Diseases of the Cornea

On this page
🏗️ Corneal Architecture: The Five-Layer Fortress
Master corneal anatomy, and you unlock the logic behind every keratitis, dystrophy, and surgical complication. The cornea's five distinct layers form an optical and barrier system so precise that even 1 mm of edema destroys transparency. Understanding each layer's cellular machinery, metabolic demands, and failure modes transforms pattern recognition across infectious, degenerative, and traumatic pathology.
The cornea achieves what no other tissue can: perfect transparency combined with structural resilience. This 550-micron thick window maintains its clarity through precise cellular architecture, active metabolic pumps consuming 30% of corneal oxygen, and an avascular design that relies entirely on diffusion. Each layer contributes distinct mechanical properties-epithelial regeneration completes in 5-7 days, while endothelial cells never regenerate after birth, making their density of 2500-3000 cells/mm² a non-renewable resource that declines 0.6% annually.
📌 Remember: ABCDE layers from front to back-Anterior epithelium, Bowman's layer, Corneal stroma (substantia propria), Descemet's membrane, Endothelium. Bowman's never regenerates (surgical scars permanent), Descemet's regenerates continuously (explains thickening with age from 3 microns at birth to 10-12 microns by age 80).

Cellular Organization: The Workforce Distribution
-
Epithelial Stratification (50-micron thickness, 40% of refractive power)
- Superficial cells: 2-3 layers of flattened squames with tight junctions (zonulae occludentes) creating barrier function
- Wing cells: 2-3 layers providing transition zone with decreasing metabolic activity
- Basal columnar cells: Single layer of mitotic cells (10-14% turnover rate weekly) anchored to basement membrane
- Hemidesmosomes attach to type IV collagen and laminin
- Complete epithelial renewal cycle: 5-7 days from limbus to center
- Limbal stem cells in palisades of Vogt provide continuous replenishment
- Metabolic demands: Glycolysis predominates (85% of glucose), minimal oxidative phosphorylation
-
Stromal Keratocytes (500-micron thickness, 90% of corneal volume)
- Density: 300-400 cells/mm³ with highest concentration in anterior third
- Modified fibroblasts maintaining extracellular matrix homeostasis
- Collagen type I fibrils arranged in 200-250 lamellae with orthogonal orientation
- Fibril diameter: 25-35 nm with precise 64-nm spacing (critical for transparency)
- Proteoglycans (keratan sulfate, dermatan sulfate) maintain interfibrillar spacing
- Metabolic activity: 70% aerobic respiration, dependent on oxygen diffusion from tears and aqueous
- Wound response: Transform to fibroblasts/myofibroblasts causing scarring
-
Endothelial Hexagonal Mosaic (5-micron thickness)
- Birth density: 3500-4000 cells/mm² declining to 2500-3000 cells/mm² in adults
- Critical threshold: 500-800 cells/mm² before corneal decompensation
- No mitotic activity post-birth; cells enlarge (polymegathism) and lose hexagonal shape (pleomorphism) to cover defects
- Na⁺-K⁺-ATPase pumps on basolateral membrane maintain 78% stromal deturgescence
- Pump failure causes stromal edema, epithelial microcysts, and Descemet's folds
- Metabolic demands: Highest ATP consumption per cell in cornea
⭐ Clinical Pearl: Endothelial cell density below 1000 cells/mm² predicts post-cataract surgery decompensation in 15-20% of cases. Specular microscopy showing >50% polymegathism or coefficient of variation >40% indicates compromised pump function even with adequate density.
| Layer | Thickness | Regeneration | Key Protein | Clinical Failure | Detection Method |
|---|---|---|---|---|---|
| Epithelium | 50 μm | Complete in 5-7 days | Keratin K3/K12 | Recurrent erosions, basement membrane disease | Fluorescein staining, OCT |
| Bowman's | 8-14 μm | Never regenerates | Collagen I/III | Anterior dystrophies, LASIK flap interface | Slit-lamp, confocal microscopy |
| Stroma | 500 μm | Scar formation only | Collagen I | Keratitis, ectasia, edema | Pachymetry, topography |
| Descemet's | 3-12 μm | Continuous growth | Collagen IV/VIII | Breaks (Haab's striae), detachment | Gonioscopy, OCT |
| Endothelium | 5 μm | Enlargement only | Na⁺-K⁺-ATPase | Fuchs' dystrophy, decompensation | Specular microscopy |

Transparency Mechanisms: The Optical Engineering
The cornea's transparency depends on three critical factors working in concert-any disruption causes immediate light scattering:
-
Lattice Theory of Collagen Arrangement
- Stromal collagen fibrils maintain uniform 25-35 nm diameter and precise 64-nm interfibrillar spacing
- Orthogonal lamellar orientation causes destructive interference of scattered light
- Proteoglycans (primarily keratan sulfate with 25-30 disaccharide repeats) maintain hydration-dependent spacing
- Disruption mechanisms:
- Edema increases spacing beyond 70 nm → light scattering appears clinically as haze
- Inflammation deposits irregular collagen → permanent scarring
- Ectasia disrupts lamellar organization → irregular astigmatism
-
Relative Stromal Dehydration (maintained at 78% water content)
- Endothelial pump function: Na⁺-K⁺-ATPase creates osmotic gradient pulling water from stroma
- Epithelial barrier: Tight junctions prevent tear film water entry
- Stromal swelling pressure: 40-60 mmHg constantly opposed by pump activity
- Pump failure sequence:
- Stromal edema appears at >85% hydration (thickness increases >600 microns)
- Epithelial edema follows at >90% hydration (microcystic changes)
- Bullous keratopathy develops when pumps fail completely
-
Avascularity Maintenance
- Anti-angiogenic factors: Endostatin, thrombospondin-1, PEDF (pigment epithelium-derived factor)
- Limbal barrier prevents vascular invasion from conjunctival vessels
- Corneal privilege lost with inflammation: VEGF upregulation overcomes anti-angiogenic balance
- Clinical threshold: >2 mm of neovascularization significantly reduces transplant success by 30-40%
💡 Master This: Corneal edema follows a predictable sequence-stromal thickening precedes epithelial changes by 12-24 hours. Early morning blurred vision improving throughout the day indicates borderline endothelial function: overnight lid closure reduces evaporation, increasing hydration beyond pump capacity. Measure pachymetry: central thickness >640 microns confirms subclinical edema even with clear cornea.
Innervation Density: The Sensory Powerhouse
The cornea possesses the highest sensory nerve density of any human tissue-300-600 times more sensitive than skin, with 7000 nociceptors/mm² in the central epithelium:
-
Nerve Architecture
- Origin: Long ciliary nerves from nasociliary branch of ophthalmic division (V1)
- Stromal entry: 60-80 nerve trunks penetrate peripheral stroma at mid-depth
- Subbasal plexus: Dense network 5-10 microns below epithelium with 20-25 mm/mm² nerve density
- Branches penetrate epithelium losing myelin sheaths
- Terminal nerve endings reach superficial cell layer
- In vivo confocal microscopy visualizes this plexus directly
- Nerve density gradient: Central > peripheral by 3-4 fold
-
Functional Consequences
- Blink reflex arc: Afferent V1 → efferent VII (facial nerve) with 40-60 ms latency
- Tear secretion: Trigeminal stimulation → lacrimal gland via parasympathetic fibers
- Trophic support: Neuropeptides (substance P, CGRP) maintain epithelial health
- Denervation causes neurotrophic keratopathy: epithelial breakdown, poor healing
- Diabetes reduces nerve density by 30-50% before clinical neuropathy
- Herpes simplex destroys nerves → persistent epithelial defects
-
Clinical Testing Thresholds
- Cochet-Bonnet aesthesiometry: Normal >55 mm filament detection
- Reduced sensation <40 mm predicts neurotrophic complications
- Complete anesthesia: Risk of exposure keratopathy, perforation without pain warning
⭐ Clinical Pearl: Unilateral reduced corneal sensation in a patient with red eye suggests herpes simplex keratitis until proven otherwise-90% of HSV keratitis cases show decreased sensation compared to fellow eye. Test both eyes systematically; asymmetry >15 mm on Cochet-Bonnet is pathological.

Metabolic Pathways: Powering the Fortress
Corneal metabolism balances transparency requirements (no blood vessels) with high energy demands for active transport and protein synthesis:
-
Oxygen Supply Chain
- Epithelium sources: 60% from atmosphere through tear film, 40% from aqueous humor
- Oxygen consumption: 3-4 μL O₂/cm²/hour (epithelium uses 50%, endothelium 20%, stroma 30%)
- Contact lens hypoxia threshold: <10 mmHg oxygen tension causes epithelial edema
- Modern silicone hydrogel lenses: >100 Dk/t (oxygen transmissibility) prevents hypoxia
- PMMA lenses: 0 Dk/t caused severe chronic hypoxia, now obsolete
- Stromal oxygen: Diffuses from limbal vessels and aqueous; central stroma most vulnerable
-
Glucose Metabolism Pathways
- Primary source: Aqueous humor glucose concentration 2.5-5.0 mmol/L (80% of plasma levels)
- Epithelial metabolism: 85% anaerobic glycolysis (lactate production), 15% pentose phosphate pathway
- Glycolysis generates 2 ATP per glucose rapidly for Na⁺-K⁺-ATPase pumps
- Pentose phosphate pathway produces NADPH for glutathione reduction (antioxidant defense)
- Endothelial metabolism: >90% aerobic respiration (Krebs cycle, oxidative phosphorylation)
- Generates 36-38 ATP per glucose to power ion pumps
- Explains endothelial vulnerability to hypoxia and mitochondrial toxins
-
Hydration Control Mechanisms
- Endothelial pump: Na⁺-K⁺-ATPase consumes >50% of endothelial ATP production
- Bicarbonate-dependent mechanism: Carbonic anhydrase facilitates HCO₃⁻ transport
- Epithelial barrier resistance: 2000-3000 Ω·cm² prevents passive water influx
- Clinical manipulation: Hyperosmotic agents (5% NaCl drops/ointment) temporarily reduce edema by increasing tear osmolarity to >400 mOsm/L
💡 Master This: Endothelial pump failure from Fuchs' dystrophy follows a 20-30 year progression: guttae formation → decreased cell density → early morning blur (overnight edema) → persistent edema → epithelial bullae → pain. Central pachymetry monitoring guides intervention: >640 microns indicates pump insufficiency, >700 microns predicts bullous keratopathy within 2-3 years without transplantation.
Corneal metabolism's dependence on diffusion rather than perfusion makes it uniquely vulnerable to hypoxia, yet resistant to vascular inflammation. Connect these metabolic principles through to understand how tear film instability disrupts oxygen delivery, and through to grasp why endothelial cell density determines graft survival.
🏗️ Corneal Architecture: The Five-Layer Fortress
🔬 Epithelial Dynamics: The Renewable Shield
The corneal epithelium operates as a self-renewing fortress, completely replacing itself every 5-7 days through a precisely orchestrated migration and differentiation cascade originating from limbal stem cells. This 50-micron thick stratified squamous barrier provides 80% of the cornea's barrier function while contributing 40% of total refractive power. Understanding epithelial cell kinetics, adhesion mechanisms, and failure modes predicts healing patterns in trauma, infection, and dystrophies.
Epithelial homeostasis depends on three synchronized processes: stem cell activation at the limbus, centripetal migration toward the corneal center, and vertical differentiation from basal to superficial layers. Disruption at any level causes distinct pathology-limbal stem cell deficiency produces conjunctivalization, basement membrane disease causes recurrent erosions, and neurotrophic damage halts healing despite intact stem cells.
📌 Remember: STEM for epithelial renewal-Stem cells in limbal palisades, Transit amplifying cells migrate centripetally, Epithelial differentiation proceeds vertically, Maturation completes at superficial layer with tight junctions. Entire cycle 5-7 days; disruption at any step produces specific pathology patterns.
Limbal Stem Cell Niche: The Source Code
The limbus harbors corneal epithelial stem cells in specialized microenvironments called palisades of Vogt, visible as radial ridges at the corneoscleral junction:
-
Stem Cell Location and Characteristics
- Palisades of Vogt: 0.5-1.0 mm wide interpalisade zones containing stem cells
- Higher density in superior and inferior limbus (60-70% of total) vs nasal/temporal
- Stem cell markers: ΔNp63α (nuclear transcription factor), ABCG2 (ATP-binding cassette transporter), keratin 15
- Absence of differentiation markers: No keratin K3/K12 expression
- Slow-cycling: Retain BrdU label for >4 weeks (label-retaining cells)
- Population size: 500-1000 stem cells per mm of limbal circumference
- Clinical threshold: >3 clock hours of limbal damage causes stem cell deficiency
-
Transit Amplifying Cell Cascade
- Stem cell division produces: 1 stem cell + 1 transit amplifying cell (asymmetric division)
- Transit amplifying cells: Undergo 4-5 divisions while migrating centripetally
- Migration rate: 0.12-0.14 mm/day from limbus to center
- Centripetal movement: Demonstrated by X-Y-Z marking studies showing peripheral-to-central flow
- Complete corneal surface replacement from limbal origin: 5-7 days
- Vertical differentiation occurs simultaneously during migration
- Basal cell turnover: 10-14% weekly mitotic rate maintains population
-
Limbal Microenvironment Protection
- Vascular support: Limbal arcades provide nutrients, growth factors (EGF, KGF, HGF)
- Melanocytes: Absorb UV radiation protecting stem cell DNA from mutation
- Extracellular matrix: Specialized basement membrane with laminin-10, nidogen-2
- Clinical vulnerability: Chemical burns, Stevens-Johnson syndrome, contact lens overwear destroy niche
⭐ Clinical Pearl: Limbal stem cell deficiency (LSCD) presents with conjunctival epithelium growing onto cornea (conjunctivalization), visible as whorl-like patterns with goblet cells on impression cytology. Fluorescein shows late staining as conjunctival epithelium is more permeable. Severe LSCD (>6 clock hours affected) requires stem cell transplantation; autologous CLET (cultivated limbal epithelial transplantation) shows 70-75% success at 2 years.

Adhesion Complex Architecture: Anchoring the Layers
Epithelial stability depends on three adhesion systems-each failure mode produces distinct clinical pathology:
-
Hemidesmosomes: Basal Cell-Basement Membrane Attachment
- Structure: Integrin α6β4 links intracellular keratin filaments to extracellular laminin-332
- Components: Plectin, BP180 (collagen XVII), BP230 (dystonin)
- Density: 1200-1500 hemidesmosomes per basal cell
- Maturation time: 48-72 hours after epithelial resurfacing
- Explains early erosion risk after epithelial debridement
- Bandage contact lens protection until maturation complete
- Genetic defects: Epidermolysis bullosa causes fragile epithelial adhesion
-
Basement Membrane Complex
- Composition: Type IV collagen, laminin-332, nidogen, perlecan
- Thickness: 40-60 nm with lamina lucida and lamina densa zones
- Anchoring fibrils: Type VII collagen extends from basement membrane into Bowman's layer
- Turnover: Continuous remodeling by matrix metalloproteinases (MMP-2, MMP-9)
- Pathology: Basement membrane dystrophy shows multilaminar thickening with abnormal collagen deposition
-
Recurrent Corneal Erosion Mechanism
- Primary cause: Defective hemidesmosome formation or basement membrane abnormalities
- Trauma-induced: 30-50% follow epithelial abrasion, symptoms begin weeks to months later
- Dystrophy-associated: Epithelial basement membrane dystrophy (map-dot-fingerprint) in 40-60% of cases
- Clinical pattern: Early morning pain (overnight lid adhesion tears loose epithelium)
- Pathophysiology: Hemidesmosomes fail to mature → epithelium slides on basement membrane → spontaneous erosions
- Healing interventions target adhesion:
- Hypertonic saline 5%: Dehydrates epithelium, promotes hemidesmosome formation
- Anterior stromal puncture: Creates focal scarring for mechanical adhesion
- Phototherapeutic keratectomy (PTK): Removes abnormal basement membrane, 85-90% cure rate
💡 Master This: Recurrent erosion syndrome follows a 48-72 hour vulnerability window after any epithelial injury-the time required for hemidesmosome maturation. Patients report "waking with severe pain" because overnight lid closure increases epithelial hydration and adhesion to lid; morning eye opening tears incompletely attached epithelium. Prophylactic hyperosmotic ointment at bedtime for 3-6 months prevents recurrence in 70-80% of post-traumatic cases.
Epithelial Barrier Function: The Selective Gate
The epithelial barrier maintains corneal hydration and prevents pathogen entry through sophisticated tight junction networks:
-
Tight Junction Molecular Architecture
- Location: Between superficial epithelial cells (2-3 layers)
- Proteins: Occludin, claudins (1, 4, 7), ZO-1, ZO-2 (zonula occludentes)
- Function: Create size-selective and charge-selective barrier
- Electrical resistance: 2000-3000 Ω·cm² (high resistance = tight barrier)
- Permeability: Allows molecules <500 Da to pass; blocks larger molecules and ions
-
Barrier Compromise Patterns
- Dry eye disease: Hyperosmolarity >316 mOsm/L disrupts tight junctions
- Increased permeability allows fluorescein penetration (positive staining)
- Inflammatory cytokines (IL-1, TNF-α) downregulate occludin expression
- Contact lens wear: Chronic hypoxia reduces ZO-1 expression by 30-40%
- Infection: Bacterial toxins and proteases actively degrade junction proteins
- Pseudomonas aeruginosa exotoxin A: Disrupts tight junctions within 2-4 hours
- Dry eye disease: Hyperosmolarity >316 mOsm/L disrupts tight junctions
-
Wound Healing Response Cascade
- Phase 1 (0-6 hours): Cell migration-adjacent cells flatten and slide to cover defect
- No mitosis initially; movement driven by lamellipodia and filopodia
- Migration rate: 60-80 microns/hour for small defects
- Phase 2 (6-24 hours): Cell proliferation-mitotic activity increases 5-10 fold
- Growth factors: EGF, TGF-α, KGF released from tears and limbal vessels
- Basal cells divide; daughter cells migrate superficially
- Phase 3 (24-72 hours): Differentiation and stratification
- Cells organize into basal, wing, and superficial layers
- Tight junctions reform at superficial layer
- Phase 4 (72 hours-7 days): Hemidesmosome maturation
- Integrin clustering and basement membrane remodeling
- Complete adhesion strength returns by 7 days
- Phase 1 (0-6 hours): Cell migration-adjacent cells flatten and slide to cover defect
⭐ Clinical Pearl: Epithelial defect healing rate predicts underlying pathology-normal epithelium heals 1-1.5 mm²/day. Defects healing slower than 0.5 mm²/day suggest neurotrophic keratopathy, limbal stem cell deficiency, or persistent toxic exposure. Defects >10 mm² or persisting >2 weeks require aggressive intervention: bandage contact lens, autologous serum tears (growth factor supplementation), or amniotic membrane transplantation.
| Adhesion Structure | Primary Components | Maturation Time | Failure Pathology | Clinical Detection |
|---|---|---|---|---|
| Hemidesmosomes | Integrin α6β4, BP180, laminin-332 | 48-72 hours | Recurrent erosions, epidermolysis bullosa | Electron microscopy, genetic testing |
| Tight Junctions | Occludin, claudins, ZO-1/2 | 24-48 hours | Barrier failure, fluorescein penetration | Fluorescein staining, permeability assays |
| Desmosomes | Desmoglein, desmocollin, plakoglobin | 12-24 hours | Acantholysis (rare in cornea) | Immunofluorescence |
| Basement Membrane | Collagen IV, laminin, nidogen | 7-14 days | EBMD, recurrent erosions | Confocal microscopy, OCT |
| Anchoring Fibrils | Collagen VII | 14-21 days | Epithelial-stromal separation | Electron microscopy |
Understanding epithelial dynamics explains why produces dendritic ulcers that heal slowly (viral cytopathic effect disrupts migration), why causes punctate epitheliopathy (barrier dysfunction from hyperosmolarity), and why patients need extended prophylaxis (hemidesmosome maturation delay).
🔬 Epithelial Dynamics: The Renewable Shield
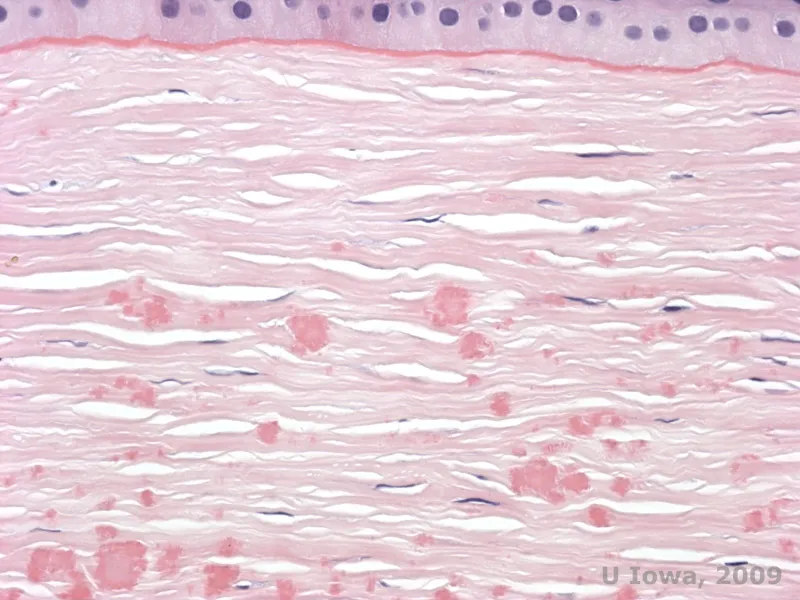
🌊 Stromal Biomechanics: The Structural Core
The corneal stroma constitutes 90% of corneal thickness (~500 microns) and provides essential biomechanical strength while maintaining optical transparency through precise collagen architecture. This acellular matrix of 200-250 collagen lamellae arranged in orthogonal patterns withstands intraocular pressure averaging 15 mmHg while allowing >90% light transmission. Stromal keratocytes maintain this delicate balance, producing extracellular matrix components and responding to injury with wound healing that often sacrifices transparency for structural integrity.
Stromal biomechanics determine corneal shape, refractive power, and resistance to ectasia. The anterior 40% of stroma contains 2-3 times more interwoven lamellae than posterior stroma, explaining why LASIK flaps (created at 90-120 microns depth) cause more biomechanical weakening than PRK (removes 50-80 microns). Understanding stress-strain relationships, hydration effects, and collagen cross-linking principles guides management of keratoconus, post-refractive surgery ectasia, and corneal edema.
📌 Remember: LAMELLAR for stromal organization-Lamellae 200-250 layers, Anterior third most interwoven, Mechanical strength from orthogonal arrangement, Extracellular matrix spacing 64 nm critical, Light scatter prevented by uniform fibril diameter, Lamellar keratoplasty respects depth-dependent properties, Anterior stroma biomechanically strongest, Refractive surgery weakens anterior stroma most. Anterior 1/3 provides 60-70% of total stromal strength.
Collagen Architecture: The Optical Lattice
Stromal transparency depends on collagen fibril organization following Maurice's lattice theory-uniform spacing and diameter prevent light scattering:
-
Collagen Fibril Specifications
- Type I collagen: >90% of stromal collagen (type V collagen 5-10% regulates fibril diameter)
- Fibril diameter: 25-35 nm (remarkably uniform, coefficient of variation <15%)
- Interfibrillar spacing: 64 nm (maintained by proteoglycans)
- Critical threshold: Spacing >70 nm causes visible light scatter (clinically: haze)
- Edema increases spacing proportionally to water content increase
- Fibril length: >1 cm continuous through multiple lamellae
- Lateral organization: Quasi-hexagonal packing within each lamella
-
Lamellar Organization Patterns
- Total lamellae: 200-250 layers spanning stromal thickness
- Lamellar thickness: 1.5-2.5 microns each
- Orientation: Orthogonal arrangement-adjacent lamellae cross at ~90 degrees
- Anterior stroma: Highly interwoven with frequent branching and interweaving
- Posterior stroma: Parallel lamellae with less interweaving
- Preferential orientation: Nasal-temporal and superior-inferior directions predominate
- Explains astigmatism patterns in keratoconus (inferior steepening)
- Biomechanical consequence: Orthogonal arrangement distributes stress multidirectionally
-
Proteoglycan Spacing Control
- Keratan sulfate: 65% of corneal proteoglycans (primarily lumican, keratocan, mimecan)
- Dermatan sulfate: 30% of proteoglycans (decorin predominates)
- Charge repulsion: Negatively charged glycosaminoglycan chains maintain interfibrillar distance
- Hydration regulation: Proteoglycans bind water molecules but prevent excessive swelling
- Pathological states:
- Mucopolysaccharidoses: Abnormal proteoglycan accumulation → corneal clouding
- Macular corneal dystrophy: Keratan sulfate deficiency → irregular spacing → opacity
⭐ Clinical Pearl: Corneal haze after PRK correlates with depth of ablation and healing response-ablations >100 microns (treating myopia >-6.00 D) produce clinically significant haze (grade 2+) in 15-20% of cases. Prophylactic mitomycin-C 0.02% applied for 12-30 seconds reduces haze incidence to <5% by inhibiting keratocyte transformation to myofibroblasts (the haze-producing cell type).
Biomechanical Properties: Engineering the Cornea
The cornea functions as a pressurized shell resisting IOP while maintaining precise curvature for optical power:
-
Stress-Strain Relationships
- Young's modulus (stiffness): 0.3-0.5 MPa in physiologic range
- Increases non-linearly with strain (stress-stiffening behavior)
- Anterior stroma: 2-3 times stiffer than posterior stroma
- Tensile strength: 3.8-5.0 MPa at failure
- Hysteresis: Cornea exhibits viscoelastic behavior-loading/unloading curves differ
- Corneal hysteresis (CH): 10.7 ± 1.8 mmHg measured by ocular response analyzer
- Low CH (<9 mmHg) predicts keratoconus progression risk
- High CH (>12 mmHg) indicates stiffer cornea, lower ectasia risk
- Young's modulus (stiffness): 0.3-0.5 MPa in physiologic range
-
Depth-Dependent Strength Distribution
- Anterior 1/3 (0-165 microns): 60-70% of total biomechanical strength
- High lamellar interweaving creates stress distribution network
- LASIK flap creation at 100-120 microns removes 30-40% of corneal strength
- Middle 1/3 (165-330 microns): 20-25% of strength
- Posterior 1/3 (330-500 microns): 10-15% of strength
- Parallel lamellae provide less multidirectional resistance
- Clinical implications:
- PRK (surface ablation): Preserves anterior lamellar integrity, less weakening
- SMILE (small incision lenticule extraction): Preserves anterior 100-120 microns, maintains >80% biomechanical strength
- Deep anterior lamellar keratoplasty (DALK): Preserves Descemet's and endothelium but removes all stromal strength
- Anterior 1/3 (0-165 microns): 60-70% of total biomechanical strength
-
Hydration Effects on Mechanics
- Normal hydration: 78% water content (4:1 water:dry weight ratio)
- Swelling pressure: 40-60 mmHg constantly opposed by endothelial pump
- Hydration-stiffness relationship: Edematous cornea 50-70% softer than normal
- Pachymetry >600 microns indicates decreased stiffness
- Ectasia risk increases with edema (transient or chronic)
- Riboflavin-UV collagen cross-linking (CXL): Increases stiffness by 70-300%
- Creates covalent bonds between collagen molecules
- Halts keratoconus progression in 90-95% of cases
- Requires minimum stromal thickness 400 microns (after epithelial removal) to avoid endothelial toxicity
💡 Master This: Iatrogenic ectasia risk after LASIK depends on residual stromal bed thickness and biomechanical quality-minimum safe residual bed is 250-280 microns, but this assumes normal preoperative biomechanics. Calculate total tissue altered (TTA): flap thickness + ablation depth. TTA >40% of corneal thickness significantly increases ectasia risk. Preoperative screening must identify forme fruste keratoconus: topography showing inferior steepening, pachymetry showing inferior thinning, low corneal hysteresis (<9 mmHg).
Keratocyte Biology: The Maintenance Crew
Stromal keratocytes maintain extracellular matrix homeostasis and respond to injury with wound healing cascades:
-
Quiescent Keratocyte Characteristics
- Density: 300-400 cells/mm³ (highest in anterior stroma)
- Morphology: Dendritic cells with extensive cytoplasmic processes connecting to neighbors
- Metabolic state: Low activity, maintain existing matrix components
- Crystallin expression: Produce α-crystallins (ALDH3A1) contributing to transparency
- Matrix turnover: Secrete collagen, proteoglycans, matrix metalloproteinases (MMPs) at basal levels
- MMP-2 and MMP-9: Degrade damaged collagen for replacement
- TIMP-1 and TIMP-2: Inhibit excessive MMP activity
-
Wound Healing Transformation
- Injury triggers: Epithelial defect, UV exposure, inflammation, surgery
- Apoptosis phase (0-6 hours): Keratocytes immediately beneath injury undergo apoptosis
- Creates acellular zone 50-100 microns deep
- Prevents immediate scarring response
- Activation phase (6-24 hours): Surviving keratocytes activate and proliferate
- Cytokine stimulation: TGF-β, PDGF, FGF from tears and epithelium
- Transform to repair phenotype: Increased metabolic activity
- Fibroblast transformation (1-7 days): Activated keratocytes become fibroblasts
- Migrate to wound site
- Produce disorganized collagen (types I, III, V in abnormal ratios)
- Loss of crystallin expression → reduced transparency
- Myofibroblast differentiation (7-30 days): Subset of fibroblasts become myofibroblasts
- Express α-smooth muscle actin (contractile apparatus)
- Produce dense extracellular matrix with abnormal collagen organization
- Major source of corneal haze and scarring
- Stimulated by TGF-β1 (most potent profibrotic cytokine)
-
Scar Formation vs Regeneration
- Superficial wounds (<1/3 stromal depth): Often heal with minimal scarring
- Keratocytes may revert to quiescent phenotype after 3-6 months
- Deep wounds (>1/3 depth): Persistent myofibroblasts cause permanent scarring
- Collagen organization never returns to normal lamellar structure
- Scar tissue: Irregular fibril diameter, increased spacing, random orientation
- Therapeutic modulation:
- Corticosteroids: Reduce TGF-β expression, decrease myofibroblast formation
- Mitomycin-C: Induces apoptosis in activated keratocytes/fibroblasts
- Anti-TGF-β antibodies: Experimental, reduce scarring in animal models
- Superficial wounds (<1/3 stromal depth): Often heal with minimal scarring
⭐ Clinical Pearl: Corneal haze grading predicts visual outcomes-Grade 0 (clear), Grade 0.5 (trace haze visible with careful slit-lamp examination), Grade 1 (mild haze not interfering with iris details), Grade 2 (moderate haze with slightly blurred iris), Grade 3 (marked haze obscuring iris details), Grade 4 (completely opaque stroma). Haze ≥Grade 2 reduces best-corrected visual acuity by ≥2 lines and requires intervention.
| Stromal Property | Normal Value | Keratoconus | Post-CXL | Clinical Significance |
|---|---|---|---|---|
| Corneal Hysteresis | 10.7 ± 1.8 mmHg | 7.5-9.0 mmHg | 9.5-11.0 mmHg | Predicts progression risk, treatment response |
| Young's Modulus | 0.3-0.5 MPa | 0.15-0.25 MPa | 0.5-0.9 MPa | Biomechanical stiffness measure |
| Central Thickness | 540 ± 30 μm | 450-500 μm | 440-480 μm | Ectasia risk, surgical planning |
| Anterior Strength % | 60-70% | Reduced 40-50% | Increased 65-75% | LASIK safety, ectasia prevention |
| Keratocyte Density | 300-400 cells/mm³ | 200-250 cells/mm³ | 150-200 cells/mm³ | Matrix maintenance capacity |
Stromal biomechanics connect directly to pathophysiology (progressive biomechanical failure), interpretation (elevation maps detect early ectasia), and techniques (DALK preserves endothelium while replacing diseased stroma).
🌊 Stromal Biomechanics: The Structural Core
🔋 Endothelial Pump Physiology: The Dehydration Engine
The corneal endothelium maintains stromal transparency through active metabolic pumping, creating the osmotic gradient that keeps stromal hydration at 78% despite constant swelling pressure of 40-60 mmHg. This single-layer hexagonal mosaic of 2500-3000 cells/mm² in adults consumes more ATP per cell than any other corneal layer, running Na⁺-K⁺-ATPase pumps continuously to prevent edema. Endothelial cells never regenerate after birth-damage causes neighboring cells
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app


















