Infectious Diseases

On this page
🦟 Tropical Parasites: The Geographic Disease Architects
Tropical infections dominate global disease burden, with >1 billion people infected by soil-transmitted helminths and >200 million malaria cases annually. Understanding geographic distribution patterns, vector biology, and life cycle vulnerabilities transforms abstract parasitology into clinical diagnostic frameworks.
Malaria: The Plasmodium Powerhouse
Five Plasmodium species cause human malaria, but P. falciparum drives >90% of mortality through cytoadherence and microvascular sequestration. The erythrocytic cycle repeats every 48 hours (tertian pattern) for P. falciparum, P. vivax, and P. ovale; 72 hours (quartan) for P. malariae; and 24 hours (quotidian) for P. knowlesi-the emerging zoonotic species with 10% mortality untreated.
📌 Remember: FEVER mnemonic for severe malaria criteria-Failure (renal: creatinine >3 mg/dL), Encephalopathy (Glasgow <11), Ventilation (ARDS), Extreme anemia (Hb <5 g/dL), Retinal hemorrhages (blackwater fever: hemoglobinuria from massive intravascular hemolysis)
- Pathogenesis cascade
- Sporozoites (mosquito saliva) → hepatocytes → 10-14 days incubation
- Merozoites released → invade RBCs → trophozoites ("ring forms") → schizonts
- P. vivax and P. ovale form dormant hypnozoites → relapses up to 5 years later
- Requires primaquine (0.5 mg/kg daily × 14 days) after G6PD screening
- G6PD deficiency prevalence: 10-15% in malaria-endemic zones
- P. falciparum cytoadherence via PfEMP1 → rosetting → cerebral malaria (15% mortality)
⭐ Clinical Pearl: Cerebral malaria presents with symmetric hyperreflexia and extensor posturing, distinguishing it from bacterial meningitis (asymmetric focal signs). Retinal hemorrhages (Roth spots with white centers) appear in 15% and predict 2.5× mortality risk.
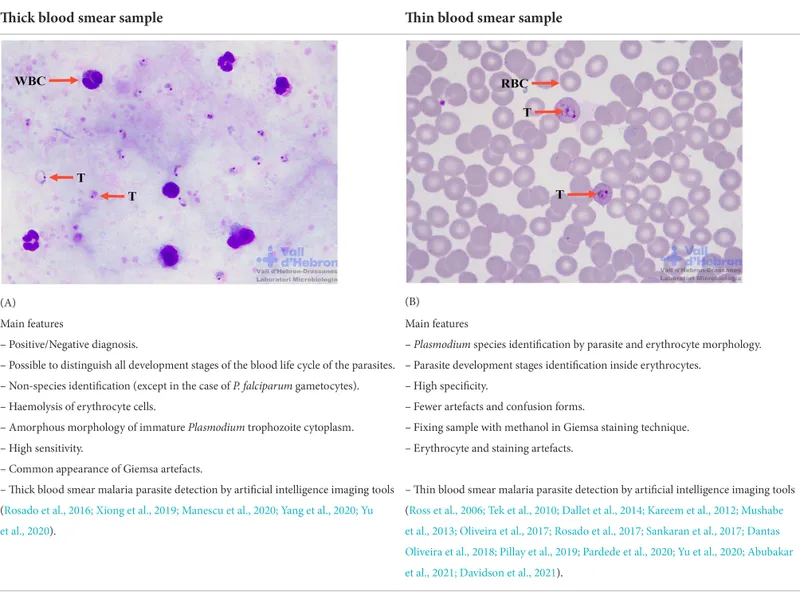
- Diagnostic precision
- Thick smear: 10-20× sensitivity vs thin smear, detects 50-100 parasites/μL
- Thin smear: species identification, quantifies parasitemia (severe if >5%)
- Rapid diagnostic tests (RDTs): HRP2 antigen (95% sensitivity for P. falciparum)
- False negatives: HRP2 gene deletions (5-10% in South America/Horn of Africa)
- PCR gold standard: detects <1 parasite/μL, identifies mixed infections (10-15% prevalence)
💡 Master This: Severe malaria requires IV artesunate (2.4 mg/kg at 0, 12, 24h, then daily) over quinine-35% mortality reduction in African children, 23% reduction in Asian adults. Artesunate causes delayed hemolysis in 10-15% at day 7-14; monitor Hb weekly × 4 weeks.
Helminthic Hierarchies: Worm Warfare
Helminths infect >2 billion globally, causing chronic morbidity through immune modulation and mechanical obstruction. Soil-transmitted helminths (Ascaris, hookworm, Trichuris) peak in school-age children (40-50% prevalence); tissue-invasive nematodes (Strongyloides, filariae) cause life-threatening hyperinfection in immunocompromised hosts.
| Helminth | Vector/Route | Key Pathology | Diagnostic Test | Treatment | Resistance Risk |
|---|---|---|---|---|---|
| Ascaris lumbricoides | Fecal-oral (eggs) | Loeffler syndrome (10% eosinophilia), intestinal obstruction | Stool O&P (fertilized eggs) | Albendazole 400 mg × 1 | Low (<5%) |
| Hookworm (Necator, Ancylostoma) | Skin penetration (larvae) | Iron-deficiency anemia (0.2 mL blood/worm/day) | Stool O&P (thin-shelled eggs) | Albendazole 400 mg × 3d | Moderate (10-15%) |
| Strongyloides stercoralis | Skin penetration (filariform larvae) | Autoinfection → hyperinfection (50-85% mortality) | Serology (ELISA), stool PCR | Ivermectin 200 μg/kg × 2d | Low (<2%) |
| Wuchereria bancrofti | Mosquito (microfilariae) | Lymphatic filariasis → elephantiasis (after 10-15 years) | Nocturnal blood smear, CFA card | DEC 6 mg/kg × 12d + albendazole | Low (<5%) |
| Schistosoma spp. | Freshwater snails (cercariae) | Katayama fever, portal hypertension (15-20%) | Stool/urine O&P (terminal spine eggs), serology | Praziquantel 40 mg/kg × 1 | Emerging (5-10%) |
📌 Remember: SWIM for schistosomiasis species-S*. mansoni* (colon, lateral spine), W*. haematobium* (bladder, terminal spine), Intercalatum (colon, terminal spine), M*. japonicum* (colon, no spine, 50-100 eggs/miracidium vs 1-10 for others)

- Strongyloides hyperinfection syndrome
- Triggers: corticosteroids (most common), HTLV-1 coinfection (75% seroprevalence in endemic areas), hematologic malignancy
- Pathophysiology: accelerated autoinfection → 10⁶-10⁹ larvae/day vs 10²-10³ in chronic infection
- Larvae penetrate colonic mucosa → bacteremia with enteric flora (E. coli, Klebsiella)
- Disseminated strongyloidiasis: larvae in sputum, CSF, peritoneum
- Clinical triad: abdominal pain + eosinophilia (often absent in severe cases) + gram-negative sepsis
- Mortality: 50-85% despite treatment; prevention via ivermectin screening before immunosuppression
⭐ Clinical Pearl: Eosinophilia <500/μL in suspected strongyloidiasis suggests hyperinfection (consumptive eosinopenia) or HTLV-1 coinfection. Obtain stool PCR (sensitivity >90%) and serology (ELISA sensitivity 85-95%); negative tests don't exclude disease-treat empirically if high pretest probability.
Protozoal Precision: Beyond Malaria
Non-malarial protozoa cause >50 million infections annually, with Entamoeba histolytica and Giardia dominating diarrheal disease, while Leishmania and Trypanosoma produce visceral and CNS syndromes with >10% mortality untreated.
- Visceral leishmaniasis (kala-azar)
- Vector: Phlebotomus sandfly; reservoir: dogs (zoonotic), humans (anthroponotic L. donovani)
- Incubation: 2-6 months (range 10 days-2 years)
- Classic pentad: fever + hepatosplenomegaly + pancytopenia + hypergammaglobulinemia + darkening skin
- Splenomegaly massive (15-20 cm below costal margin)
- Pancytopenia: WBC <4000/μL, Hb <10 g/dL, platelets <100,000/μL
- Polyclonal IgG: >5 g/dL (inverted albumin:globulin ratio)
- Diagnosis: rK39 RDT (sensitivity >95% in South Asia, <70% in East Africa), splenic aspirate (sensitivity >95%, risk <0.1%)
- Treatment: liposomal amphotericin B 10 mg/kg single dose (cure >95%) or 3 mg/kg daily × 5 days
💡 Master This: Post-kala-azar dermal leishmaniasis (PKDL) develops in 5-10% after treatment, presenting 6 months-3 years later with hypopigmented macules progressing to nodules. PKDL patients serve as human reservoirs sustaining transmission; treatment requires extended courses (miltefosine 2.5 mg/kg/day × 12 weeks).

🦟 Tropical Parasites: The Geographic Disease Architects
💊 Antimicrobial Mastery: The Pharmacodynamic Framework
Antimicrobial efficacy depends on achieving target drug concentrations at infection sites while minimizing toxicity and resistance selection. Understanding pharmacokinetic/pharmacodynamic (PK/PD) indices-time above MIC (T>MIC), peak:MIC ratio (Cmax:MIC), and area under curve:MIC (AUC:MIC)-transforms empiric prescribing into precision therapeutics.
PK/PD Indices: The Dosing Determinants
Different antibiotic classes require distinct PK/PD optimization strategies. β-lactams exhibit time-dependent killing, requiring prolonged T>MIC; aminoglycosides show concentration-dependent killing with extended post-antibiotic effect (PAE); fluoroquinolones demonstrate AUC-dependent efficacy balancing both mechanisms.
📌 Remember: CAPITAL for concentration-dependent drugs-Colistin, Aminoglycosides, Polymyxins, Isoniazid (early bactericidal), Tigecycline, Amphotericin, Lipopeptides (daptomycin). All others primarily time-dependent (β-lactams, glycopeptides, oxazolidinones).
- β-lactam optimization strategies
- Target T>MIC: 40-50% for bacteriostasis, 60-70% for maximal killing
- Standard dosing achieves target for MIC ≤1 μg/mL; resistant organisms (MIC 4-8 μg/mL) require intensification
- Extended infusion: piperacillin-tazobactam 4.5 g over 4h q8h vs 4.5 g over 30min q6h
- Continuous infusion: meropenem 6 g/day continuous after 2 g loading (maintains >16 μg/mL)
- Critical illness pharmacokinetics: ↑ volume of distribution (+30-50%), augmented renal clearance (CrCl >130 mL/min in 30-65%)
- Requires higher doses: meropenem 2 g q8h vs standard 1 g q8h
- Therapeutic drug monitoring (TDM): target trough >4× MIC (free drug concentration)
⭐ Clinical Pearl: Augmented renal clearance (ARC) in young trauma/burn patients causes subtherapeutic β-lactam levels despite "normal" dosing. Suspect when CrCl >130 mL/min by 8-hour urine collection; measure trough concentrations and escalate doses empirically (+50-100%) while awaiting results.
- Aminoglycoside precision dosing
- Target Cmax:MIC >8-10 for maximal killing and PAE (>6 hours against gram-negatives)
- Once-daily dosing: gentamicin 5-7 mg/kg achieves Cmax 20-30 μg/mL (vs 4-6 μg/mL with divided dosing)
- Equivalent efficacy, ↓30-50% nephrotoxicity vs traditional q8h dosing
- Contraindications: endocarditis (requires sustained levels), pregnancy, CrCl <30 mL/min
- Extended-interval nomogram: dose based on CrCl, monitor levels at 6-14 hours post-dose
- Target random level <5 μg/mL at 12h (low nephrotoxicity risk)
- Synergy with β-lactams: gentamicin 3 mg/kg/day divided for enterococcal endocarditis (time-dependent synergy)
| Drug Class | PK/PD Index | Target Value | Dosing Strategy | TDM Parameter | Target Level |
|---|---|---|---|---|---|
| Penicillins | T>MIC | 60-70% interval | Extended/continuous infusion | Trough (free) | 4-6× MIC |
| Carbapenems | T>MIC | 40-50% interval | Extended infusion (4h) | Trough (free) | 4-5× MIC |
| Vancomycin | AUC:MIC | 400-600 | Loading 25-30 mg/kg, then q8-12h | Trough, AUC | 15-20 μg/mL, AUC 400-600 |
| Aminoglycosides | Cmax:MIC | 8-10 | Once daily 5-7 mg/kg | Peak, random level | 20-30 μg/mL, <5 at 12h |
| Fluoroquinolones | AUC:MIC | 125-250 | High-dose once daily | AUC (rarely measured) | AUC >100-125 |
| Daptomycin | AUC:MIC | ≥666 | 8-10 mg/kg/day (high-dose) | Trough | ≥24.3 μg/mL |
💡 Master This: Vancomycin dosing shifted from trough-based (15-20 μg/mL) to AUC-based (400-600) after 2020 consensus guidelines. Bayesian calculators estimate AUC from two levels (peak and trough); AUC >600 increases nephrotoxicity risk 2.5× without added efficacy. Target lower AUC (400-500) for non-bacteremic infections.
Antimicrobial Spectrum: Class Characteristics
Antibiotic selection requires matching spectrum to likely pathogens while considering tissue penetration, resistance patterns, and toxicity profiles. Empiric therapy balances broad coverage against collateral damage (resistance selection, C. difficile risk).
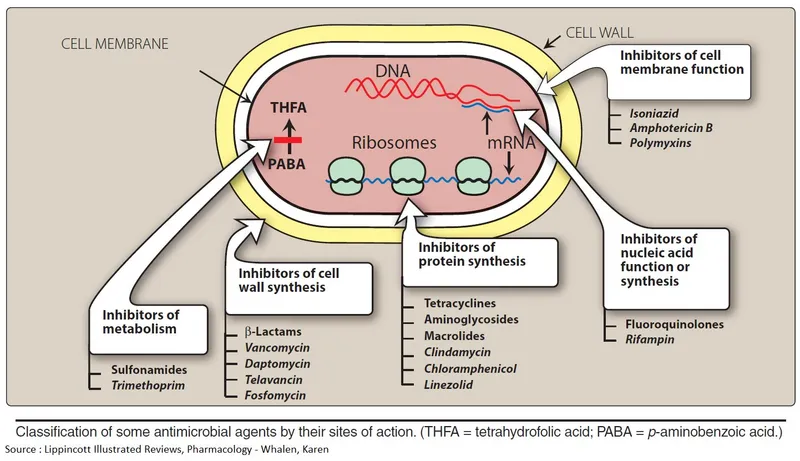
- β-lactam spectrum architecture
- Penicillins: narrow (benzylpenicillin) → antistaphylococcal (nafcillin) → aminopenicillins (ampicillin + E. coli) → antipseudomonal (piperacillin)
- β-lactamase inhibitors restore activity: amoxicillin-clavulanate (MSSA, ESBL-negative E. coli)
- Piperacillin-tazobactam: 85% P. aeruginosa susceptibility, covers ESBL producers (tazobactam inhibits TEM/SHV)
- Cephalosporins: generation correlates with gram-negative coverage, inverse with gram-positive
- 1st generation (cefazolin): MSSA, Proteus, E. coli (surgical prophylaxis gold standard)
- 3rd generation (ceftriaxone): Enterobacteriaceae, S. pneumoniae, N. meningitidis (meningitis empiric)
- 4th generation (cefepime): **+**P. aeruginosa, **+**AmpC stability (ceftriaxone-resistant Enterobacter)
- 5th generation (ceftaroline): **+**MRSA (binds PBP2a), **−**Pseudomonas (narrow gram-negative)
- Carbapenems: broadest spectrum, reserve for ESBL/AmpC producers and severe sepsis
- Imipenem: **+**enterococci, **+**Nocardia, seizure risk 1-2% (avoid if CNS pathology)
- Meropenem: **−**enterococci, **↓**seizure risk (0.5%), preferred for CNS infections
- Ertapenem: **−**Pseudomonas, **−**Acinetobacter, once-daily dosing (outpatient OPAT)
- Penicillins: narrow (benzylpenicillin) → antistaphylococcal (nafcillin) → aminopenicillins (ampicillin + E. coli) → antipseudomonal (piperacillin)
📌 Remember: CAMPFIRE organisms induce chromosomal AmpC β-lactamases-Citrobacter, Aeromonas, Morganella, Providencia, Freundii (Enterobacter), Indole-positive Proteus, Retgeri (Serratia), Enterobacter. Avoid 3rd-gen cephalosporins (select resistance); use cefepime or carbapenems.
- Gram-positive specialist agents
- Vancomycin: MRSA gold standard, **↓**penetration (lung 20-30% serum), nephrotoxicity 15-20%
- MIC creep: MRSA isolates with vancomycin MIC ≥2 μg/mL show ↑treatment failure
- Daptomycin: concentration-dependent, 8-10 mg/kg for bacteremia (>6 mg/kg for endocarditis)
- Inactivated by surfactant-never use for pneumonia
- Monitor CPK weekly (myopathy 3-5%, rhabdomyolysis <1%)
- Linezolid: 100% oral bioavailability, excellent tissue penetration (lung, bone, CSF)
- Reversible myelosuppression (30-40% >14 days), serotonin syndrome with SSRIs
- Bacteriostatic-avoid for endocarditis/bacteremia with high inoculum
- Ceftaroline: MRSA β-lactam option, 600 mg q8h (q12h insufficient for bacteremia)
- Vancomycin: MRSA gold standard, **↓**penetration (lung 20-30% serum), nephrotoxicity 15-20%
⭐ Clinical Pearl: Vancomycin failure in MRSA bacteremia (persistent positivity >5-7 days) occurs in 15-20%, associated with MIC ≥1.5 μg/mL, endovascular infection, or inadequate AUC. Switch to daptomycin 8-10 mg/kg + ceftaroline 600 mg q8h for synergy (combination therapy shows ↓mortality in observational studies).
💊 Antimicrobial Mastery: The Pharmacodynamic Framework
🧬 Resistance Mechanisms: The Bacterial Counterstrike
Antimicrobial resistance (AMR) threatens modern medicine, with >700,000 annual deaths projected to reach 10 million by 2050 without intervention. Resistance mechanisms-enzymatic degradation, target modification, efflux pumps, permeability barriers-evolve through mutation and horizontal gene transfer, selected by antibiotic pressure.
β-lactamase Evolution: From TEM to NDM
β-lactamases hydrolyze β-lactam rings, categorized by Ambler molecular class (A-D) and Bush-Jacoby functional groups. Extended-spectrum β-lactamases (ESBLs) and carbapenemases represent escalating threats, with carbapenem-resistant Enterobacteriaceae (CRE) conferring 40-50% mortality in bloodstream infections.
- ESBL architecture
- Derived from TEM-1, SHV-1, and CTX-M families through point mutations
- Hydrolyze penicillins, cephalosporins (including 3rd/4th generation), aztreonam
- Inhibited by clavulanate, tazobactam, sulbactam-preserved activity for β-lactamase inhibitor combinations
- Amoxicillin-clavulanate, piperacillin-tazobactam retain 70-85% susceptibility
- Prevalence: 15-30% of E. coli in community-acquired UTI (India, Southeast Asia), >40% in healthcare settings
- Risk factors: recent antibiotic exposure (90 days), healthcare contact, international travel
📌 Remember: ESBL TRAP for risk factors-Travel (endemic areas), Recent antibiotics (<90d), Age >65, Prior ESBL infection (50-70% recurrence within 1 year). Empiric carbapenem if ≥2 risk factors + sepsis.
- Carbapenemase classification
- Class A serine carbapenemases: KPC (most common in USA, 30-40% CRE)
- Inhibited by avibactam, vaborbactam-ceftazidime-avibactam >90% susceptibility
- Class B metallo-β-lactamases (MBLs): NDM, VIM, IMP (dominant in South Asia, >60% CRE)
- Require zinc for activity-not inhibited by clavulanate or avibactam
- Aztreonam remains stable (monobactam structure) but requires MBL inhibitor combination
- Class D oxacillinases: OXA-48 (Middle East, Mediterranean), weak carbapenemase activity
- May show carbapenem susceptibility in vitro despite resistance gene
- Detection: modified carbapenem inactivation method (mCIM), molecular PCR for resistance genes
- Class A serine carbapenemases: KPC (most common in USA, 30-40% CRE)
| Resistance Mechanism | Enzyme/Mutation | Affected Drugs | Preserved Activity | Detection Method | Prevalence |
|---|---|---|---|---|---|
| ESBL (TEM, SHV, CTX-M) | Class A β-lactamase | 3rd/4th cephalosporins, aztreonam | Carbapenems, cephamycins | ESBL phenotypic test | 15-40% E. coli |
| KPC carbapenemase | Class A serine | All β-lactams including carbapenems | Ceftazidime-avibactam | mCIM, PCR | 30-40% USA CRE |
| NDM metallo-β-lactamase | Class B zinc-dependent | All β-lactams except aztreonam | Aztreonam + MBL inhibitor | EDTA test, PCR | 60-70% South Asia CRE |
| OXA-48 carbapenemase | Class D serine | Carbapenems (variable), penicillins | Ceftazidime-avibactam (variable) | Temocillin disk test | 20-30% Mediterranean CRE |
| AmpC overexpression | Class C chromosomal | 3rd-gen cephalosporins, cephamycins | Cefepime, carbapenems | AmpC disk test | 10-15% Enterobacter |
⭐ Clinical Pearl: CRE bacteremia requires combination therapy for severe infections (septic shock, high-risk sources). Preferred regimens: ceftazidime-avibactam 2.5 g q8h + aztreonam 2 g q8h (covers both KPC and MBL), or meropenem 2 g q8h extended + colistin 5 mg/kg loading, then 2.5 mg/kg q12h (synergy for KPC).
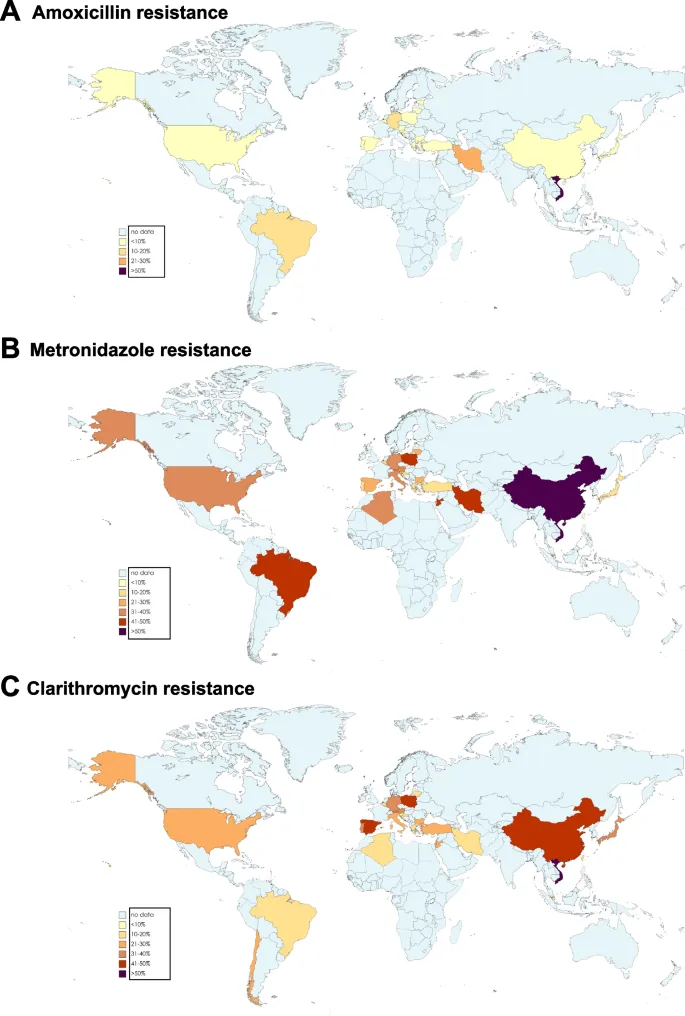
Glycopeptide and Oxazolidinone Resistance
Vancomycin resistance in enterococci (VRE) emerged in the 1980s, mediated by van gene clusters modifying peptidoglycan D-Ala-D-Ala targets to D-Ala-D-Lac (↓affinity 1000×). MRSA vancomycin resistance remains rare (<1%), but heterogeneous vancomycin-intermediate S. aureus (hVISA) causes cryptic treatment failures.
- Vancomycin resistance mechanisms
- VanA phenotype: inducible high-level resistance (vancomycin MIC >64 μg/mL, teicoplanin >16 μg/mL)
- Transferable plasmid-mediated, most common VRE type (>80%)
- VanB phenotype: variable resistance (vancomycin MIC 16-512 μg/mL, teicoplanin susceptible)
- Chromosomal or plasmid, 15-20% VRE
- VanC phenotype: intrinsic low-level resistance (E. gallinarum, E. casseliflavus)
- Vancomycin MIC 2-32 μg/mL, not clinically significant
- hVISA: subpopulations with vancomycin MIC 4-8 μg/mL (standard MIC ≤2 μg/mL)
- Detected by population analysis profile (PAP), not routine testing
- Associated with vancomycin treatment failure despite "susceptible" MIC
- VanA phenotype: inducible high-level resistance (vancomycin MIC >64 μg/mL, teicoplanin >16 μg/mL)
💡 Master This: VRE bacteremia requires daptomycin 8-10 mg/kg/day (monitor CPK) or linezolid 600 mg q12h (monitor CBC). Daptomycin preferred for endocarditis (bactericidal); linezolid for pneumonia/CNS (excellent penetration). Combination therapy (daptomycin + ampicillin or ceftaroline) shows synergy in vitro but unclear clinical benefit.
- Linezolid resistance emergence
- Mechanism: 23S rRNA mutations (G2576T) or cfr methyltransferase gene
- Prevalence: <1% globally, but 5-10% in prolonged therapy (>28 days)
- Cross-resistance: cfr confers resistance to linezolid, tedizolid, chloramphenicol, florfenicol
- Alternatives: tedizolid (lower resistance selection), daptomycin, tigecycline
🧬 Resistance Mechanisms: The Bacterial Counterstrike
🛡️ HIV/AIDS: The Immune Erosion Paradigm
HIV infects 38 million globally, with 1.5 million new infections and 650,000 deaths annually despite antiretroviral therapy (ART). Understanding HIV pathogenesis-CD4 depletion kinetics, viral reservoirs, immune reconstitution-enables prediction of opportunistic infection (OI) risk and optimization of treatment timing.
HIV Pathogenesis: The CD4 Countdown
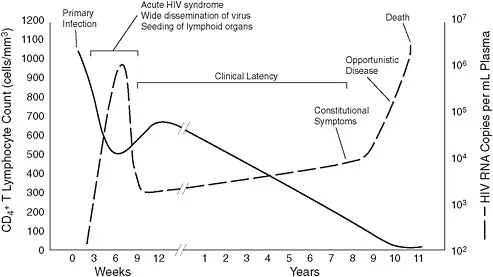
HIV-1 preferentially infects CD4+ T cells via gp120 binding to CD4 and CCR5/CXCR4 coreceptors. Acute infection produces 10⁶-10⁷ copies/mL viremia, followed by immune-mediated suppression to set point (10³-10⁵ copies/mL). Without treatment, CD4 count declines 50-100 cells/μL/year, reaching AIDS threshold (<200 cells/μL) in 8-10 years (median).
- HIV natural history stages
- Acute HIV syndrome (50-90% of infections): fever, lymphadenopathy, rash at 2-4 weeks post-exposure
- High viremia (>10⁶ copies/mL), transient CD4 drop, window period (antibody-negative)
- Diagnosis: HIV RNA PCR (detectable 10-14 days post-exposure vs antibody 3-12 weeks)
- Chronic asymptomatic phase: 5-10 years (variable), gradual CD4 decline
- Set point viral load predicts progression: >100,000 copies/mL → AIDS in 3-5 years, <10,000 → >10 years
- AIDS-defining conditions: CD4 <200 cells/μL or specific OIs regardless of CD4
- CD4 <200: Pneumocystis jirovecii pneumonia (PCP), disseminated histoplasmosis
- CD4 <100: toxoplasma encephalitis, cryptococcal meningitis
- CD4 <50: Mycobacterium avium complex (MAC), CMV retinitis
- Acute HIV syndrome (50-90% of infections): fever, lymphadenopathy, rash at 2-4 weeks post-exposure
📌 Remember: CCMMTT for AIDS-defining OIs by CD4 threshold-Candida esophagitis (<200), Cryptococcus (<100), MAC (<50), Mycobacterium tuberculosis (any CD4), Toxoplasma (<100), Tuberculosis (any CD4, but ↑risk if <350)
- Opportunistic infection risk stratification
- CD4 350-500: ↑TB reactivation risk (3-5× baseline), bacterial pneumonia (5-10× baseline)
- CD4 200-350: oral candidiasis (20-30%), herpes zoster (5-10%)
- CD4 100-200: PCP (40% lifetime risk without prophylaxis), disseminated endemic fungi
- PCP prophylaxis: TMP-SMX DS 1 tab daily or 3× weekly (↓risk >90%)
- CD4 50-100: toxoplasma encephalitis (30% if seropositive), cryptococcal meningitis (5-10%)
- Toxoplasma prophylaxis: TMP-SMX DS (dual coverage with PCP) if IgG-positive
- CD4 <50: MAC bacteremia (20-40% without prophylaxis), CMV retinitis (15-30%)
- MAC prophylaxis: azithromycin 1200 mg weekly (preferred) or clarithromycin 500 mg BID
⭐ Clinical Pearl: Immune reconstitution inflammatory syndrome (IRIS) occurs in 10-25% starting ART, typically 2-8 weeks after initiation. Paradoxical worsening of treated OI (TB-IRIS most common, 15-20%) or unmasking of subclinical infection. Treat with corticosteroids (prednisone 1 mg/kg/day) if severe; continue ART unless life-threatening.
Antiretroviral Therapy: The Suppression Strategy
Modern ART achieves viral suppression (<50 copies/mL) in >95% with single-tablet regimens. Integrase strand transfer inhibitors (INSTIs) form the backbone of first-line therapy, combined with nucleoside reverse transcriptase inhibitors (NRTIs) for high genetic barrier to resistance.
| ART Class | Mechanism | Key Agents | Advantages | Disadvantages | Resistance Barrier |
|---|---|---|---|---|---|
| NRTIs | Chain termination | Tenofovir alafenamide (TAF), emtricitabine (FTC) | Well-tolerated, dual HBV activity | Mitochondrial toxicity (stavudine, zidovudine) | Low (single mutation) |
| NNRTIs | Allosteric RT inhibition | Efavirenz, rilpivirine, doravirine | High potency, low pill burden | CNS effects (efavirenz), low barrier (single mutation) | Low |
| PIs | Protease inhibition | Darunavir, atazanavir | High barrier, salvage therapy | GI effects, drug interactions (CYP3A4) | High (multiple mutations) |
| INSTIs | Integrase inhibition | Dolutegravir, bictegravir, cabotegravir | Rapid suppression, high barrier, minimal interactions | Neural tube defects (dolutegravir, periconception) | High (dolutegravir, bictegravir) |
| Entry inhibitors | CCR5 blockade | Maraviroc | Tropism-specific | Requires tropism testing (CCR5 vs CXCR4) | Moderate |
💡 Master This: First-line ART regimens-bictegravir/TAF/FTC (Biktarvy) or dolutegravir + TAF/FTC-achieve 95-98% virologic suppression at 48 weeks. Start ART immediately upon diagnosis (same-day initiation safe, ↑linkage to care). Delay only for TB meningitis (↑IRIS risk) or cryptococcal meningitis (start 4-6 weeks after antifungal initiation).
- ART resistance and salvage therapy
- Transmitted resistance: 10-15% in treatment-naive patients (NNRTIs most common)
- Acquired resistance: virologic failure (viral load >200 copies/mL after suppression or failure to suppress)
- NRTI resistance: M184V mutation (FTC/3TC) emerges first, minimal clinical impact with INSTI backbone
- NNRTI resistance: single mutation (K103N, Y181C) confers high-level resistance, class cross-resistance
- INSTI resistance: rare with dolutegravir/bictegravir (<1%), higher with raltegravir/elvitegravir (3-5%)
- Genotypic testing: perform at baseline (detect transmitted resistance) and virologic failure
- Salvage regimens: use ≥2 fully active agents from different classes, often including boosted PI + INSTI
🛡️ HIV/AIDS: The Immune Erosion Paradigm
🔥 Sepsis and Septic Shock: The Dysregulated Host Response
Sepsis affects 49 million and kills 11 million annually worldwide, representing 20% of global deaths. Sepsis-3 definitions emphasize organ dysfunction (Sequential Organ Failure Assessment [SOFA] score ≥2) over systemic inflammatory response syndrome (SIRS), recognizing dysregulated host response-not just infection-drives mortality.
Sepsis Recognition: The SOFA Framework
Sepsis requires documented or suspected infection plus acute organ dysfunction (↑SOFA ≥2 points). Septic shock adds persistent hypotension requiring vasopressors (MAP ≥65 mmHg) and lactate >2 mmol/L despite adequate fluid resuscitation-conferring >40% mortality vs 10% for sepsis without shock.
- SOFA score components (0-4 points each)
- Respiration: PaO₂/FiO₂ ratio (<400 = 1 point, <100 = 4 points)
- Coagulation: platelets (<150,000 = 1 point, <20,000 = 4 points)
- Liver: bilirubin (1.2-1.9 mg/dL = 1 point, >12 mg/dL = 4 points)
- Cardiovascular: MAP <70 mmHg or vasopressors (dopamine ≤5 μg/kg/min = 2 points, norepinephrine >0.1 μg/kg/min = 4 points)
- CNS: Glasgow Coma Scale (13-14 = 1 point, <6 = 4 points)
- Renal: creatinine (**1.2-1.9 mg/
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app


















