Hemoglobin and Myoglobin - Heme Team Intro
-
Myoglobin (Mb):
- Structure: Monomeric protein in muscle; $O_2$ storage.
- Components: Single polypeptide chain, one heme group (Protoporphyrin IX + $Fe^{2+}$).
- Key Histidines:
- Proximal (F8): Directly binds $Fe^{2+}$.
- Distal (E7): Stabilizes $O_2$-$Fe^{2+}$ bond, prevents $Fe^{2+}$ oxidation to $Fe^{3+}$.

-
Hemoglobin (Hb):
- Structure: Tetrameric protein (e.g., $\alpha_2\beta_2$ in HbA) in RBCs; $O_2$ transport.
- Components: Four polypeptide chains, each with a heme group (Protoporphyrin IX + $Fe^{2+}$).

-
Hemoglobin Types & Chains:
- HbA (Adult): $\alpha_2\beta_2$; >95%.
- HbA2 (Adult): $\alpha_2\delta_2$; 2-3.5%.
- HbF (Fetal): $\alpha_2\gamma_2$; <2% in adults, predominant in fetus.
⭐ Heme, composed of Protoporphyrin IX and a central ferrous ($Fe^{2+}$) ion, is the crucial site for reversible $O_2$ binding.
Oxygen Binding Dynamics - O2 Delivery Service
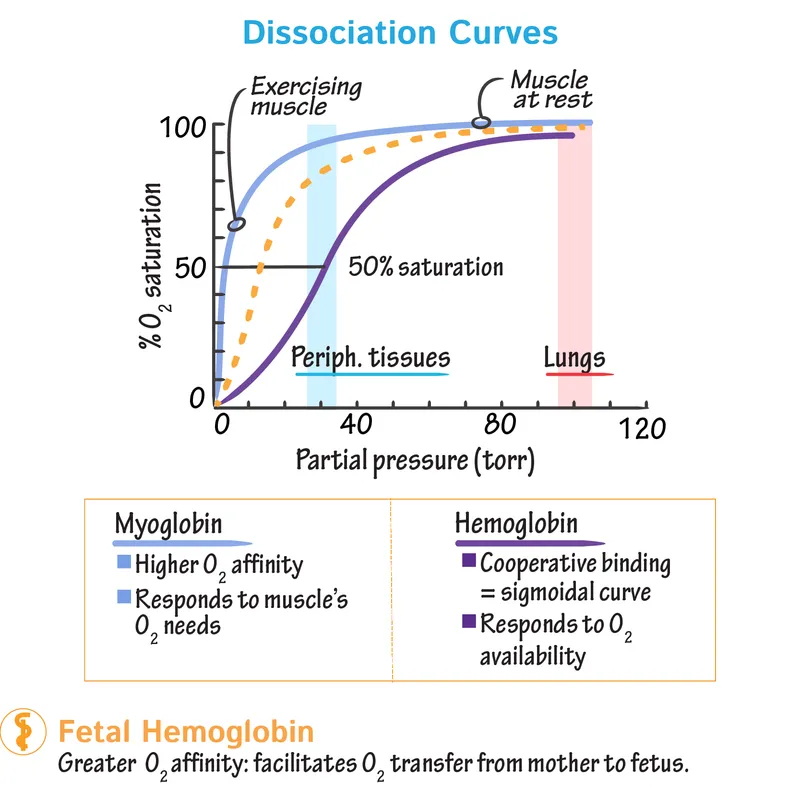
- Myoglobin (Mb):
- Monomer, 1 $O_2$ site. Hyperbolic curve.
- High $O_2$ affinity (P50 Mb ≈ 1 mmHg). $O_2$ storage (muscle).
- Hemoglobin (Hb):
- Tetramer ($Hb + 4O_2 \rightleftharpoons Hb(O_2)_4$), 4 $O_2$ sites. Sigmoidal curve (cooperative binding).
- T (taut) state: Low $O_2$ affinity.
- R (relaxed) state: High $O_2$ affinity.
- Lower $O_2$ affinity (P50 Hb ≈ 26.6 mmHg). $O_2$ transport (blood).
-
Factors Shifting ODC Right (↓ Affinity, ↑ $O_2$ Release):
- 📌 CADET, right shift!: ↑ CO₂, ↑ Acid (↓pH), ↑ 2,3-DPG, ↑ Exercise, ↑ Temperature.
-
Bohr Effect: ↓pH or ↑$CO_2$ ($H^+$ binds Hb) → stabilizes T-state → ↓$O_2$ affinity (right shift) → $O_2$ release in tissues.
-
Haldane Effect: DeoxyHb ↑ affinity for $CO_2$/$H^+$ vs OxyHb. Promotes $CO_2$ transport from tissues; $O_2$ binding in lungs releases $H^+$, $CO_2$.
⭐ HbF ($\alpha_2\gamma_2$) has ↓ affinity for 2,3-BPG vs HbA ($\alpha_2\beta_2$) → ↑ $O_2$ affinity, aiding $O_2$ transfer mother-fetus.
Hemoglobin Variants & Issues - When Heme Goes Haywire
- Sickle Cell Anemia (HbS):
- Cause: β-globin point mutation (Glu6Val).
- Patho: HbS polymerizes in low $O_2$ → RBC sickling, vaso-occlusion, hemolysis.

⭐ In Sickle Cell Anemia, glutamic acid is replaced by valine at the 6th position of the β-globin chain.
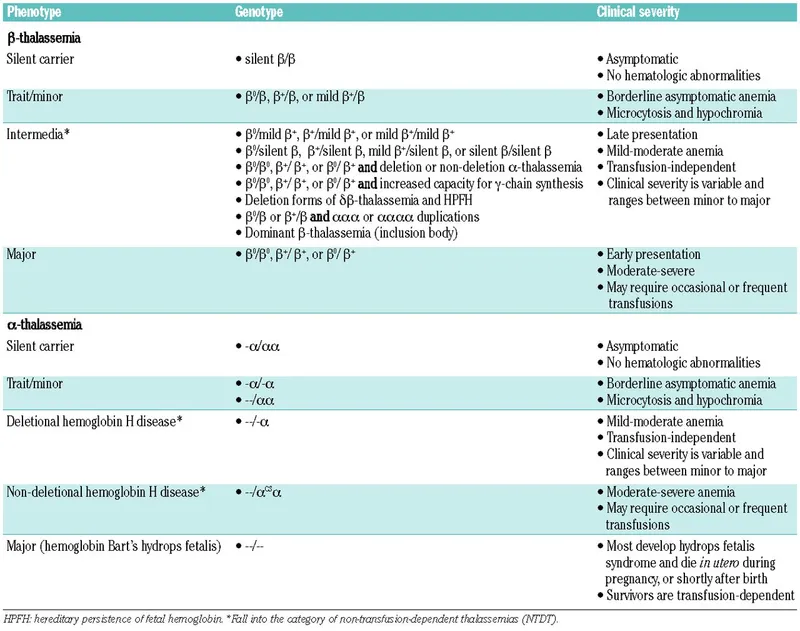
- Thalassemias:
- Defect: Reduced α or β globin chain synthesis → chain imbalance.
- α-Thalassemia: ↓ α-chain. Severity depends on gene deletions.
- β-Thalassemia: ↓ β-chain. Major (Cooley's) = severe anemia, ineffective erythropoiesis.
- Methemoglobinemia (MetHb):
- Issue: Heme iron oxidized to $Fe^{3+}$; cannot bind $O_2$.
- Causes: Oxidant drugs (nitrites, dapsone), congenital.
- Signs: Cyanosis, "chocolate blood".
- Rx: Methylene blue. 📌 MetHb: Methylene Blue.
- Carboxyhemoglobinemia (COHb):
- Cause: $CO$ poisoning. $CO$ has ~200-250x greater affinity for Hb than $O_2$.
- Effect: Shifts ODC left (↓ $O_2$ delivery).
- Signs: Headache, confusion; classic "cherry-red" skin/mucosa.
High‑Yield Points - ⚡ Biggest Takeaways
- Myoglobin (Mb): Monomeric, higher O₂ affinity, hyperbolic curve; O₂ storage in muscle.
- Hemoglobin (Hb): Tetrameric (α₂β₂), sigmoidal O₂ binding (cooperativity); O₂ transport in blood.
- T (taut) state has low O₂ affinity; R (relaxed) state has high O₂ affinity.
- 2,3-BPG stabilizes T state, promoting O₂ release (right shift).
- Bohr effect: ↓pH or ↑CO₂ shifts curve right (↓O₂ affinity).
- Haldane effect: Deoxygenated Hb binds CO₂/H⁺ better.
- HbF (α₂γ₂) has ↑O₂ affinity than HbA (poor 2,3-BPG binding).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

