Nucleic Acid Vaccines - Genetic Code Shots
- Delivers genetic material (DNA or mRNA) encoding specific antigen(s) into host cells.
- Host cells transcribe (DNA) and translate (mRNA/DNA) the genetic material to produce the antigen.
- This in-situ antigen production mimics natural infection, eliciting robust cellular (T-cell) and humoral (B-cell) immunity.
- Types:
- DNA Vaccines:
- Plasmid DNA containing antigen gene.
- Requires entry into nucleus for transcription.
- Good stability; elicits strong Th1 responses.
- mRNA Vaccines:
- In vitro transcribed mRNA encoding antigen.
- Direct translation in cytoplasm; no nuclear entry needed.
- Rapid development, potent immunogenicity (e.g., COVID-19 vaccines).
- Often encapsulated in Lipid Nanoparticles (LNPs) for delivery & stability.
- DNA Vaccines:
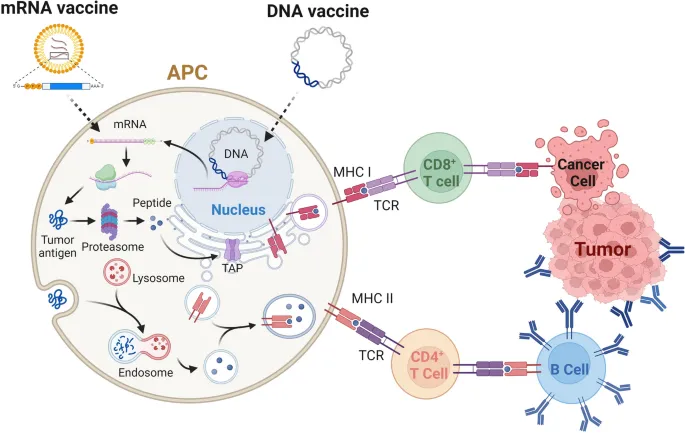
⭐ mRNA vaccines (e.g., Pfizer-BioNTech, Moderna for COVID-19) are known for their rapid development and ability to elicit strong neutralizing antibody responses and T-cell immunity. They bypass the need for nuclear entry, leading to faster antigen expression compared to DNA vaccines.
- Advantages: Rapid development, scalability, safety (non-infectious, no integration risk for mRNA), induces broad immunity.
- Challenges: Delivery systems (especially for DNA), stability (mRNA more labile than DNA), potential for anti-DNA antibodies (theoretical for DNA vaccines).
Viral Vector Vaccines - Trojan Horse Tactics
- Harmless virus (vector) delivers pathogen's genetic material (DNA/RNA) into host cells. 📌 Viral Vans Deliver Genes.
- Host cells produce pathogen antigens, triggering robust immune response (cellular & humoral).
- "Trojan Horse" delivery of antigen-coding genes.
- Vector Types:
- Replicating: e.g., Measles, VSV. ↑Antigen, stronger immunity.
- Non-replicating: e.g., Adenovirus (Ad26, ChAdOx1), AAV. Safer; may need boosters.
- Pros: Strong T & B-cell response; good stability.
- Cons: Pre-existing vector immunity can ↓ efficacy; manufacturing complexity.
- Examples:
- COVID-19: J&J (Ad26), AstraZeneca (ChAdOx1).
- Ebola: rVSV-ZEBOV.

⭐ Pre-existing adenovirus immunity can ↓ vector vaccine efficacy.
Subunit & Particle Vaccines - Protein Power-Ups
- Uses purified antigens (proteins, polysaccharides) or Virus-Like Particles (VLPs). No whole organisms.
- Pros: ↑ Safety (no infection risk), defined composition.
- Cons: Often need adjuvants; immunity may be less broad/durable.
- Examples:
- Protein Subunit: Hepatitis B (HBsAg), Acellular pertussis.
- Polysaccharide: Pneumococcal (PPSV23), Meningococcal. Conjugation (e.g., Hib, PCV13) vital for infant T-dependent response.
- Virus-Like Particles (VLPs): HPV, Hepatitis B. Self-assembling viral proteins; mimic virus, highly immunogenic, non-infectious.
⭐ HPV vaccine, a VLP type, shows high efficacy by mimicking viral structure, inducing robust protective immunity against specific oncogenic HPV types.
Emerging Vaccine Tech & Adjuvants - Frontier Technologies
- Nanoparticle Vaccines: VLPs, liposomes. ↑ immunogenicity.
- Virus-Like Particles (VLPs): Non-infectious, mimic native pathogen structures (e.g., HPV, HBV).
- Outer Membrane Vesicles (OMVs): From Gram-negative bacteria, potent immunogens (e.g., MenB).
- Reverse Vaccinology 2.0: Genomic/proteomic antigen discovery.
- Structural Vaccinology: Structure-based rational antigen design.
- Self-amplifying RNA (saRNA): Lower dose, prolonged antigen expression than mRNA.
- Modern Adjuvants: Enhance magnitude & duration of immune response.
- TLR Agonists: CpG ODN (TLR9), MPLA (TLR4).
- Saponins: QS-21 (potent saponin from Quillaja saponaria).
- Emulsions: MF59, AS03 (oil-in-water).
- STING Agonists: Potent innate immune activation via STING.

⭐ AS04 adjuvant combines MPLA (TLR4 agonist) and alum, used in HPV (Cervarix) and HBV (Fendrix) vaccines.
High‑Yield Points - ⚡ Biggest Takeaways
- mRNA vaccines (e.g., Pfizer, Moderna) use mRNA for antigen synthesis; rapid development, no infection risk.
- Viral vector vaccines (e.g., Covishield) use a modified virus to deliver antigen genes.
- DNA vaccines (e.g., ZyCoV-D) use plasmid DNA; stable, elicit cellular and humoral immunity.
- Recombinant protein vaccines (e.g., Novavax) use purified antigens; fewer side effects.
- Virus-Like Particles (VLPs) (e.g., HPV) are non-infectious, highly immunogenic structural mimics.
- Modern adjuvants enhance immunogenicity and response duration.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

