Reproductive Endocrinology

On this page
🧭 The Reproductive Endocrine Command Center: Your Strategic Overview
Reproductive endocrinology orchestrates fertility, metabolic health, and sexual development through an intricate hormonal network. Master the hypothalamic-pituitary-ovarian (HPO) axis, and you decode menstrual disorders, infertility, and systemic metabolic dysfunction. This lesson integrates 10 interconnected domains-from hirsutism patterns to ovulation induction protocols-building your clinical reasoning from molecular signals to bedside management. Understanding these pathways transforms isolated facts into diagnostic precision, enabling you to predict complications, select targeted therapies, and optimize reproductive outcomes across the lifespan.
🏗️ Hormonal Architecture: Building the HPO Foundation
The HPO axis functions as a three-tiered regulatory system where hypothalamic GnRH pulses drive pituitary gonadotropin release, which in turn orchestrates ovarian steroidogenesis and folliculogenesis. This architecture determines menstrual cyclicity, ovulation timing, and metabolic homeostasis.
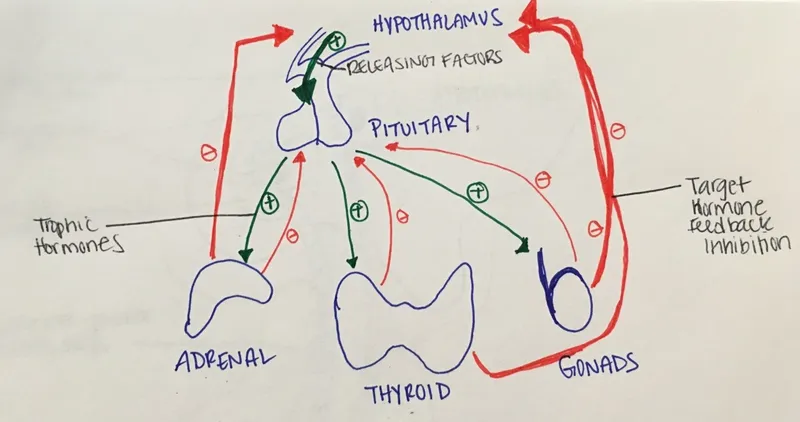
The Hierarchical Control System
-
Hypothalamic Command Center
- GnRH neurons in the arcuate nucleus fire in pulsatile patterns
- Pulse frequency determines FSH vs LH predominance: fast pulses (90 min) favor LH, slow pulses (>120 min) favor FSH
- Kisspeptin neurons integrate metabolic signals (leptin, insulin) with reproductive readiness
- Leptin threshold: <4 ng/mL suppresses GnRH pulsatility, causing hypothalamic amenorrhea
- Body fat: <22% disrupts menstrual cyclicity in 85% of athletes
- Stress-induced CRH and opioids suppress GnRH secretion within 2-4 hours
-
Pituitary Amplification Station
- Gonadotrophs respond to GnRH pulses by releasing FSH and LH
- FSH half-life: 3-4 hours (sustained follicular phase levels)
- LH half-life: 20-30 minutes (enables mid-cycle surge dynamics)
- Mid-cycle LH surge: >25 IU/L triggers ovulation within 34-36 hours
- Surge amplitude: 3-10 fold above baseline, lasting 48-72 hours
- Negative feedback from estradiol and inhibin B modulates FSH secretion
- Positive feedback: Estradiol >200 pg/mL for >48 hours triggers LH surge
-
Ovarian Response Theater
- Granulosa cells express FSH receptors, driving aromatase activity
- Theca cells possess LH receptors, producing androgens via two-cell, two-gonadotropin model
- Theca cells: Cholesterol → Androstenedione (requires LH)
- Granulosa cells: Androstenedione → Estradiol (requires FSH + aromatase)
- Dominant follicle selection occurs at 8-10 mm diameter (days 5-7)
- Corpus luteum produces 25-50 mg progesterone/day for 12-14 days
📌 Remember: GnRH Pulse Code - "Fast LH, Slow FSH" - Rapid GnRH pulses (<90 min) favor LH synthesis, while slower pulses (>120 min) favor FSH. This frequency coding determines which gonadotropin dominates, explaining why continuous GnRH (as in pump therapy or agonist downregulation) paradoxically suppresses both FSH and LH by desensitizing receptors.
⭐ Clinical Pearl: Functional hypothalamic amenorrhea affects 20-35% of reproductive-age women with menstrual irregularity. Weight restoration to BMI >18.5 kg/m² and stress reduction restore GnRH pulsatility in 60-70% within 3-6 months, avoiding unnecessary hormonal interventions.
Feedback Loop Sophistication
The HPO axis employs both negative and positive feedback mechanisms, creating dynamic regulation across the menstrual cycle.
| Feedback Type | Signal | Threshold | Target | Clinical Effect | Time Frame |
|---|---|---|---|---|---|
| Negative | Estradiol | <150 pg/mL | FSH/LH | Suppresses gonadotropins | Continuous |
| Positive | Estradiol | >200 pg/mL × 48h | LH | Triggers ovulatory surge | Mid-cycle |
| Negative | Progesterone | >3 ng/mL | GnRH frequency | Slows pulse, suppresses LH | Luteal phase |
| Negative | Inhibin B | Elevated | FSH | Dominant follicle selection | Follicular |
| Negative | Testosterone | >80 ng/dL | GnRH/LH | Suppresses axis (PCOS) | Chronic |
💡 Master This: The estradiol threshold paradox-low estradiol (<150 pg/mL) suppresses FSH/LH through negative feedback, but sustained high estradiol (>200 pg/mL for 48+ hours) switches to positive feedback, triggering the LH surge. This biphasic response explains why exogenous estrogen can both suppress ovulation (low-dose oral contraceptives) and induce ovulation (high-dose estradiol priming in IVF protocols). Recognizing this threshold transforms your understanding of hormonal contraception and ovulation induction strategies.
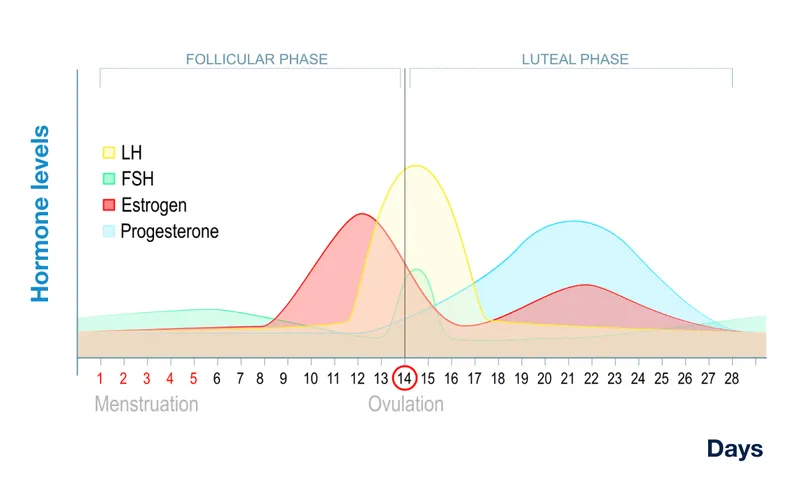
Metabolic Integration Points
-
Insulin's Reproductive Impact
- Hyperinsulinemia stimulates ovarian androgen production independent of LH
- Insulin resistance present in 50-70% of PCOS patients
- Compensatory hyperinsulinemia: >15 μIU/mL fasting correlates with anovulation
- Insulin directly stimulates theca cell androgen synthesis
- Suppresses hepatic SHBG production, increasing free testosterone by 30-50%
- Metformin improves ovulation rates by 25-40% in insulin-resistant PCOS
-
Thyroid-Reproductive Cross-Talk
- Hypothyroidism (TSH >4.5 mIU/L) causes anovulation in 15-20% of cases
- TRH stimulates prolactin release, creating hyperprolactinemic hypogonadism
- Thyroid hormone receptors on granulosa cells modulate FSH sensitivity
- Levothyroxine normalization restores ovulation in 60-75% within 3 months
⭐ Clinical Pearl: Check TSH in every patient with menstrual irregularity or infertility. Subclinical hypothyroidism (TSH 2.5-4.5 mIU/L with normal T4) reduces conception rates by 20-30% and increases miscarriage risk by 2-3 fold. Treatment to TSH <2.5 mIU/L before conception optimizes reproductive outcomes.
The HPO axis doesn't operate in isolation-it integrates metabolic signals, stress responses, and systemic health status. This integration explains why weight changes, thyroid dysfunction, and insulin resistance profoundly affect menstrual cyclicity and fertility. Connect these foundational principles through to understand how disruptions manifest as clinical disorders.
🏗️ Hormonal Architecture: Building the HPO Foundation
🔬 Androgen Excess: Decoding the Hyperandrogenic State
Hyperandrogenism represents the most common endocrine disorder in reproductive-age women, affecting 5-10% of the population. Understanding androgen physiology-from production sites to peripheral conversion-enables systematic diagnosis and targeted therapy.
Androgen Production Architecture
-
Ovarian Androgen Factory
- Theca cells produce 60-70% of circulating androgens
- Androstenedione: Primary ovarian androgen (1.5-3.0 ng/mL normal range)
- Testosterone: 25% from direct ovarian secretion, 25% from adrenal precursors, 50% from peripheral conversion
- Normal total testosterone: 15-70 ng/dL in women
- Free testosterone: <2.5% of total (most bound to SHBG)
- Ovarian stromal hyperthecosis produces severe hyperandrogenism (testosterone >150 ng/dL)
-
Adrenal Contribution
- DHEA-S: >90% adrenal origin (best marker of adrenal androgen excess)
- Normal DHEA-S: 35-430 μg/dL (age-dependent decline)
- Elevated DHEA-S (>700 μg/dL) suggests adrenal source
- Non-classic CAH (21-hydroxylase deficiency) in 1-9% of hyperandrogenic women
- 17-OHP: >200 ng/dL baseline or >1000 ng/dL post-ACTH stimulation confirms diagnosis
- Cushing's syndrome: Rare but must exclude when virilization present
-
Peripheral Conversion Dynamics
- 5α-reductase converts testosterone → DHT in skin
- Type 2 isoform: 3-5 fold more active in genital skin
- DHT: 2-3 times more potent at androgen receptor than testosterone
- Explains why some women have hirsutism despite normal testosterone
- Aromatase converts androgens → estrogens in adipose tissue
📌 Remember: Source Localization - "DHEA-S = Adrenal Address, Testosterone = Ovarian Origin" - DHEA-S >700 μg/dL points to adrenal pathology (CAH, tumor), while isolated testosterone elevation (>80 ng/dL) with normal DHEA-S indicates ovarian source (PCOS, tumor). Combined elevation (both >2× upper limit) raises concern for androgen-secreting tumor requiring imaging.
Clinical Manifestations: The Virilization Spectrum
-
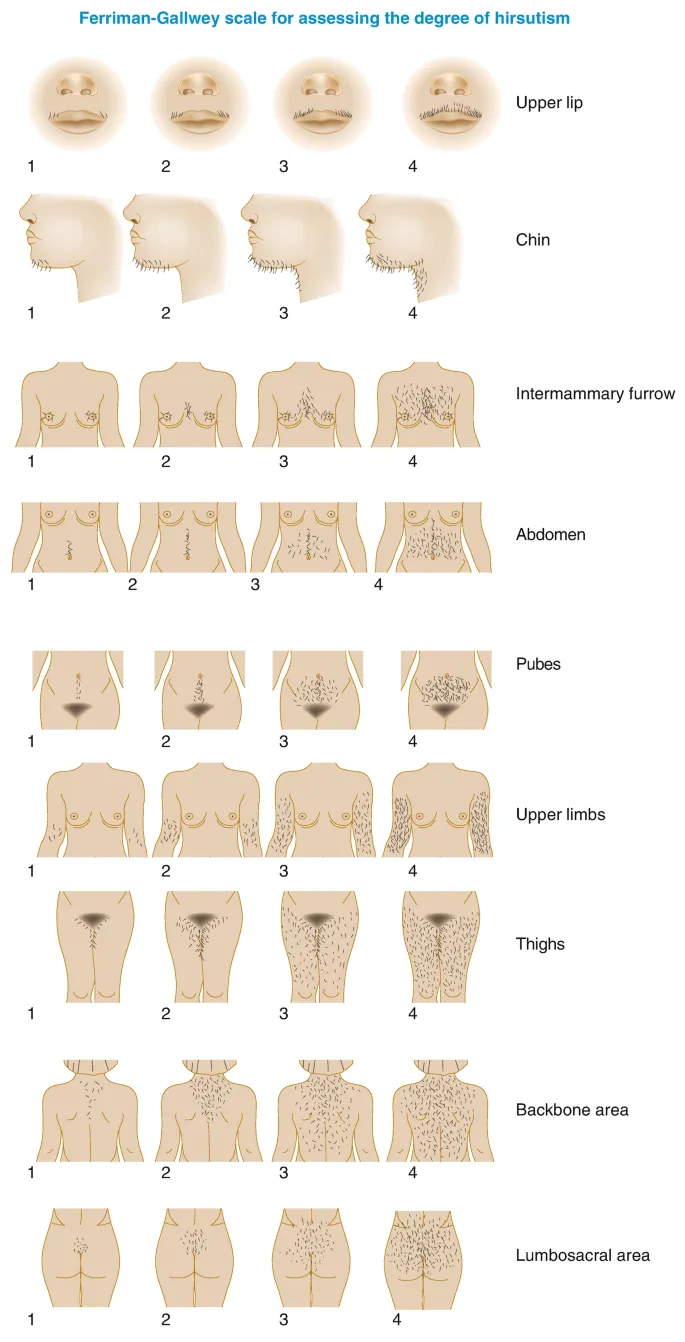
Hirsutism: Terminal Hair Distribution
- Ferriman-Gallwey score: 9 body areas scored 0-4 (maximum 36)
- Score ≥8 defines hirsutism in Caucasian/Mediterranean women
- Score ≥6 appropriate for Asian women (lower baseline hair growth)
- Upper lip, chin, chest, upper back: Most androgen-sensitive areas
- Affects 5-15% of reproductive-age women
- Rate of progression: Gradual onset (months-years) suggests PCOS; rapid (<6 months) suggests tumor
-
Virilization: Red Flag Features
- Clitoromegaly: Clitoral index >35 mm² (length × width) or length >10 mm
- Voice deepening: Irreversible laryngeal cartilage changes
- Temporal/vertex balding: Male-pattern hair loss
- Increased muscle mass and male body habitus
- Breast atrophy and loss of female body contours
- Virilization indicates testosterone >150-200 ng/dL (tumor threshold)
- Requires immediate pelvic ultrasound and adrenal CT imaging
⭐ Clinical Pearl: Rapid-onset hirsutism (<6 months) with virilization signs mandates tumor exclusion. Ovarian androgen-secreting tumors (Sertoli-Leydig, hilus cell) typically present with testosterone >150 ng/dL and unilateral ovarian mass >3 cm. Adrenal tumors show DHEA-S >700 μg/dL with adrenal mass on CT. Benign PCOS never causes true virilization-its presence changes your diagnostic algorithm entirely.
Diagnostic Algorithm: Systematic Workup
| Test | Normal Range | PCOS | Non-Classic CAH | Tumor Threshold | Interpretation |
|---|---|---|---|---|---|
| Total Testosterone | 15-70 ng/dL | 70-120 ng/dL | 60-150 ng/dL | >150 ng/dL | Ovarian source marker |
| DHEA-S | 35-430 μg/dL | Normal-mild ↑ | 430-700 μg/dL | >700 μg/dL | Adrenal source marker |
| 17-OHP (AM) | <200 ng/dL | Normal | 200-1000 ng/dL | >1000 ng/dL | CAH screening |
| LH:FSH ratio | 1:1 | >2:1 in 60% | Normal | Normal | PCOS supportive |
| Free Testosterone | 0.3-1.9 pg/mL | >1.9 pg/mL | Variable | >4.0 pg/mL | Bioavailable fraction |
| SHBG | 18-114 nmol/L | Low (↓ by insulin) | Normal | Variable | Affects free T calculation |
💡 Master This: The testosterone-DHEA-S matrix guides imaging decisions. If testosterone >150 ng/dL → pelvic ultrasound/MRI for ovarian tumor. If DHEA-S >700 μg/dL → adrenal CT for adrenal tumor. If 17-OHP >200 ng/dL → ACTH stimulation test for non-classic CAH. This systematic approach prevents missing androgen-secreting tumors while avoiding unnecessary imaging in benign PCOS. When both testosterone and DHEA-S exceed tumor thresholds, image both ovaries and adrenals simultaneously.
PCOS: The Hyperandrogenic Prototype
-
Rotterdam Criteria (2 of 3 Required)
- Oligo-ovulation or anovulation: <9 cycles/year or >35-day intervals
- Clinical or biochemical hyperandrogenism
- Polycystic ovarian morphology: ≥12 follicles (2-9 mm) per ovary or ovarian volume >10 mL
- Affects 6-12% of reproductive-age women globally
- Diagnosis requires exclusion of other causes (CAH, Cushing's, prolactinoma, thyroid)
-
Pathophysiologic Triad
- Insulin resistance: Present in 50-70%, independent of obesity
- LH hypersecretion: LH:FSH ratio >2:1 in 60% (not required for diagnosis)
- Chronic anovulation: Arrested follicular development at 5-8 mm
- Elevated LH stimulates theca cells → excess androgens
- Androgens inhibit aromatase → reduced estradiol
- Lack of dominant follicle → no LH surge → anovulation
-
Metabolic Consequences
- Metabolic syndrome: 30-40% prevalence (vs 15% in general population)
- Type 2 diabetes: 7.5-10% by age 40 (4-fold increased risk)
- Cardiovascular disease: 2-fold increased risk long-term
- Endometrial hyperplasia: 20-30% due to unopposed estrogen from anovulation
⭐ Clinical Pearl: PCOS patients with BMI >30 kg/m² have 2-3 fold higher testosterone and insulin levels compared to lean PCOS. Weight loss of just 5-10% body weight restores ovulation in 55-75% of anovulatory obese PCOS women within 6 months, often eliminating need for ovulation induction medications. Lifestyle modification remains first-line therapy before pharmacologic intervention.
Hyperandrogenism requires systematic evaluation to distinguish benign PCOS from life-threatening tumors. The diagnostic algorithm-testosterone and DHEA-S levels guiding imaging-prevents missed malignancies while avoiding overinvestigation. Connect androgen physiology through and metabolic implications through to build comprehensive understanding.
🔬 Androgen Excess: Decoding the Hyperandrogenic State
🎯 Pattern Recognition: The Clinical Endocrine Detective
Reproductive endocrine disorders present with overlapping symptoms-menstrual irregularity, infertility, hirsutism-requiring systematic pattern recognition to differentiate etiologies. Master the "see X, think Y" correlations, and you rapidly narrow differentials before ordering tests.
Menstrual Pattern Signatures
-
Primary Amenorrhea (No Menarche by Age 15)
- With breast development (estrogen present): Outflow tract obstruction or androgen insensitivity
- Imperforate hymen, transverse vaginal septum: Hematocolpos on exam
- Müllerian agenesis (MRKH): Normal external genitalia, absent uterus on ultrasound (1 in 4500 females)
- Androgen insensitivity: 46,XY karyotype, blind vaginal pouch, testes in inguinal canals
- Without breast development (estrogen deficient): Gonadal dysgenesis or hypothalamic-pituitary failure
- Turner syndrome (45,X): Short stature (<5th percentile), webbed neck, shield chest
- Kallmann syndrome: Anosmia + hypogonadotropic hypogonadism (1 in 10,000 females)
- Constitutional delay: Positive family history, bone age delayed >2 years
- With breast development (estrogen present): Outflow tract obstruction or androgen insensitivity
-
Secondary Amenorrhea (Absence >3 Months)
- Pregnancy: Always rule out first (β-hCG sensitivity >99% at 5 weeks)
- Hypothalamic: Low LH, low FSH, low estradiol + stress/weight loss/exercise
- BMI <18.5 kg/m² or >10% weight loss in 3-6 months
- Exercise >10 hours/week with negative energy balance
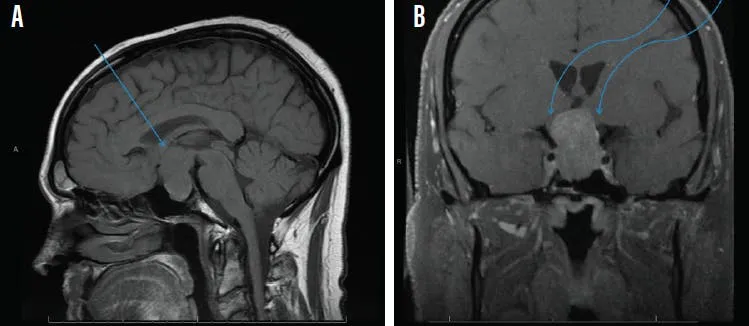
- Pituitary: Hyperprolactinemia (>25 ng/mL), Sheehan's syndrome, pituitary adenoma
- Ovarian: POI (FSH >25 IU/L on 2 occasions >4 weeks apart before age 40)
- Uterine: Asherman's syndrome (intrauterine adhesions post-D&C or endometritis)
- Endocrine: PCOS, thyroid dysfunction, Cushing's syndrome
📌 Remember: Amenorrhea Workup - "Pregnancy, Prolactin, Thyroid, Then Sort" - First check β-hCG (pregnancy), then TSH (thyroid), then prolactin. If all normal, check FSH to distinguish ovarian failure (high FSH) from hypothalamic-pituitary causes (low-normal FSH). This sequence prevents missing common treatable causes before pursuing rare diagnoses.
Galactorrhea-Amenorrhea Pattern
-
Hyperprolactinemia Thresholds
- Normal prolactin: <25 ng/mL (varies by lab, some use <30 ng/mL)
- Mild elevation (25-100 ng/mL): Medications, stress, hypothyroidism, PCOS
- Moderate elevation (100-200 ng/mL): Microadenoma (<10 mm) or drug effect
- Severe elevation (>200 ng/mL): Macroadenoma (>10 mm) in >90% of cases
- Prolactin >500 ng/mL: Virtually diagnostic of prolactinoma
- Hook effect: Falsely low prolactin with giant adenomas (request 1:100 dilution)
-
Common Medication Culprits
- Dopamine antagonists: Metoclopramide, domperidone (60-80% cause elevation)
- Antipsychotics: Risperidone, haloperidol (increase 3-10 fold)
- Antidepressants: SSRIs, tricyclics (mild elevation in 20-30%)
- Antihypertensives: Methyldopa, verapamil
- Estrogens: Oral contraceptives (physiologic elevation, rarely symptomatic)
⭐ Clinical Pearl: Visual field defects occur in 20-30% of macroadenomas due to suprasellar extension compressing optic chiasm. Bitemporal hemianopsia is classic but occurs in only 10-15%; superior temporal field defects are more common initially. Any prolactinoma >10 mm requires formal visual field testing before and during treatment. Dopamine agonist therapy shrinks tumors by >50% in 80-90% of cases within 3-6 months.
Anovulation Phenotypes
| Clinical Feature | PCOS | Hypothalamic | POI | Hyperprolactinemia | Thyroid |
|---|---|---|---|---|---|
| Menstrual Pattern | Oligomenorrhea | Amenorrhea | Amenorrhea | Oligo/Amenorrhea | Variable |
| FSH Level | Normal-low | Low-normal | Elevated (>25) | Normal-low | Normal |
| LH Level | Elevated | Low | Elevated | Normal-low | Normal |
| Estradiol | Normal-high | Low | Low | Normal-low | Normal |
| Androgen Status | Elevated | Normal | Normal | Normal | Normal |
| Body Habitus | Often obese | Often thin | Variable | Variable | Variable |
| Associated Signs | Hirsutism, acne | Stress, ↓ weight | Hot flashes | Galactorrhea | Fatigue, ↓ BMR |
💡 Master This: The FSH level distinguishes ovarian from central causes of anovulation. Elevated FSH (>25 IU/L) indicates ovarian failure (POI, menopause), while low-normal FSH suggests hypothalamic-pituitary dysfunction (stress, weight loss, hyperprolactinemia) or PCOS. This single test narrows your differential dramatically-high FSH means the ovaries can't respond (poor prognosis for ovulation induction), while low FSH means the signal is inadequate (often reversible with lifestyle changes or hormonal therapy).
Infertility Pattern Recognition
-
Time-to-Pregnancy Thresholds
- Infertility definition: 12 months of unprotected intercourse without conception
- Age >35 years: Evaluate after 6 months (declining ovarian reserve)
- Known risk factors: Evaluate immediately (amenorrhea, severe oligomenorrhea, male factor)
- Monthly pregnancy rate in fertile couples: 20-25% (peak age 25-30)
- Cumulative pregnancy rate: 85% by 12 months, 93% by 24 months
-
Ovulation Detection Methods
- Mid-luteal progesterone: >3 ng/mL confirms ovulation (day 21 of 28-day cycle)
- Basal body temperature: 0.5-1.0°F rise after ovulation (sensitivity 70-80%)
- LH surge detection kits: Predict ovulation 24-36 hours in advance (specificity >95%)
- Transvaginal ultrasound: Dominant follicle >18 mm + endometrial thickness >7 mm
-
Ovarian Reserve Markers
- AMH (Anti-Müllerian Hormone): Best single marker of ovarian reserve
- Normal: 1.0-4.0 ng/mL (age-dependent decline)
- Low reserve: <1.0 ng/mL (poor IVF response predicted)
- Very low: <0.5 ng/mL (consider donor eggs counseling)
- Day 3 FSH: >10 IU/L suggests diminished reserve, >20 IU/L indicates poor prognosis
- Antral follicle count (AFC): <5 follicles total indicates low reserve
- AMH (Anti-Müllerian Hormone): Best single marker of ovarian reserve
⭐ Clinical Pearl: AMH correlates with IVF oocyte yield better than FSH or age. AMH <1.0 ng/mL predicts poor response (<4 oocytes) in 70-80% of IVF cycles. Unlike FSH (which varies cycle-to-cycle), AMH remains stable and can be measured any day. Women with AMH <0.5 ng/mL have <5% pregnancy rate per IVF cycle, warranting early donor egg discussion. AMH doesn't predict egg quality-only quantity-so age remains the primary quality determinant.
Pattern recognition accelerates diagnosis by matching clinical presentations to hormonal profiles. When you see amenorrhea with galactorrhea, think prolactin first. When you encounter hirsutism with oligomenorrhea, PCOS tops your list. These patterns guide initial testing, preventing shotgun laboratory approaches. Connect diagnostic strategies through and specific disorders through .
🎯 Pattern Recognition: The Clinical Endocrine Detective
🏥 Therapeutic Algorithms: Evidence-Based Management Pathways
Reproductive endocrine management requires matching treatment intensity to diagnosis severity, patient goals, and metabolic context. Systematic algorithms prevent both undertreatment and unnecessary intervention.
Ovulation Induction: The Stepwise Approach
- First-Line: Lifestyle + Oral Agents
- Weight loss in obese PCOS: 5-10% reduction restores ovulation in 55-75%
- Clomiphene citrate: 50 mg days 3-7, increase by 50 mg to maximum 150 mg
- Ovulation rate: 70-80% of PCOS patients
- Pregnancy rate: 30-40% after 6 cycles
- Multiple pregnancy risk: 8-10% (mostly twins)
- Monitoring: Mid-cycle ultrasound optional, confirm ovulation with day 21 progesterone
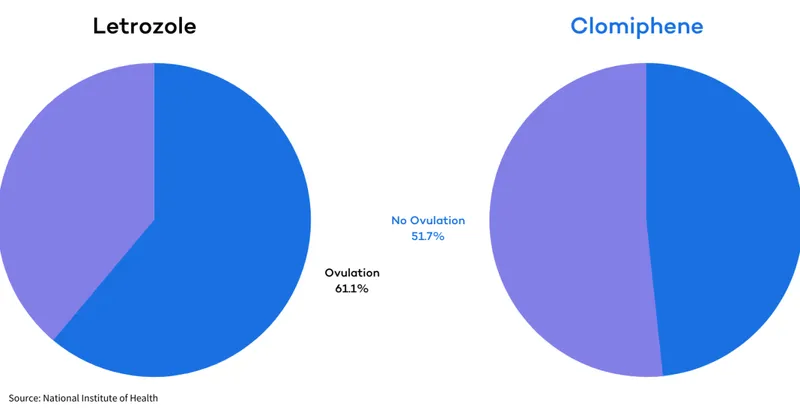
- Letrozole (aromatase inhibitor): 2.5 mg days 3-7, increase to 5-7.5 mg
- Superior to clomiphene in PCOS: Live birth 27.5% vs 19.1% per cycle
- Lower multiple pregnancy rate: 3-4% vs 8% with clomiphene
- Mechanism: Reduces estrogen → increased FSH → follicle recruitment
📌 Remember: Letrozole Advantages - "Letrozole: Less Multiples, More Singles" - Letrozole produces fewer follicles than clomiphene (typically 1-2 vs 2-4), reducing multiple pregnancy risk while maintaining higher pregnancy rates in PCOS. It's now preferred first-line for PCOS ovulation induction, with clomiphene reserved for letrozole failures or cost constraints.
-
Second-Line: Gonadotropin Therapy
- Low-dose step-up protocol: Start FSH 37.5-75 IU/day, increase by 37.5 IU every 7 days
- Goal: 1-2 mature follicles (≥18 mm) to minimize multiples
- Monitoring: Ultrasound + estradiol every 2-3 days during stimulation
- Cancel cycle if >3 follicles ≥14 mm or estradiol >2500 pg/mL (OHSS risk)
- hCG trigger: 5000-10,000 IU when lead follicle ≥18 mm
- Timed intercourse or IUI 36 hours post-trigger
- Ovulation rate: 70-90% in clomiphene-resistant PCOS
- Multiple pregnancy rate: 10-30% (requires careful monitoring)
-
Third-Line: Assisted Reproductive Technology
- IVF indicated for: Failed gonadotropin cycles (3-6 attempts), tubal factor, male factor
- Live birth rate per cycle: 30-40% (age <35), 10-15% (age >40)
- PCOS-specific protocols: GnRH antagonist + letrozole priming reduces OHSS risk
- Metformin pretreatment: Reduces OHSS by 50% in PCOS IVF cycles
⭐ Clinical Pearl: Ovarian hyperstimulation syndrome (OHSS) risk increases dramatically with >20 follicles or estradiol >3500 pg/mL during gonadotropin stimulation. Preventive strategies include coasting (withholding gonadotropins 1-3 days), GnRH agonist trigger instead of hCG (reduces severe OHSS by 80%), and cryopreservation of all embryos with delayed transfer. OHSS peaks 7-10 days post-trigger; severe cases (3-5%) require hospitalization for ascites, hemoconcentration, and thrombosis risk management.
Hyperandrogenism Management Matrix
| Therapy | Mechanism | Efficacy Timeline | Hirsutism Improvement | Metabolic Benefit | Contraceptive Effect |
|---|---|---|---|---|---|
| Combined OCP | ↓ LH, ↑ SHBG, ↓ adrenal androgens | 6-12 months | 60-70% reduction | Minimal | Yes |
| Spironolactone | Androgen receptor blocker | 6-9 months | 40-50% reduction | Minimal | No (teratogenic) |
| Metformin | ↓ Insulin, ↓ ovarian androgens | 3-6 months | 20-30% reduction | ↓ Insulin resistance | No |
| Eflornithine cream | Inhibits hair follicle ornithine decarboxylase | 2-3 months | 30-40% facial hair | None | No |
| Finasteride | 5α-reductase inhibitor | 6-12 months | 30-50% reduction | Minimal | No (teratogenic) |
- Combination Therapy Rationale
- OCP + Spironolactone: Synergistic effect (70-80% improvement vs 50-60% monotherapy)
- Metformin + Lifestyle: Additive metabolic benefits in insulin-resistant PCOS
- Mechanical hair removal + Medical therapy: Fastest cosmetic improvement
- Laser hair removal: 70-90% reduction after 6-8 sessions (works best on dark hair)
- Electrolysis: Permanent but time-intensive, suitable for small areas
💡 Master This: Hirsutism treatment requires 6-12 months to show significant improvement because hair growth cycles last 6-8 weeks. Set realistic expectations-medical therapy slows new growth and thins existing hair but doesn't remove established terminal hairs. Combining OCP (suppresses new androgen production) with spironolactone (blocks existing androgen effects) plus mechanical removal (addresses current hair) provides comprehensive management. Patients often abandon therapy prematurely at 3 months when improvement isn't yet visible; counseling about the timeline prevents discontinuation.
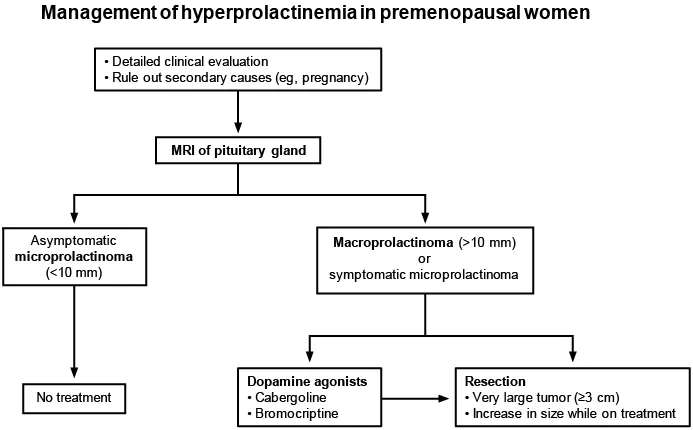
Hyperprolactinemia Treatment Protocol
-
Dopamine Agonist Therapy
- Cabergoline: 0.25 mg twice weekly, increase by 0.25 mg every 4 weeks to maximum 1.5 mg twice weekly
- Preferred over bromocriptine: Better tolerability (nausea 10% vs 50%)
- Prolactin normalization: 85-95% of microadenomas, 70-80% of macroadenomas
- Tumor shrinkage: >50% reduction in 80-90% within 3-6 months
- Menstrual cycle restoration: 75-90% within 3 months of normalization
- Bromocriptine: 1.25 mg daily, increase to 2.5 mg twice daily
- Start at bedtime with food to minimize nausea
- Pregnancy-preferred agent (longer safety data than cabergoline)
- Cabergoline: 0.25 mg twice weekly, increase by 0.25 mg every 4 weeks to maximum 1.5 mg twice weekly
-
Monitoring Strategy
- Prolactin levels: Check 4 weeks after dose adjustment, then every 3 months until stable
- MRI: Repeat at 1 year for macroadenomas, optional for microadenomas
- Visual fields: Test every 6 months if macroadenoma with visual symptoms
- Dose reduction trial: After 2 years of normal prolactin and tumor shrinkage
- 30-40% remain normoprolactinemic off medication
- Higher success in microadenomas and pregnancy-related prolactinomas
⭐ Clinical Pearl: Pregnancy in prolactinoma patients requires individualized management. Microadenomas have <5% risk of significant growth during pregnancy-most clinicians discontinue dopamine agonists once pregnancy confirmed. Macroadenomas carry 15-35% risk of symptomatic enlargement; consider continuing cabergoline or switching to bromocriptine. Monitor symptoms (headache, visual changes) rather than prolactin levels during pregnancy. Breastfeeding is safe and doesn't worsen tumor growth in most cases.
Primary Ovarian Insufficiency Management
-
Hormone Replacement Therapy (Mandatory)
- Physiologic estrogen replacement: Prevents osteoporosis, cardiovascular disease, cognitive decline
- Transdermal estradiol: 100 μg patch twice weekly (avoids first-pass hepatic metabolism)
- Micronized progesterone: 200 mg nightly (for endometrial protection)
- Continue until age 50-51 (natural menopause age)
- Higher doses than typical menopause HRT (replaces premenopausal levels)
- Bone density: Baseline DEXA, repeat every 2-5 years
- Cardiovascular screening: Lipid panel, blood pressure monitoring annually
-
Fertility Considerations
- Spontaneous pregnancy: 5-10% of POI patients conceive naturally (intermittent ovarian function)
- Ovulation induction: Generally ineffective (FSH already maximally elevated)
- Donor egg IVF: 50-60% live birth rate per cycle (similar to age-matched donors)
- Fertility preservation: Consider oocyte cryopreservation in women with transitional POI (FSH 25-40 IU/L with intermittent menses)
💡 Master This: POI before age 40 increases long-term health risks: Osteoporosis (2-fold risk of fractures), cardiovascular disease (2-fold risk of MI/stroke), cognitive decline, and premature mortality (hazard ratio 1.7-2.0). Hormone replacement therapy until natural menopause age mitigates these risks-it's not optional cosmetic treatment but essential preventive medicine. Distinguish this from menopausal HRT (which has risks in older women); physiologic replacement in young POI patients restores normal hormonal milieu without excess risk.
Evidence-based algorithms prevent both therapeutic nihilism and overtreatment. Start with lifestyle and oral agents, escalate to gonadotropins only after failures, and reserve ART for specific indications. Each step has defined success rates and timeframes, enabling shared decision-making with patients about when to advance therapy. Connect treatment strategies through and specific disorder management through .
🏥 Therapeutic Algorithms: Evidence-Based Management Pathways
⚖️ Metabolic-Reproductive Integration: The PCOS Paradigm
PCOS exemplifies the intricate relationship between metabolic dysfunction and reproductive pathology. Understanding this integration reveals why reproductive endocrinologists must think beyond ovaries, addressing insulin resistance, cardiovascular risk, and long-term metabolic health.
Insulin Resistance: The Metabolic Driver
- Pathophysiologic Mechanisms
- Insulin receptor dysfunction: Post-receptor signaling defects in 50-70% of PCOS patients
- Compensatory hyperinsulin
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app










