Hemoglobinopathies - Basics & Blood Bonds
- Hemoglobinopathies: Genetic disorders affecting hemoglobin (Hb) structure or synthesis.
- Qualitative: Abnormal globin chain structure (e.g., Sickle Cell Disease).
- Quantitative: Reduced synthesis of normal globin chains (e.g., Thalassemia).
- Normal Adult Hemoglobins:
- HbA ($α_2β_2$): >95%
- HbA2 ($α_2δ_2$): 1.5-3.5%
- HbF ($α_2γ_2$): <1-2% (higher in infants)
- Inheritance: Most are Autosomal Recessive.
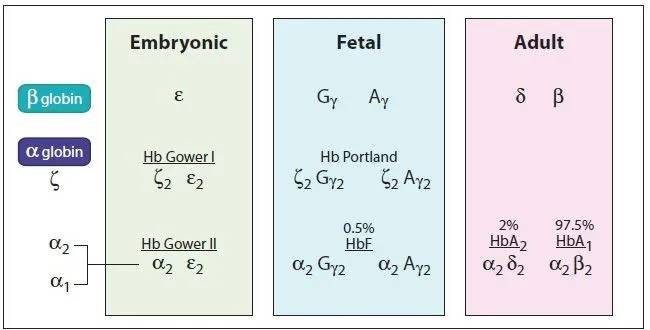
⭐ HbF is the predominant hemoglobin during fetal life, which gradually decreases after birth as HbA production increases.
Sickle Cell Syndromes - Crescent Calamity
- Autosomal recessive; β-globin gene mutation (GAG→GTG; Glu→Val at codon 6) → HbS.
- Pathophysiology: Deoxygenation → HbS polymerization → RBC sickling → microvascular occlusion, hemolysis.
- Clinical Features:
- Vaso-occlusive crisis (VOC): Severe pain.
- Acute Chest Syndrome (ACS): Fever, cough, chest pain, hypoxia.
- Splenic sequestration (infants) → autosplenectomy (adults) → ↑ risk: encapsulated organisms (S. pneumo, H. flu, N. menin).
- Aplastic crisis (Parvovirus B19).
- Dactylitis (hand-foot syndrome; often 1st sign).
- Stroke, priapism, avascular necrosis.
- Diagnosis:
- Newborn screening.
- Hb electrophoresis/HPLC: HbS predominant, ↑ HbF, no HbA (in HbSS).
- Peripheral smear: Sickled cells, Howell-Jolly bodies.

- Management:
- Hydroxyurea: ↑ HbF, ↓ VOCs.
- Folic acid supplementation.
- Vaccinations (Pneumococcal, etc.).
- Pain control, hydration.
- Transfusions.
- Stem cell transplant (curative).
⭐ Howell-Jolly bodies on peripheral smear indicate splenic dysfunction/asplenia.
Thalassemias - Chain Chain Woes
Quantitative defect in globin chain synthesis (α or β). Results in microcytic hypochromic anemia.
-
Alpha (α) Thalassemia: Due to gene deletions on chromosome 16 (αα/αα).
- 1-2 deletions (-α/αα or -α/-α or --/αα): Silent carrier or α-thalassemia trait. Usually asymptomatic or mild microcytic anemia.
- 3 deletions (--/-α): HbH disease. Excess β chains form HbH (β4). Chronic hemolytic anemia, jaundice, splenomegaly. Golf ball inclusions (Heinz bodies) with supravital stain.
- 4 deletions (--/--): Hb Barts. Excess γ chains form Hb Barts (γ4). Severe intrauterine hypoxia, hydrops fetalis. Usually fatal.
- Diagnosis: Genetic testing. Hb electrophoresis shows HbH or Hb Barts.
-
Beta (β) Thalassemia: Due to point mutations in β-globin gene on chromosome 11.
- β-Thalassemia Minor (Trait): Heterozygous (β/β⁰ or β/β⁺). Mild asymptomatic anemia. ↑HbA2 (>3.5%), sometimes ↑HbF.
- β-Thalassemia Intermedia: Homozygous mild or compound heterozygous moderate mutations. Variable severity, may not require regular transfusions.
- β-Thalassemia Major (Cooley's Anemia): Homozygous (β⁰/β⁰) or severe compound heterozygous (β⁰/β⁺). Severe transfusion-dependent anemia by 4-6 months. Massive hepatosplenomegaly, bone marrow expansion (frontal bossing, chipmunk facies, "crew-cut" on X-ray), iron overload.
- Diagnosis: Hb electrophoresis (↓/absent HbA, ↑↑HbF, variable ↑HbA2).
-
Management (Moderate/Severe):
- Regular packed RBC transfusions (maintain Hb >9-10 g/dL).
- Iron chelation therapy (e.g., Deferoxamine, Deferasirox, Deferiprone) to prevent iron overload complications.
- Folic acid supplementation.
- Splenectomy (if hypersplenism).
- Allogeneic hematopoietic stem cell transplantation (HSCT) is curative.

⭐ Target cells (codocytes) are a characteristic, though not specific, finding on peripheral smear in thalassemias due to relative membrane redundancy.
Diagnostic Approach & Other Variants - Spotting the Suspects
- Screening: CBC (↓Hb, abnormal MCV/MCH), Retic count, Peripheral Smear (RBC morphology: target cells, sickled cells, crystals).
- Confirmation: HPLC (gold standard), Hb Electrophoresis. Genetic tests for unclear/atypical cases.
- Newborn Screening (NBS): Crucial for early detection (SCA, β-Thal Major), allows timely intervention.

- Key Variants:
- HbC: Mild hemolysis, target cells, HbC crystals (rod-like).
- HbE: SE Asia; mild microcytic anemia; HbE co-migrates with HbA2.
- HbD-Punjab: Often asymptomatic; co-migrates with HbS (differentiate via solubility test - HbD is soluble).
⭐ On alkaline electrophoresis (pH 8.6), Hb migration: A > F > S/D/G > E/O/C (Anode to Cathode). 📌 Mnemonic: All Fine Sick Doctors Go East Or Centre.
High‑Yield Points - ⚡ Biggest Takeaways
- Sickle Cell Anemia (SCA): HbS (β-globin Glu→Val), autosomal recessive. Features vaso-occlusive crises; hydroxyurea ↑HbF.
- β-Thalassemia Major: Absent/reduced β-globin chains cause severe microcytic hypochromic anemia; transfusion-dependent with iron chelation.
- α-Thalassemia: α-globin gene deletions lead to Hb Bart's (hydrops fetalis) or HbH disease.
- Diagnosis: HPLC is gold standard. Newborn screening is key.
- SCA Complications: Aplastic crisis (Parvovirus B19), risk of sepsis (prophylaxis needed).
- HbE Trait/Disease: Common in NE India/SE Asia; often mild. HbE/β-thalassemia is severe_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

