Introduction to Targeted Therapy - Precision Power
- Drugs acting on specific molecular targets (proteins, genes) vital for cancer cell growth, progression, and survival.
- Mechanism: "Lock and key" model; drugs precisely interact with targets often unique to or overexpressed in cancer cells.
- Patient selection: Requires predictive biomarker identification (e.g., EGFR, HER2, BRAF mutations) via genetic/protein tests.
- Contrasts conventional chemotherapy, which non-selectively targets all rapidly dividing cells.
- Goal: Enhanced efficacy, reduced systemic toxicity ("Precision Medicine"), improving therapeutic index by sparing normal cells.
⭐ Many targeted therapies are monoclonal antibodies (suffix "-mab") targeting cell surface receptors or small molecule kinase inhibitors (suffix "-ib") affecting intracellular signals.
Tyrosine Kinase Inhibitors - Signal Jammers
- Small molecule inhibitors targeting intracellular kinase domains, blocking ATP binding sites.
- Interfere with signaling pathways essential for tumor cell proliferation and survival.
- Generally orally administered. Many end in "-tinib". 📌 (Imatinib)
- Common class side effects: GI toxicity, rash, fatigue.
- Resistance via gatekeeper mutations is a challenge.
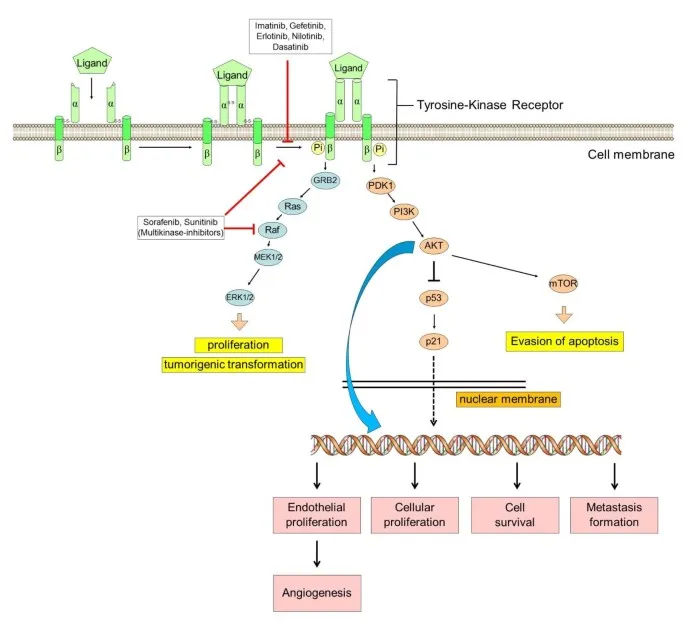
| Drug | Primary Target(s) | Key Indication(s) | Hallmark Side Effect(s) |
|---|---|---|---|
| Imatinib | BCR-ABL, c-KIT | CML, GIST | Fluid retention, myelosuppression |
| Erlotinib | EGFR | NSCLC (EGFR+) | Acneiform rash, diarrhea |
| Crizotinib | ALK, ROS1 | NSCLC (ALK+/ROS1+) | Visual issues, hepatotoxicity |
| Vemurafenib | BRAF V600E | Melanoma (BRAF V600E+) | Arthralgia, photosensitivity, skin SCC |
| Sunitinib | Multi-kinase (VEGFR) | RCC, GIST (Imat-res) | Fatigue, HTN, hand-foot synd. |
Monoclonal Antibodies - Guided Missiles
- Large proteins targeting specific cell surface antigens (e.g., receptors) or circulating proteins.
- Nomenclature indicates origin & structure:
- -omab: Murine (100% mouse)
- -ximab: Chimeric (e.g., Rituximab; ~30% mouse)
- -zumab: Humanized (e.g., Trastuzumab; ~5-10% mouse)
- -umab: Human (e.g., Nivolumab; 100% human)
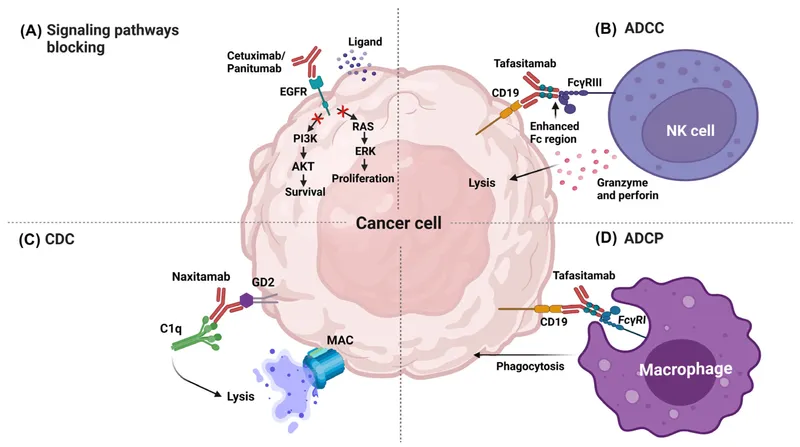
- Key Examples & Targets:
mAb Target Key Indications Trastuzumab HER2 Breast Ca, Gastric Ca Rituximab CD20 NHL, CLL, RA Bevacizumab VEGF CRC, NSCLC, RCC, Glioblastoma Cetuximab EGFR CRC, H&N Ca Pembrolizumab PD-1 Melanoma, NSCLC, RCC, Hodgkin Ipilimumab CTLA-4 Melanoma - Adverse Effects (AEs):
- General: Infusion reactions, hypersensitivity (less with human/humanized).
- Specific:
- Trastuzumab: Cardiotoxicity (monitor LVEF).
- Bevacizumab: Hypertension, bleeding, GI perforation, impaired wound healing.
- Cetuximab/Panitumumab: Acneiform rash.
- Checkpoint Inhibitors (CPIs): Immune-related AEs (irAEs) - colitis, hepatitis, pneumonitis, endocrinopathies.
⭐ Trastuzumab is only effective in HER2-positive cancers; HER2 testing is mandatory before therapy initiation.
Other Targets & Resistance - New Paths, Old Foes
- Key Alternative Targets & Agents:
- PARP Inhibitors (Olaparib, Niraparib): BRCA1/2 mutated cancers (ovarian, breast, prostate). MOA: Synthetic lethality.
- Proteasome Inhibitors (Bortezomib, Carfilzomib): Multiple Myeloma. MOA: Inhibit proteasome, ↑apoptosis.
- mTOR Inhibitors (Everolimus, Sirolimus): Renal Cell Ca, HR+ Breast Ca. MOA: Block mTOR, ↓cell growth.
- HDAC Inhibitors (Vorinostat): CTCL. MOA: Epigenetic modulation.
- Hedgehog Pathway Inhibitors (Vismodegib): Basal cell carcinoma.
- Mechanisms of Acquired Resistance:
- Target gene mutations (e.g., EGFR T790M).
- Bypass pathway activation (e.g., MET amplification).
- Drug efflux (P-glycoprotein).
- Phenotypic transformation.
- Combating Resistance:
- Next-gen inhibitors (Osimertinib for T790M).
- Combination therapies.
- Liquid biopsies for early detection.

⭐ Osimertinib, a 3rd-gen EGFR-TKI, is highly effective against the T790M "gatekeeper" resistance mutation in NSCLC.
High‑Yield Points - ⚡ Biggest Takeaways
- Imatinib: A TKI for CML (BCR-ABL) and GIST (c-KIT).
- Monoclonal antibodies: Trastuzumab (HER2+ breast cancer), Rituximab (CD20+ lymphomas).
- EGFR inhibitors: Gefitinib (NSCLC with EGFR mutation), Cetuximab (KRAS wild-type colorectal cancer).
- Angiogenesis inhibitors: Bevacizumab (anti-VEGF mAb), Sunitinib (multi-TKI).
- Immune Checkpoint Inhibitors: Pembrolizumab (anti-PD-1), Ipilimumab (anti-CTLA-4) boost T-cell activity.
- BRAF inhibitors: Vemurafenib for melanoma with BRAF V600E mutation.
- PARP inhibitors: Olaparib for BRCA-mutated ovarian/prostate cancers.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

