "SHIP")
- Sulfation: SULT; PAPS.
- Methylation: Methyltransferase; SAM.
- Glutathione Conjugation: GST; GSH. Detoxifies electrophiles.
- Usually inactivates drugs (e.g., Morphine-6-glucuronide is active).

⭐ N-acetyltransferase (NAT2) polymorphism causes slow/fast acetylator status, affecting toxicity (e.g., isoniazid neuropathy in slow acetylators).
Drug Excretion: Renal & Others - The Great Escape
- Main Route: Kidneys. Others: Biliary, pulmonary, saliva, sweat, milk.
- Renal Mechanisms:
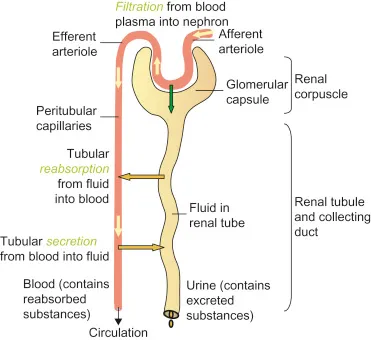
- Glomerular Filtration (GF): Unbound drug; depends on GFR.
- Tubular Reabsorption: Passive (lipid-soluble, non-ionized). Ion trapping (↑ ionized drug, e.g., $A^-$, $BH^+$): acidic drugs → alkaline urine; basic drugs → acidic urine. 📌 Urine pH change traps ionized drug.
- Tubular Secretion: Active (OATs, OCTs), saturable, competition.
- Factors Affecting Renal Excretion:
- Biliary Excretion: Drugs >300 Da MW, conjugates. Enterohepatic circulation.
- Milk: Basic, lipid-soluble drugs. ⚠️ Risk to nursing infant.
⭐ Alkalinization of urine (e.g., NaHCO₃) enhances excretion of acidic drugs (Aspirin); acidification (NH₄Cl) for basic drugs (Amphetamine).
Clinical Pharmacokinetics: Metabolism & Excretion - Dose & Duration
- Clearance ($Cl$): Rate of drug elimination. $Cl = (0.693 \times Vd) / t½$. Governs maintenance dose.
- Half-life ($t½$): Time for drug concentration to decrease by 50%. $t½ = (0.693 \times Vd) / Cl$. Determines dosing interval; steady state reached in 4-5 $t½$.
- First-pass metabolism: Hepatic/gut wall metabolism before systemic circulation; ↓ bioavailability (e.g., lignocaine, propranolol).
- Factors: Genetic (e.g., slow/fast acetylators 📌 INH), liver/kidney disease (↓ $Cl$, ↑ $t½$).

⭐ Steady state concentration ($Css$) is achieved after approximately 4-5 elimination half-lives of a drug when administered by continuous infusion or fixed intermittent doses.
High‑Yield Points - ⚡ Biggest Takeaways
- Phase I reactions (oxidation, reduction, hydrolysis) via CYP450 make drugs polar.
- Phase II reactions (conjugation, e.g., glucuronidation) form highly polar, inactive, excretable metabolites.
- Enzyme induction (e.g., rifampicin, phenytoin) ↑ metabolism, ↓ drug effect.
- Enzyme inhibition (e.g., ketoconazole, erythromycin) ↓ metabolism, ↑ drug toxicity.
- First-pass metabolism in liver significantly ↓ oral drug bioavailability.
- Zero-order kinetics: constant amount eliminated/time (e.g., Warfarin, Aspirin, Tolbutamide, Ethanol, Phenytoin).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

