Cell Cycle - Cancer's Clockwork
- Phases & Key Events:
- G0: Quiescent/Resting.
- G1: Growth, protein/RNA synth. G1/S checkpoint (p53).
- S: DNA synthesis.
- G2: Pre-mitotic growth. G2/M checkpoint.
- M: Mitosis (Prophase, Metaphase, Anaphase, Telophase).
- Drug Classification:
- Cell Cycle Specific (CCS): Target specific phases.
- S-phase: Antimetabolites (Methotrexate, 5-FU), Etoposide.
- M-phase: Vinca alkaloids (Vincristine), Taxanes (Paclitaxel). 📌 Microtubules Targeted in M-phase.
- G2-phase: Bleomycin.
- Cell Cycle Non-Specific (CCNS): Act on all phases (incl. G0). E.g., Alkylating agents, Cisplatin.
- Cell Cycle Specific (CCS): Target specific phases.
- Log-Kill Hypothesis: Kills constant fraction of cells/cycle (e.g., 3-log kill: $10^{12} \rightarrow 10^9$ cells).

⭐ Most human solid tumors have a low growth fraction, making them more sensitive to CCNS drugs or requiring recruitment strategies for CCS drugs.
Treatment Aims - Strategic Strikes
-
Primary Goals of Chemotherapy:
- Cure: Complete eradication of all neoplastic cells.
- Possible in select cancers (e.g., testicular, Hodgkin's, ALL, choriocarcinoma).
- Control: Prevent further tumor growth and spread when cure is unlikely.
- Aims to prolong survival and maintain patient's Quality of Life (QoL).
- Palliation: Alleviate symptoms (e.g., pain, obstruction) and improve QoL in advanced, incurable cancers.
- Cure: Complete eradication of all neoplastic cells.
-
Key Therapeutic Strategies & Timing:
⭐ Gompertzian Growth Model: Tumor growth fraction is highest when small, decreasing as tumor enlarges. Chemotherapy is most effective against rapidly dividing cells (high growth fraction), explaining why smaller tumors/micrometastases are more chemosensitive.
Drug Resistance - Cancer's Defenses
- Types:
- Primary (Intrinsic): No prior drug exposure.
- Acquired: Develops post-exposure.
- Mechanisms:
- ↓ Drug influx/uptake (e.g., methotrexate).
- ↑ Drug efflux: P-glycoprotein (MDR1 gene) pumps drugs out. 📌 MDR1: Many Drugs Removed.

- Altered drug targets (e.g., enzyme mutation, gene amplification).
- ↑ Drug inactivation (e.g., glutathione conjugation).
- ↓ Drug activation (pro-drugs not converted).
- ↑ DNA repair mechanisms.
- Evasion of apoptosis (e.g., p53 mutation, ↑Bcl-2).
- Multidrug Resistance (MDR): Cross-resistance to multiple, structurally unrelated drugs.
⭐ P-glycoprotein (encoded by ABCB1 or MDR1 gene) is a major cause of MDR, effluxing natural product drugs like anthracyclines, vinca alkaloids, and taxanes.
- Overcoming Resistance:
- Combination chemotherapy.
- High-dose therapy + rescue (e.g., Leucovorin for Methotrexate).
- MDR modulators (e.g., verapamil; limited by toxicity).
- Targeted therapies bypassing resistance mechanisms.
Side Effects - Battle Scars
- Common (Rapidly Dividing Cells):
- GIT: Nausea, vomiting (CTZ), mucositis, diarrhea.
- Bone Marrow Suppression (BMS):
- Neutropenia (ANC < 500/µL ↑infection risk).
- Thrombocytopenia (↑bleeding).
- Anemia.
- Alopecia.
- Gonadal dysfunction: Infertility.
- Key Organ-Specific Toxicities:
- Cardio: Doxorubicin (cumulative; Dexrazoxane).
- Nephro: Cisplatin (hydrate, amifostine), Methotrexate (high dose).
- Neuro: Vincristine (peripheral), Cisplatin (ototoxicity, peripheral).
- Pulmonary: Bleomycin, Busulfan.
- Hemorrhagic Cystitis: Cyclophosphamide/Ifosfamide (Acrolein metabolite; MESNA, hydration). 📌 "MESNA for Cyclo's Bladder"
- Tumor Lysis Syndrome (TLS):
- Lab: ↑K⁺, ↑PO₄³⁻, ↑Uric acid, ↓Ca²⁺.
- Manage: Hydration, allopurinol/rasburicase.
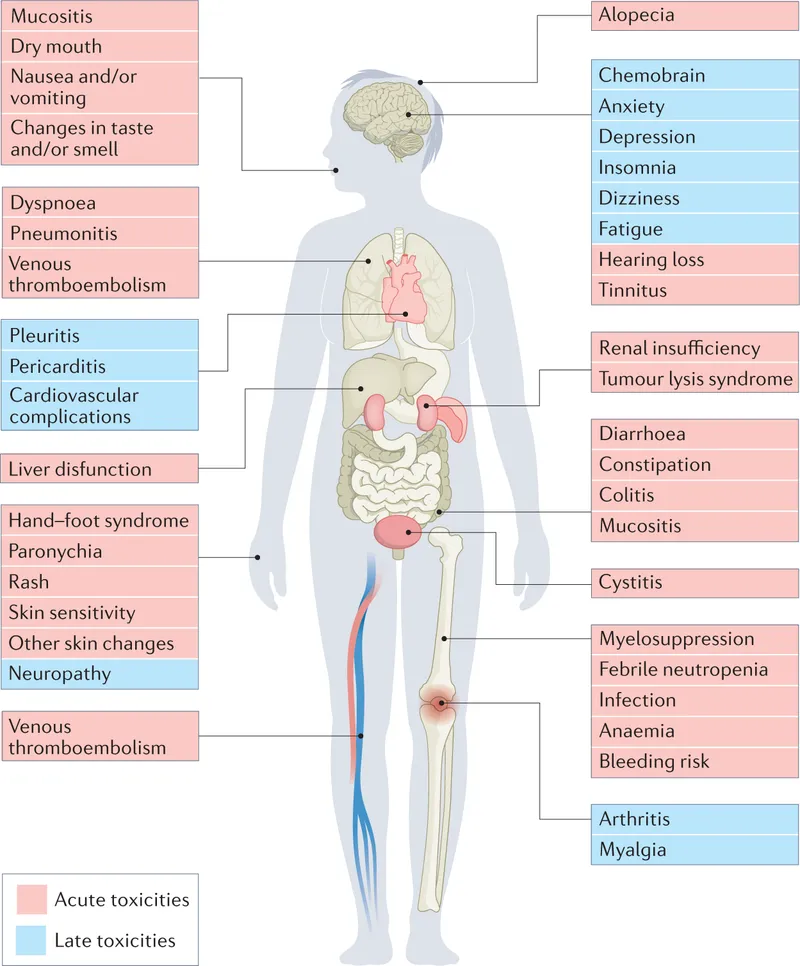
⭐ Cisplatin: major dose-limiting toxicities are nephrotoxicity and ototoxicity. Amifostine & hydration for nephroprotection.
High‑Yield Points - ⚡ Biggest Takeaways
- Cell cycle-specific (CCS) drugs (e.g., antimetabolites, vincas) target dividing cells; CCNS drugs act on all cells.
- Log-kill hypothesis: Chemotherapy kills a constant fraction of tumor cells per cycle.
- Combination therapy is key: ↑ efficacy, ↓ resistance, manages toxicity, targets heterogeneity.
- Drug resistance mechanisms: P-glycoprotein efflux, altered targets, ↑ DNA repair, ↓ drug activation.
- Major dose-limiting toxicities: myelosuppression, mucositis, nausea/vomiting.
- Sanctuary sites (e.g., CNS, testes) hinder drug penetration and efficacy.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

