Hospital Acquired Infections

On this page
🦠 Hospital Acquired Infections: The Invisible Battlefield
Hospital-acquired infections transform places of healing into battlegrounds where patients face threats they never anticipated. You'll master how to identify the most dangerous nosocomial pathogens, understand why standard defenses fail, and deploy evidence-based prevention strategies that save lives. From recognizing early infection patterns to implementing rapid treatment protocols, you'll build the clinical judgment needed to protect vulnerable patients and interrupt transmission chains before they spread.
⭐ Critical Reality: HAIs affect 1.7 million patients annually in the US, causing 99,000 deaths and costing healthcare systems $28-45 billion yearly.
The HAI Landscape: Epidemiological Architecture
Hospital-acquired infections follow predictable patterns that reveal systematic vulnerabilities:
- Temporal Definition: Infections occurring ≥48 hours after hospital admission
- Excludes community-acquired infections in incubation phase
- Includes infections up to 30 days post-discharge for surgical sites
- 90-day window for implant-related infections
- Prevalence Distribution:
- Urinary tract infections: 32% of all HAIs
- Surgical site infections: 22% of cases
- Bloodstream infections: 14% of occurrences
- Pneumonia: 15% of hospital-acquired cases
📌 Remember: CUPS - Catheter UTIs (32%), Under-the-knife SSIs (22%), Pneumonia (15%), Sepsis/bloodstream (14%)
| HAI Type | Prevalence | Primary Pathogens | Mortality Rate | Cost per Episode | Prevention Focus |
|---|---|---|---|---|---|
| CAUTI | 32% | E. coli, Enterococcus | 2-3% | $13,793 | Catheter stewardship |
| SSI | 22% | S. aureus, E. coli | 3-5% | $25,546 | Perioperative bundles |
| CLABSI | 14% | CoNS, S. aureus | 12-25% | $45,814 | Central line care |
| VAP | 15% | P. aeruginosa, MRSA | 20-50% | $40,144 | Ventilator weaning |
| CDI | 12% | C. difficile | 6-8% | $11,285 | Antibiotic stewardship |
The microbial ecosystem within hospitals creates unique selective pressures favoring antibiotic-resistant organisms. ESKAPE pathogens dominate HAI epidemiology, representing >70% of multidrug-resistant infections.
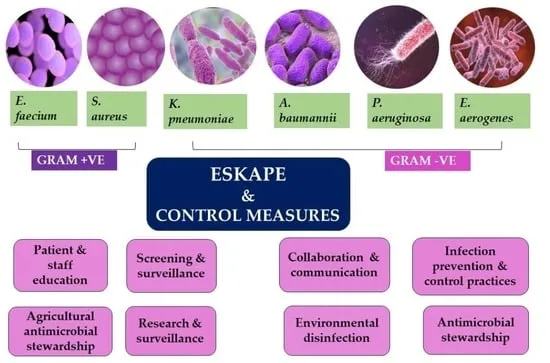
Understanding HAI epidemiology provides the foundation for recognizing high-risk scenarios and implementing targeted prevention strategies that address specific pathogen-device combinations.
🦠 Hospital Acquired Infections: The Invisible Battlefield
🎯 Pathogen Warfare: The Microbial Arsenal
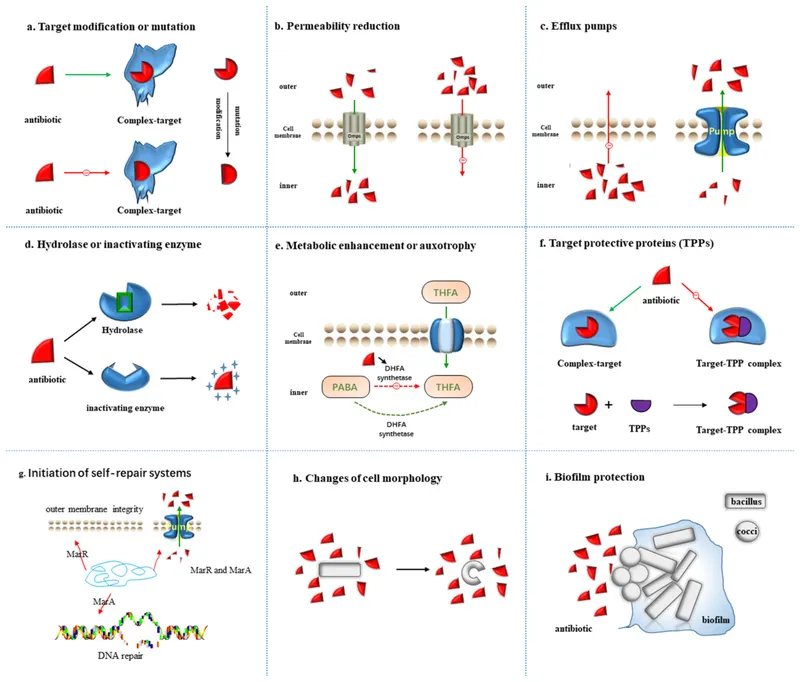
Hospital pathogens employ distinct strategies that exploit healthcare vulnerabilities, creating predictable infection patterns based on organism characteristics and environmental pressures.
ESKAPE Pathogen Mastery Framework
The ESKAPE acronym represents the most dangerous HAI pathogens, each with unique resistance mechanisms and clinical presentations:
- Enterococcus faecium: Vancomycin resistance via vanA/vanB genes
- Intrinsic ampicillin resistance in E. faecium (vs. sensitive E. faecalis)
- Survives >7 days on dry surfaces
- Causes 15% of healthcare-associated bacteremia
- Staphylococcus aureus: Methicillin resistance via mecA gene
- MRSA prevalence: 46% of S. aureus HAIs
- Biofilm formation on medical devices
- 30-day mortality: 20-25% for MRSA bacteremia
- Klebsiella pneumoniae: Carbapenemase production (KPC, NDM, OXA-48)
- CRE (Carbapenem-Resistant Enterobacteriaceae) mortality: 40-50%
- Hypervirulent strains cause >90% mortality in immunocompromised
- Acinetobacter baumannii: Multidrug resistance via efflux pumps
- Survives >150 days on dry surfaces
- XDR (Extensively Drug-Resistant) rates: >60% in ICUs
- Pseudomonas aeruginosa: Intrinsic resistance plus acquired mechanisms
- MDR prevalence: 35% of P. aeruginosa isolates
- Biofilm production increases antibiotic MIC by 1000-fold
- Enterobacter species: AmpC β-lactamase induction
- Cephalosporin resistance develops during therapy in >20% cases
📌 Remember: ESKAPE Artists - Enterococci Survive Klebsiella Kills Acinetobacter Adheres Pseudomonas Persists Enterobacter Evolves
Resistance Mechanism Categories
Hospital pathogens develop resistance through four primary mechanisms, each requiring specific therapeutic approaches:
- Enzymatic Inactivation (40% of resistance)
- β-lactamases: >1000 variants identified
- ESBL prevalence: 25% of E. coli, 35% of K. pneumoniae
- Carbapenemases: KPC (45%), NDM (30%), OXA-48 (20%)
- Target Modification (30% of resistance)
- mecA gene: Altered PBP2a in MRSA
- vanA/vanB: Modified peptidoglycan precursors
- Quinolone resistance: gyrA/parC mutations
- Efflux Pump Overexpression (20% of resistance)
- MexAB-OprM in P. aeruginosa
- AcrAB-TolC in Enterobacteriaceae
- Increases MIC by 4-32 fold
- Permeability Changes (10% of resistance)
- Porin loss in OmpK35/36 (Klebsiella)
- OprD deficiency (Pseudomonas)
⭐ Clinical Pearl: Combination resistance occurs in >60% of ESKAPE isolates, requiring susceptibility-guided therapy rather than empirical treatment protocols.
| Pathogen | Primary Resistance | Survival Time | Biofilm Capacity | Mortality Rate | Key Clinical Feature |
|---|---|---|---|---|---|
| E. faecium | VanA/VanB (85%) | 7 days | Moderate | 15% | Ampicillin intrinsic resistance |
| MRSA | mecA (46%) | 14 days | High | 20-25% | Device-associated infections |
| K. pneumoniae | KPC (25%) | 30 days | High | 40-50% | Hypervirulent strains |
| A. baumannii | MDR (60%) | 150 days | Very High | 35-45% | Environmental persistence |
| P. aeruginosa | Efflux (35%) | 21 days | Very High | 25-35% | Intrinsic resistance |
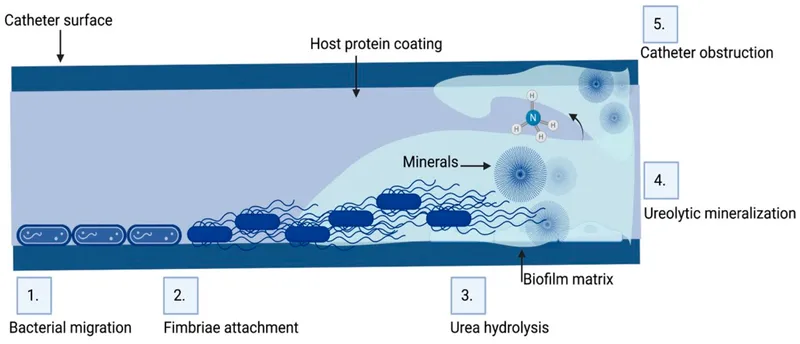
Pathogen-specific characteristics determine transmission patterns, with environmental survivors (Acinetobacter, Enterococcus) causing outbreak scenarios, while biofilm producers (Staphylococcus, Pseudomonas) dominate device-associated infections. This knowledge guides targeted prevention strategies and outbreak investigation approaches.
🎯 Pathogen Warfare: The Microbial Arsenal
🛡️ Prevention Strategies: The Defense Matrix
Bundle-Based Prevention Architecture
Evidence-based bundles represent all-or-nothing intervention packages where 100% compliance with every element achieves maximum risk reduction:
- Central Line Bundle (CLABSI Prevention)
- Hand hygiene before insertion (99% compliance required)
- Maximal sterile barriers during insertion
- Chlorhexidine skin antisepsis (>0.5% concentration)
- Optimal catheter site selection (subclavian > internal jugular > femoral)
- Daily review of line necessity with prompt removal
- Impact: 66% reduction in CLABSI rates when >95% compliant
📌 Remember: CHAMP Bundle - Chlorhexidine prep, Hand hygiene, Aseptic insertion, Maximal barriers, Prompt removal
- Ventilator Bundle (VAP Prevention)
- Head of bed elevation 30-45 degrees (unless contraindicated)
- Daily sedation vacation and spontaneous breathing trials
- Peptic ulcer prophylaxis for high-risk patients
- DVT prophylaxis unless contraindicated
- Daily oral care with chlorhexidine 0.12%
- Subglottic secretion drainage when available
- Impact: 45% VAP reduction with >90% bundle compliance
Device-Specific Prevention Protocols
Each invasive device requires tailored prevention strategies based on pathogen ecology and insertion site characteristics:
-
Urinary Catheter Management
- Aseptic insertion with sterile technique
- Smallest appropriate catheter size (14-16 Fr for adults)
- Closed drainage system maintenance
- Daily necessity assessment (target <5 days duration)
- Proper positioning: Drainage bag below bladder level
- CAUTI reduction: 32% decrease with systematic protocols
-
Surgical Site Protection
- Preoperative antibiotic prophylaxis within 60 minutes of incision
- Appropriate hair removal (clippers, not razors)
- Normothermia maintenance (>36°C perioperatively)
- Glucose control (<180 mg/dL in diabetics)
- Smoking cessation ≥30 days preoperatively
- SSI reduction: 27% decrease with complete bundle adherence
⭐ Clinical Pearl: Catheter-days serve as the primary denominator for HAI rates - CLABSI rate = (Number of CLABSIs × 1000) ÷ Central line-days, with benchmark targets <1.0 per 1000 catheter-days.
| Prevention Bundle | Core Elements | Compliance Target | Risk Reduction | Monitoring Metric | Implementation Barrier |
|---|---|---|---|---|---|
| Central Line | 5 components | >95% | 66% CLABSI ↓ | Per 1000 line-days | Sterile barrier cost |
| Ventilator | 6 components | >90% | 45% VAP ↓ | Per 1000 vent-days | Sedation protocols |
| Urinary Catheter | 4 components | >85% | 32% CAUTI ↓ | Per 1000 cath-days | Necessity assessment |
| Surgical Site | 7 components | >80% | 27% SSI ↓ | Per 100 procedures | Timing coordination |
| Hand Hygiene | 5 moments | >80% | 40% overall HAI ↓ | Opportunities observed | Behavioral change |
Hospital environments require systematic decontamination protocols targeting high-touch surfaces and pathogen reservoirs:
-
Terminal Cleaning Protocols
- UV-C disinfection: 99.9% pathogen reduction in 15-30 minutes
- Hydrogen peroxide vapor: 6-log reduction of spores
- Enhanced cleaning of high-touch surfaces every 2-4 hours
- ATP bioluminescence monitoring for cleaning verification
-
Isolation Precautions Implementation
- Contact precautions: Gown and gloves for MDRO patients
- Droplet precautions: Surgical mask within 3 feet
- Airborne precautions: N95 respirator in negative pressure rooms
- Cohorting strategies: Geographic separation during outbreaks
💡 Master This: Environmental persistence determines cleaning frequency - C. difficile spores survive >5 months, requiring sporicidal agents, while vegetative bacteria succumb to standard disinfectants within minutes.

Prevention success depends on systematic implementation rather than individual interventions, with bundle compliance serving as the primary predictor of HAI reduction. Understanding device-specific risks enables targeted resource allocation and monitoring strategies.
🛡️ Prevention Strategies: The Defense Matrix
🔬 Diagnostic Strategies: Detection Mastery
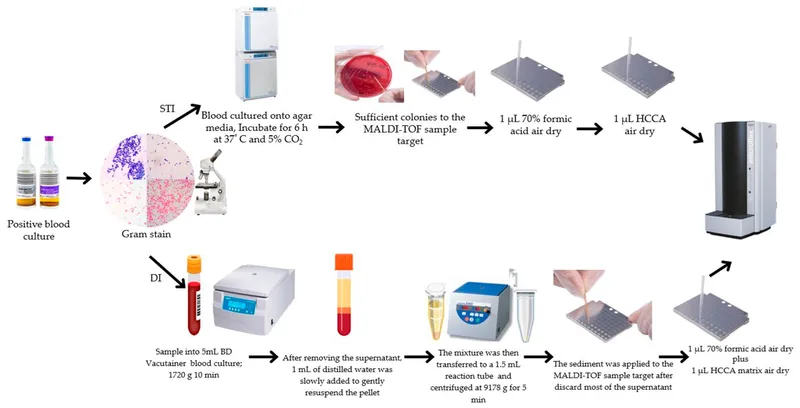
Diagnostic accuracy determines treatment appropriateness, with false positives leading to unnecessary antibiotic exposure and false negatives causing delayed intervention and increased mortality.
Clinical Syndrome Recognition Patterns
HAI diagnosis relies on syndrome-specific criteria that combine clinical signs with objective markers:
-
Bloodstream Infection Criteria
- Primary BSI: Positive blood culture unrelated to infection at another site
- Secondary BSI: Bloodstream infection related to documented infection elsewhere
- Clinical signs: Fever >38°C or hypothermia <36°C, plus ≥1 of:
- Rigors, altered mental status, oliguria, hypotension
- Laboratory markers: WBC >12,000 or <4,000, >10% bands
- Contamination exclusion: ≥2 positive cultures for CoNS or common skin flora
-
Pneumonia Diagnostic Framework
- Clinical criteria: New/progressive infiltrate plus ≥2 of:
- Fever >38°C, leukocytosis >12,000, purulent secretions
- Ventilator-associated: Pneumonia occurring ≥48 hours after intubation
- Microbiological thresholds:
- Quantitative cultures: ≥10^5 CFU/mL (endotracheal aspirate)
- BAL: ≥10^4 CFU/mL, PSB: ≥10^3 CFU/mL
- Biomarkers: Procalcitonin >0.5 ng/mL supports bacterial etiology
- Clinical criteria: New/progressive infiltrate plus ≥2 of:
📌 Remember: SIRS Plus Source - Systemic inflammatory response Infection Requires Source identification Plus Lab Unequivocal Signs
Laboratory Diagnostic Thresholds
Microbiological diagnosis requires quantitative interpretation to distinguish infection from colonization:
-
Urine Culture Interpretation
- Symptomatic patients: ≥10^3 CFU/mL (straight catheter)
- Asymptomatic patients: ≥10^5 CFU/mL (indwelling catheter)
- Mixed flora: ≥3 organisms suggests contamination
- Pyuria threshold: ≥10 WBC/hpf supports infection
-
Respiratory Specimen Analysis
- Sputum quality: <10 epithelial cells, >25 PMNs per lpf
- Endotracheal aspirate: ≥10^5 CFU/mL significant
- Bronchoalveolar lavage: ≥10^4 CFU/mL diagnostic
- Protected specimen brush: ≥10^3 CFU/mL confirms VAP
-
Blood Culture Optimization
- Volume requirements: 20-30 mL per culture set (adults)
- Number of sets: ≥2 sets from separate sites
- Timing: Before antibiotics when possible, 15-30 minutes apart
- Yield optimization: >90% sensitivity with 3 sets within 24 hours
⭐ Clinical Pearl: Time to positivity in blood cultures predicts clinical significance - CoNS growing in <12 hours suggests true bacteremia, while >24 hours indicates likely contamination.
| Specimen Type | Significant Threshold | Collection Method | Processing Time | Sensitivity | Specificity |
|---|---|---|---|---|---|
| Blood Culture | Any growth (non-CoNS) | 20-30 mL per set | 5 days incubation | 90-95% | 95-98% |
| Urine (catheter) | ≥10^3 CFU/mL | Aseptic aspiration | 24-48 hours | 85-90% | 90-95% |
| Respiratory (BAL) | ≥10^4 CFU/mL | Bronchoscopic | 24-48 hours | 80-85% | 85-90% |
| Wound Culture | Moderate/Heavy growth | Deep tissue | 24-72 hours | 75-80% | 80-85% |
| CSF Culture | Any growth | Lumbar puncture | 24-48 hours | 95-99% | 98-99% |
Modern diagnostic platforms enable faster pathogen identification and resistance detection:
-
Molecular Diagnostics
- PCR-based panels: Results in 1-6 hours vs. 24-72 hours for culture
- Blood culture identification: MALDI-TOF provides species ID in <30 minutes
- Resistance gene detection: mecA, vanA/B, KPC within 2 hours
- Syndromic panels: Respiratory, GI, meningitis panels available
-
Biomarker Integration
- Procalcitonin: >0.5 ng/mL suggests bacterial infection
- C-reactive protein: >100 mg/L supports severe bacterial infection
- Lactate: >2 mmol/L indicates tissue hypoperfusion
- Presepsin: >600 pg/mL predicts sepsis with 85% sensitivity
💡 Master This: Rapid diagnostics reduce time to appropriate therapy by 24-48 hours, decreasing mortality by 15-20% and length of stay by 1-2 days in critically ill patients.
Diagnostic precision requires integration of clinical criteria, quantitative microbiology, and rapid technologies to distinguish true infections from colonization, enabling targeted therapy and antimicrobial stewardship.
🔬 Diagnostic Strategies: Detection Mastery
⚔️ Treatment Algorithms: Therapeutic Warfare
Empirical Therapy Decision Framework
Initial antibiotic selection requires risk stratification based on patient factors, institutional resistance patterns, and infection severity:
-
MDRO Risk Factor Assessment
- Prior antibiotic exposure within 90 days
- Hospitalization ≥5 days or ICU stay
- Immunosuppression: Steroids >20 mg prednisone daily
- Invasive devices: Central lines, ventilators, urinary catheters
- Known MDRO colonization or prior MDRO infection
- High local resistance rates: >20% for specific pathogen-antibiotic combinations
-
Severity Stratification Criteria
- Severe/Septic: qSOFA ≥2, lactate >2 mmol/L, organ dysfunction
- Moderate: Localized infection without systemic signs
- Mild: Minimal symptoms, stable vital signs
Pathogen-Specific Treatment Protocols
Definitive therapy targets identified organisms with narrowest effective spectrum:
-
MRSA Treatment Options
- Vancomycin: 15-20 mg/kg q8-12h, target trough 15-20 μg/mL
- Linezolid: 600 mg q12h PO/IV, avoid >28 days (thrombocytopenia)
- Daptomycin: 6-10 mg/kg daily IV, monitor CPK weekly
- Ceftaroline: 600 mg q12h IV, active against MRSA
- Duration: 7-14 days (uncomplicated), 4-6 weeks (endocarditis)
-
Carbapenem-Resistant Enterobacteriaceae (CRE)
- Combination therapy preferred for serious infections
- Meropenem + colistin: If MIC ≤8 μg/mL
- Ceftazidime-avibactam: 2.5 g q8h IV for KPC producers
- Meropenem-vaborbactam: 4 g q8h IV for KPC, some OXA-48
- Polymyxin B: 2.5-3 mg/kg daily, monitor nephrotoxicity
-
Multidrug-Resistant Pseudomonas
- Combination therapy: β-lactam + aminoglycoside or fluoroquinolone
- Ceftolozane-tazobactam: 1.5 g q8h IV
- Ceftazidime-avibactam: 2.5 g q8h IV
- Imipenem-relebactam: 500 mg q6h IV
- Colistin: 5 mg/kg loading dose, then 2.5 mg/kg q12h
📌 Remember: MRSA Triple Threat - Vancomycin (trough 15-20), Linezolid (limit 28 days), Daptomycin (check CPK)
| Pathogen | First-Line Therapy | Alternative Options | Duration | Monitoring Parameters | Resistance Concerns |
|---|---|---|---|---|---|
| MRSA | Vancomycin 15-20 mg/kg | Linezolid, Daptomycin | 7-14 days | Trough levels, SCr | Vancomycin MIC creep |
| CRE | Combination therapy | Ceftazidime-avibactam | 10-14 days | Renal function | Pan-resistance |
| MDR Pseudomonas | Ceftolozane-tazobactam | Combination therapy | 7-10 days | Renal function | Efflux pumps |
| VRE | Linezolid | Daptomycin, Tigecycline | 7-14 days | CBC, platelets | Linezolid resistance |
| ESBL E. coli | Carbapenem | Ceftazidime-avibactam | 7-10 days | Renal function | Carbapenemase |
Therapeutic success requires pharmacokinetic optimization and clinical monitoring:
-
Dosing Optimization Principles
- Time-dependent killing: β-lactams require prolonged infusions
- Concentration-dependent: Aminoglycosides, fluoroquinolones need peak optimization
- AUC-dependent: Vancomycin dosing based on AUC/MIC >400
- Renal adjustment: Creatinine clearance-based dosing modifications
-
Therapeutic Drug Monitoring
- Vancomycin: AUC monitoring preferred over trough levels
- Aminoglycosides: Peak and trough levels, extended-interval dosing
- Colistin: Steady-state levels 2-3 mg/L for efficacy
- Voriconazole: Trough >1 μg/mL, <5 μg/mL to avoid toxicity
-
De-escalation Criteria
- Culture results available with susceptibilities
- Clinical improvement: Fever resolution, WBC normalization
- Source control achieved when applicable
- Narrow spectrum agent available with equal efficacy
⭐ Clinical Pearl: Combination therapy for CRE reduces mortality by 20-30% compared to monotherapy, but increases nephrotoxicity risk by 15-25% - monitor creatinine daily.
💡 Master This: Time to appropriate therapy represents the most critical factor in HAI outcomes - each hour delay increases mortality by 7-10% in severe infections.
Treatment algorithms must balance broad initial coverage with rapid de-escalation, using institutional antibiograms and patient-specific factors to optimize outcomes while minimizing resistance development and adverse effects.
⚔️ Treatment Algorithms: Therapeutic Warfare
🌐 Systems Integration: The Infection Control Ecosystem
Surveillance System Architecture
Modern HAI surveillance combines automated detection with clinical validation to enable real-time monitoring and rapid response:
-
Electronic Surveillance Capabilities
- Automated case detection: Laboratory-based algorithms identify >80% of HAIs
- Real-time alerts: Positive cultures trigger immediate notifications
- Risk stratification: Predictive models identify high-risk patients
- Outcome tracking: Length of stay, mortality, readmission monitoring
- Benchmarking: NHSN reporting enables national comparisons
-
Key Performance Indicators (KPIs)
- HAI rates: Device-associated infections per 1000 device-days
- Process measures: Bundle compliance rates >90% target
- Outcome measures: Standardized infection ratios (SIR) <1.0 goal
- Antimicrobial metrics: Days of therapy, defined daily doses
- Cost indicators: HAI-attributable costs per patient-day
Multidisciplinary Team Integration
Effective infection control requires coordinated expertise across clinical specialties and support services:
-
Core Team Composition
- Infection Preventionist: Surveillance, investigation, education
- Hospital Epidemiologist: Medical oversight, policy development
- Clinical Pharmacist: Antimicrobial stewardship, resistance monitoring
- Microbiology Director: Laboratory liaison, diagnostic optimization
- Quality Officer: Performance measurement, improvement initiatives
- Environmental Services: Cleaning protocols, disinfection validation
-
Specialty Integration Points
- ICU Teams: Device management, bundle implementation
- Surgical Services: Perioperative protocols, SSI prevention
- Emergency Department: Isolation decisions, empirical therapy
- Laboratory: Rapid diagnostics, resistance reporting
- Pharmacy: Formulary management, dosing optimization
📌 Remember: EPIC Teams - Epidemiologist leads, Pharmacist stewards, Infection preventionist monitors, Clinicians implement
Outbreak Investigation Framework
Systematic outbreak response follows epidemiological principles to identify sources, control transmission, and prevent recurrence:
-
Outbreak Definition Criteria
- Temporal clustering: ≥2 cases within epidemiologically linked timeframe
- Geographic clustering: Same unit or shared exposure
- Microbiological similarity: Same species with similar resistance patterns
- Molecular confirmation: PFGE, WGS for definitive relatedness
-
Investigation Steps
- Case identification: Active surveillance for additional cases
- Case-control study: Risk factor analysis for transmission modes
- Environmental sampling: Potential reservoirs and contaminated equipment
- Molecular typing: Strain relatedness confirmation
- Control measures: Enhanced precautions, cohorting, equipment replacement
-
Control Measure Implementation
- Enhanced contact precautions: Gown and gloves for all patient contact
- Cohorting strategies: Geographic separation of colonized/infected patients
- Staff cohorting: Dedicated personnel for affected patients
- Environmental decontamination: Terminal cleaning with sporicidal agents
- Equipment sterilization: Heat-sensitive items require low-temperature methods
⭐ Clinical Pearl: Molecular epidemiology using whole genome sequencing can distinguish true outbreaks from pseudo-outbreaks with >99% accuracy, preventing unnecessary interventions.
| Investigation Phase | Timeline | Key Activities | Success Metrics | Resource Requirements |
|---|---|---|---|---|
| Detection | 0-24 hours | Case identification | Time to recognition | Surveillance system |
| Assessment | 1-3 days | Risk factor analysis | Attack rate calculation | Epidemiologist |
| Control | 3-7 days | Intervention implementation | Transmission cessation | Multidisciplinary team |
| Evaluation | 1-4 weeks | Outcome monitoring | No new cases | Continued surveillance |
| Prevention | Ongoing | System improvements | Policy updates | Quality improvement |
Modern infection control leverages digital health technologies for enhanced surveillance and decision support:
-
Artificial Intelligence Applications
- Predictive modeling: Machine learning identifies high-risk patients with 85% accuracy
- Natural language processing: Automated chart review for HAI detection
- Image recognition: Wound assessment and healing progression monitoring
- Antibiotic optimization: AI-driven dosing recommendations
-
Mobile Health Solutions
- Hand hygiene monitoring: Electronic sensors track compliance rates
- Contact tracing: RFID badges map staff-patient interactions
- Environmental monitoring: Real-time temperature, humidity tracking
- Communication platforms: Secure messaging for outbreak coordination
💡 Master This: Integrated surveillance systems combining automated detection, clinical validation, and real-time reporting reduce HAI investigation time by 60-70% while improving detection sensitivity to >95%.
Systems integration transforms fragmented infection control activities into coordinated quality improvement programs that achieve sustained HAI reduction through data-driven decision making and multidisciplinary collaboration.
🌐 Systems Integration: The Infection Control Ecosystem
🎯 Clinical Mastery Arsenal: Rapid Response Toolkit
Essential HAI Recognition Patterns
Master these high-yield recognition patterns for immediate HAI identification:
-
Device-Associated Infection Triggers
- CLABSI: Fever + positive blood culture in patient with central line >48 hours
- CAUTI: Fever + pyuria in catheterized patient without other source
- VAP: New infiltrate + fever + purulent secretions in ventilated patient >48 hours
- SSI: Wound erythema + drainage within 30 days of surgery
-
MDRO Colonization Clues
- Prior hospitalization within 90 days
- Antibiotic exposure within 30 days
- Transfer from LTCF or dialysis center
- Known MDRO contact or outbreak unit
📌 Remember: CLAV Triggers - Central line fever, Lung infiltrate + vent, Asymptomatic UTI + catheter, Vascular access + bacteremia
Critical Decision Thresholds
Memorize these evidence-based thresholds for immediate clinical decisions:
| Clinical Scenario | Threshold Value | Action Required | Time Frame | Monitoring Parameter |
|---|---|---|---|---|
| Sepsis Recognition | qSOFA ≥2 | Blood cultures + antibiotics | <1 hour | Lactate, vitals |
| MRSA Bacteremia | Any positive culture | Vancomycin + echo | <6 hours | Trough levels |
| CRE Detection | Carbapenem MIC >2 | Contact isolation | Immediate | Surveillance cultures |
| C. diff Suspicion | ≥3 loose stools | Contact precautions | <2 hours | Toxin assay |
| Outbreak Threshold | ≥2 related cases | Investigation team | <24 hours | Additional cases |
Use this systematic approach for any suspected HAI:
- RAPID Assessment Protocol
- Risk factors: Device presence, immunosuppression, prior antibiotics
- Assessment: Vital signs, laboratory values, clinical signs
- Pathogen likelihood: Local resistance patterns, patient factors
- Isolation needs: Transmission precautions based on suspected organism
- Diagnostics: Appropriate cultures before antibiotics when possible
Emergency Response Protocols
Critical situations requiring immediate action:
-
Suspected Outbreak Response
- Immediate: Contact infection control within 15 minutes
- Enhanced surveillance: Screen all unit patients within 24 hours
- Environmental cultures: Sample potential sources immediately
- Staff notification: Alert all caregivers to enhanced precautions
-
MDRO Detection Protocol
- Contact isolation: Implement within 1 hour of identification
- Contact screening: Test roommates and recent contacts
- Environmental cleaning: Terminal clean with appropriate disinfectant
- Staff education: Review transmission prevention immediately
⭐ Clinical Pearl: Time-sensitive interventions - Sepsis antibiotics within 1 hour, MDRO isolation within 1 hour, outbreak investigation within 24 hours - each delay increases adverse outcomes exponentially.
Quick Reference Clinical Tools
Essential numbers for immediate recall:
-
HAI Rate Benchmarks
- CLABSI: <1.0 per 1000 central line-days
- CAUTI: <2.0 per 1000 catheter-days
- VAP: <2.0 per 1000 ventilator-days
- SSI: <2% for clean procedures
-
Antibiotic Dosing Pearls
- Vancomycin: 15-20 mg/kg q8-12h, AUC >400
- Piperacillin-tazobactam: 4.5g q6h, extended infusion
- Meropenem: 2g q8h, 3-hour infusion for resistant organisms
- Colistin: 5 mg/kg loading dose, then 2.5 mg/kg q12h
💡 Master This: Bundle compliance >90% prevents more HAIs than any single intervention - focus on systematic implementation rather than individual components.
Clinical mastery in HAI management requires immediate pattern recognition, systematic assessment, and evidence-based intervention within critical time windows to optimize patient outcomes and prevent transmission.
🎯 Clinical Mastery Arsenal: Rapid Response Toolkit
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























