Infectious Diseases

On this page
🦠 Infectious Disease Mastery: The Clinical Battlefield
Infectious diseases remain humanity's oldest adversary and our most dynamic clinical challenge, demanding you master pathogen behavior, antimicrobial selection, and the race against resistance simultaneously. You'll learn to recognize sepsis in its critical early window, interpret molecular diagnostics that have revolutionized detection, and optimize treatment through precision medicine principles that balance efficacy against the evolutionary pressures driving resistance. This is clinical decision-making where timing, pattern recognition, and strategic thinking converge to save lives.
📌 Remember: SIRS-SOFA-SHOCK - Systemic inflammatory response (≥2 criteria), Sepsis organ failure assessment (≥2 points), Septic shock (MAP <65 mmHg + lactate >2 mmol/L)
The infectious disease landscape operates on three fundamental principles that govern every clinical encounter:
-
Pathogen Recognition Hierarchy
- Primary pathogens: cause disease in healthy hosts (mortality 5-15%)
- Opportunistic pathogens: require immunocompromise (mortality 25-60%)
- CD4 count <200 cells/μL: high-risk threshold
- Neutrophil count <500 cells/μL: severe neutropenia
- Steroid equivalent >20mg prednisone daily for >2 weeks
-
Temporal Pattern Recognition
- Hyperacute onset: <6 hours (meningococcemia, toxic shock)
- Acute onset: 6-72 hours (bacterial pneumonia, UTI)
- Subacute onset: days to weeks (endocarditis, osteomyelitis)
- Chronic onset: weeks to months (tuberculosis, fungal infections)
-
Resistance Pattern Evolution
- MRSA prevalence: 15-50% in healthcare settings
- ESBL producers: 10-25% of Enterobacteriaceae
- Carbapenem resistance: <5% but rising 10-15% annually
| Pathogen Category | Onset Timeline | Mortality Risk | Key Diagnostic Window | Treatment Window |
|---|---|---|---|---|
| Bacterial Sepsis | 6-24 hours | 20-40% | First 3 hours | Hour 1 critical |
| Viral Syndromes | 1-7 days | <5% | 48-72 hours | Supportive care |
| Fungal Invasive | Days-weeks | 30-70% | Galactomannan + | Early empiric |
| Parasitic Severe | Hours-days | 10-30% | Blood smear stat | Species-specific |
| Mycobacterial | Weeks-months | 5-15% | AFB + molecular | Multi-drug combo |
💡 Master This: Every infectious disease presentation follows the pathogen-host-environment triad. Pathogen virulence factors determine invasion capacity, host immune status determines resistance level, and environmental factors determine exposure risk. Understanding this triad predicts 90% of clinical presentations.
Connect these foundational patterns through antimicrobial stewardship principles to understand how resistance shapes modern infectious disease practice.
🦠 Infectious Disease Mastery: The Clinical Battlefield
⚔️ Antimicrobial Arsenal: The Precision Strike Force
📌 Remember: CAMP-FIST - Cell wall (β-lactams), Antimetabolites (folate inhibitors), Membrane (polymyxins), Protein synthesis (aminoglycosides, macrolides), Folate synthesis (sulfonamides), Inhibitors (β-lactamase), Synthesis (quinolones), Topoisomerase (fluoroquinolones)
The antimicrobial classification system operates through five mechanistic categories with distinct resistance patterns:
-
Cell Wall Synthesis Inhibitors
- β-lactams: bactericidal against actively dividing organisms
- Penicillins: Gram-positive coverage, MIC ≤0.12 mg/L for susceptible Streptococcus
- Cephalosporins: generation-dependent spectrum expansion
- Carbapenems: broad-spectrum, reserve for ESBL producers
- Vancomycin: Gram-positive only, trough levels 15-20 mg/L for serious infections
- β-lactams: bactericidal against actively dividing organisms
-
Protein Synthesis Inhibitors
- 30S ribosome: aminoglycosides (bactericidal), tetracyclines (bacteriostatic)
- 50S ribosome: macrolides, chloramphenicol, lincosamides
- Concentration-dependent killing: peak/MIC ratio >8-10
- Time-dependent killing: time above MIC >40-50% dosing interval
-
DNA/RNA Synthesis Inhibitors
- Fluoroquinolones: topoisomerase II/IV inhibition
- AUC/MIC ratio >125 for Gram-negatives
- AUC/MIC ratio >30-40 for Gram-positives
- Metronidazole: anaerobic DNA damage, 100% bioavailability
- Fluoroquinolones: topoisomerase II/IV inhibition

📌 Remember: PK-PD-POWER - Peak concentration (aminoglycosides), Kill curves (concentration-dependent), Post-antibiotic effect (fluoroquinolones), Duration above MIC (β-lactams), Penetration (CNS, bone), Optimal dosing (therapeutic monitoring), Waste elimination (renal/hepatic), Efficacy targets (clinical cure), Resistance prevention (mutant selection window)
| Antibiotic Class | PK/PD Parameter | Target Ratio | Dosing Strategy | Resistance Risk |
|---|---|---|---|---|
| β-lactams | Time > MIC | >40-50% | Frequent dosing | Low-moderate |
| Aminoglycosides | Peak/MIC | >8-10 | Once daily | Low |
| Fluoroquinolones | AUC/MIC | >125 | Daily dosing | High |
| Vancomycin | AUC/MIC | >400 | Continuous/q12h | Moderate |
| Macrolides | Time > MIC | >40% | Extended dosing | High |
- Enzymatic inactivation: β-lactamases (>1,000 variants), aminoglycoside-modifying enzymes
- Target modification: PBP alterations (MRSA), ribosomal methylation (macrolide resistance)
- Efflux pumps: multidrug resistance in Pseudomonas, Acinetobacter
- Permeability changes: porin loss in Enterobacteriaceae
⭐ Clinical Pearl: Carbapenem resistance mechanisms include KPC (Klebsiella pneumoniae carbapenemase), NDM (New Delhi metallo-β-lactamase), and OXA (oxacillinase) enzymes. Colistin remains active against >90% of carbapenem-resistant Enterobacteriaceae but requires loading dose 9 million units followed by 4.5 million units q12h.
💡 Master This: Antimicrobial stewardship reduces resistance development by 20-30%, decreases C. difficile infections by 50%, and improves clinical outcomes while reducing costs by 15-25%. The "right drug, right dose, right duration" principle prevents collateral damage to normal flora.
Connect antimicrobial principles through sepsis recognition patterns to understand how rapid pathogen identification drives therapeutic success.
⚔️ Antimicrobial Arsenal: The Precision Strike Force
🎯 Sepsis Recognition: The Critical Hour Protocol
📌 Remember: SOFA-QUICK - Systemic organ failure assessment, Oxygen (PaO2/FiO2 <400), Factor coagulation (platelets <150,000), Alteration mental (GCS <15), Quick assessment (qSOFA ≥2), Urine output (<0.5 mL/kg/h), Inotropes required (MAP <65), Creatinine elevated (>1.2 mg/dL), Kidney dysfunction (doubling baseline)
The sepsis recognition cascade operates through three progressive severity levels with distinct mortality implications:
-
SIRS (Systemic Inflammatory Response Syndrome)
- ≥2 criteria: Temperature >38°C or <36°C, HR >90 bpm, RR >20/min, WBC >12,000 or <4,000/μL
- Sensitivity 95% but specificity only 10% for infection
- Positive predictive value <5% in general population
-
Sepsis (Sepsis-3 Definition)
- SOFA score ≥2 from baseline representing organ dysfunction
- Hospital mortality 10% with SOFA 2-6 points
- ICU mortality 25% with SOFA 7-12 points
- Respiratory: PaO2/FiO2 ratio scoring system
- Coagulation: platelet count thresholds
- Hepatic: bilirubin levels (>1.2 mg/dL = 1 point)
- Cardiovascular: MAP requirements and vasopressor needs
- Neurologic: Glasgow Coma Scale assessment
- Renal: creatinine or urine output criteria
-
Septic Shock (Sepsis-3 Definition)
- Vasopressor requirement to maintain MAP ≥65 mmHg
- Lactate >2 mmol/L despite adequate fluid resuscitation
- Hospital mortality >40% with early recognition critical
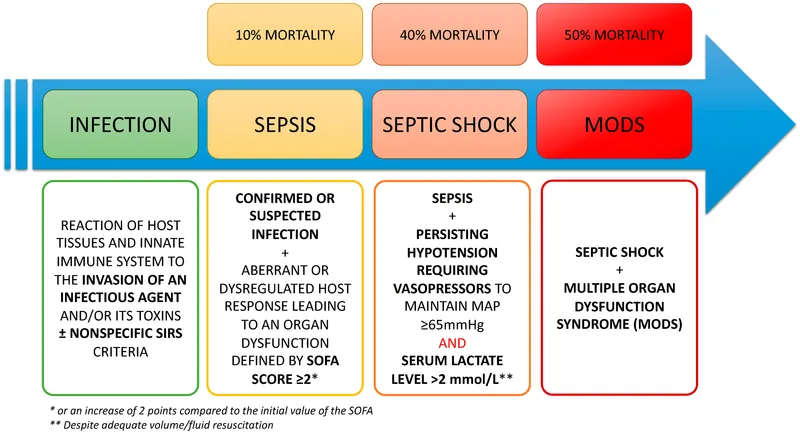
📌 Remember: LACTATE-MAP - Lactate >2 mmol/L (shock marker), Adequate fluids (30 mL/kg crystalloid), Cultures before antibiotics (blood, urine, sputum), Time-sensitive (1-hour bundle), Antibiotics broad-spectrum, Target MAP ≥65 mmHg, Evaluate source control, Monitoring serial lactate, Assess fluid responsiveness, Pressors if needed
| Sepsis Severity | SOFA Score | Mortality Risk | Key Interventions | Time Targets |
|---|---|---|---|---|
| Sepsis | 2-6 points | 10-15% | Antibiotics + cultures | 1 hour |
| Severe Sepsis | 7-12 points | 25-35% | Fluid resuscitation | 3 hours |
| Septic Shock | >12 points | 40-60% | Vasopressors + lactate | 6 hours |
| Refractory Shock | >15 points | >80% | ECMO consideration | 24 hours |
- Blood cultures before antibiotics (≥2 sets, ≥20 mL total volume)
- Lactate measurement (arterial or venous acceptable)
- Broad-spectrum antibiotics within 1 hour of recognition
- Crystalloid fluid 30 mL/kg for hypotension or lactate ≥4 mmol/L
- Source Control Priorities
- Drainage of infected fluid collections (<12 hours optimal)
- Debridement of necrotic tissue (necrotizing fasciitis emergency)
- Device removal (infected catheters, prosthetic materials)
- Definitive repair of anatomical disruption
⭐ Clinical Pearl: qSOFA (altered mental status, SBP ≤100 mmHg, RR ≥22/min) identifies high-risk patients outside ICU but misses 70% of sepsis cases. SOFA score remains gold standard for organ dysfunction assessment with each point representing 10% mortality increase.
💡 Master This: Early goal-directed therapy principles focus on hemodynamic optimization within 6 hours: CVP 8-12 mmHg, MAP ≥65 mmHg, urine output ≥0.5 mL/kg/h, ScvO2 ≥70%. However, fluid overload increases mortality, requiring dynamic assessment of fluid responsiveness.
Connect sepsis recognition through resistance pattern analysis to understand how pathogen identification guides targeted therapy.
🎯 Sepsis Recognition: The Critical Hour Protocol
🔬 Resistance Patterns: The Evolutionary Arms Race
📌 Remember: ESCAPE-FIRE - Enterococcus (VRE), Staphylococcus (MRSA), Clostridium (C. diff), Acinetobacter (MDR), Pseudomonas (XDR), Enterobacteriaceae (CRE), Fungal (azole-resistant), Influenza (oseltamivir), Respiratory (macrolide), Emerging (colistin)
The resistance evolution operates through four primary mechanisms with distinct clinical implications:
-
Enzymatic Inactivation Mechanisms
- β-lactamases: >1,000 variants identified globally
- Class A: TEM, SHV, KPC (serine-based)
- Class B: NDM, VIM, IMP (metallo-β-lactamases)
- Class C: AmpC (chromosomal, plasmid-mediated)
- Class D: OXA (oxacillinases, carbapenem-hydrolyzing)
- ESBL prevalence: 15-25% E. coli, 20-40% K. pneumoniae globally
- Carbapenemase producers: <5% but doubling every 2-3 years
- β-lactamases: >1,000 variants identified globally
-
Target Site Modifications
- MRSA: mecA gene encoding PBP2a (penicillin-binding protein)
- Healthcare-associated: clonal complex 5/8
- Community-associated: clonal complex 1/8 with PVL toxin
- Vancomycin resistance: vanA (high-level, MIC >32 mg/L), vanB (variable)
- Fluoroquinolone resistance: gyrA/parC mutations in >80% resistant isolates
- MRSA: mecA gene encoding PBP2a (penicillin-binding protein)
-
Efflux Pump Overexpression
- Pseudomonas aeruginosa: MexAB-OprM, MexXY, MexEF-OprN systems
- Acinetobacter baumannii: AdeABC, AdeIJK multidrug efflux
- Enterobacteriaceae: AcrAB-TolC system affecting multiple drug classes

📌 Remember: CRE-ALERT - Carbapenem-resistant Enterobacteriaceae, Rapid detection (<4 hours), Epidemiologic investigation, Alert laboratory, Limit transmission (contact precautions), Empire therapy (colistin + tigecycline), Reporting mandatory, Testing (carbapenemase genes)
| Resistance Pattern | Prevalence | Key Mechanisms | Therapeutic Options | Mortality Impact |
|---|---|---|---|---|
| MRSA | 15-50% | mecA gene | Vancomycin, linezolid | +15-20% |
| ESBL | 15-25% | TEM, SHV, CTX-M | Carbapenems | +10-15% |
| CRE | <5% | KPC, NDM, OXA | Colistin, tigecycline | +30-50% |
| VRE | 5-15% | vanA, vanB | Linezolid, daptomycin | +20-25% |
| MDR-TB | 3-5% | katG, rpoB | Second-line drugs | +40-60% |
- ESBL producers: co-resistance to fluoroquinolones (70-80%), aminoglycosides (60-70%)
- Carbapenemase producers: pan-resistance except colistin (90-95% susceptible)
- High-level aminoglycoside resistance: synergy loss with cell wall agents
- Emerging Resistance Threats
- Colistin resistance: mcr-1 gene (plasmid-mediated)
- Ceftazidime-avibactam resistance: KPC variants (blaKPC-3)
- Azole-resistant Aspergillus: TR34/L98H mutations (environmental selection)
⭐ Clinical Pearl: Carbapenem-sparing strategies for ESBL producers include β-lactam/β-lactamase inhibitor combinations: piperacillin-tazobactam (MIC ≤16 mg/L), ceftolozane-tazobactam, ceftazidime-avibactam. Ertapenem preferred over meropenem/imipenem to preserve anti-Pseudomonas activity.
💡 Master This: Antimicrobial stewardship reduces resistance development through de-escalation (48-72 hours), optimal dosing (PK/PD targets), duration optimization (biomarker-guided), and combination therapy for high-resistance risk pathogens. Heteroresistance requires higher dosing to prevent resistance emergence.
Connect resistance patterns through diagnostic innovation to understand how rapid pathogen identification enables targeted therapy.
🔬 Resistance Patterns: The Evolutionary Arms Race
🧬 Diagnostic Revolution: Molecular Medicine Mastery
📌 Remember: RAPID-DX - Real-time PCR (<4 hours), Antigen detection (<30 minutes), Point-of-care testing (bedside), Identification molecular (16S rRNA), Drug resistance genes (mecA, vanA), Direct specimen testing (blood, CSF), Xpert platforms (GeneXpert)
The diagnostic technology landscape operates through five revolutionary platforms with distinct clinical applications:
-
Molecular Amplification Technologies
- Real-time PCR: sensitivity >95%, specificity >98% for most pathogens
- Multiplex panels: 15-25 targets simultaneously
- Blood culture-independent: direct specimen testing
- Turnaround time: 1-6 hours vs 24-72 hours culture
- Loop-mediated isothermal amplification (LAMP): point-of-care capability
- CRISPR-based diagnostics: emerging technology with single-molecule sensitivity
- Real-time PCR: sensitivity >95%, specificity >98% for most pathogens
-
Mass Spectrometry Identification
- MALDI-TOF MS: matrix-assisted laser desorption/ionization
- Identification accuracy >95% for common bacteria
- Cost per test <$1 after initial investment
- Turnaround time <30 minutes from positive culture
- Direct specimen testing: limited sensitivity (requires >10^5 CFU/mL)
- Resistance prediction: emerging applications for β-lactamase detection
- MALDI-TOF MS: matrix-assisted laser desorption/ionization
-
Next-Generation Sequencing (NGS)
- Metagenomic sequencing: unbiased pathogen detection
- Sensitivity for rare pathogens >90%
- Turnaround time 24-48 hours
- Cost $200-500 per test
- Whole genome sequencing: outbreak investigation, resistance profiling
- 16S rRNA sequencing: bacterial identification from culture-negative specimens
- Metagenomic sequencing: unbiased pathogen detection
📌 Remember: BIOMARKER-GUIDE - Biomarkers (PCT, CRP), Inflammation markers (IL-6), Organ dysfunction (lactate), Monitoring response (serial levels), Antibiotic duration (PCT-guided), Risk stratification (SOFA), Kinetics (half-life), Early detection (trending), Range normal (reference values), Guidance therapy (algorithms), Utility clinical (decision-making), Interpretation (context-dependent), Discontinuation (stopping rules), Evidence-based (RCT data)
| Diagnostic Platform | Turnaround Time | Sensitivity | Specificity | Cost per Test | Clinical Application |
|---|---|---|---|---|---|
| Blood Culture | 24-72 hours | >95% | >98% | $20-30 | Gold standard |
| PCR Multiplex | 1-6 hours | >90% | >95% | $100-200 | Rapid identification |
| MALDI-TOF | <30 minutes | >95% | >98% | <$1 | Culture identification |
| Antigen Tests | <30 minutes | 70-90% | >95% | $10-50 | Point-of-care |
| NGS Metagenomic | 24-48 hours | >90% | >85% | $200-500 | Complex cases |
- Procalcitonin (PCT): bacterial infection marker
- <0.25 ng/mL: low bacterial probability (<10%)
- 0.25-0.5 ng/mL: possible bacterial (10-25%)
- >0.5 ng/mL: likely bacterial (>75%)
- >2.0 ng/mL: severe bacterial sepsis (>90%)
- C-reactive protein (CRP): inflammation marker
- <10 mg/L: low inflammation
- 10-50 mg/L: moderate inflammation
- >50 mg/L: severe inflammation
- Lactate: tissue hypoperfusion marker
- <2 mmol/L: normal perfusion
- 2-4 mmol/L: mild hypoperfusion
- >4 mmol/L: severe shock
- Resistance Gene Detection
- mecA/mecC: MRSA identification (sensitivity >99%)
- vanA/vanB: VRE detection (specificity >98%)
- Carbapenemase genes: KPC, NDM, OXA-48 (multiplex assays)
- ESBL genes: CTX-M, TEM, SHV (epidemiologic typing)
⭐ Clinical Pearl: PCT-guided antibiotic therapy reduces antibiotic duration by 2-3 days without increasing mortality. Stopping rule: PCT decrease >80% from peak or absolute level <0.25 ng/mL suggests bacterial clearance. Serial monitoring every 24-48 hours optimizes decision-making.
💡 Master This: Diagnostic stewardship principles include pre-test probability assessment, appropriate test selection, result interpretation in clinical context, and post-test management decisions. False-positive results from high-sensitivity tests require clinical correlation to avoid unnecessary treatment.
Connect diagnostic capabilities through treatment optimization strategies to understand how personalized therapy improves outcomes.
🧬 Diagnostic Revolution: Molecular Medicine Mastery
🎯 Treatment Optimization: The Precision Medicine Protocol

📌 Remember: OPTIMIZE-RX - Optimal dosing (PK/PD targets), Personalized therapy (patient factors), Therapeutic monitoring (drug levels), Infection source control, Monitoring response (biomarkers), Interaction assessment (drug-drug), Zero resistance (combination therapy), Early de-escalation, Renal adjustment (CrCl), Xtra considerations (pregnancy, obesity)
The treatment optimization framework operates through six integrated components with measurable clinical endpoints:
-
Pharmacokinetic/Pharmacodynamic Optimization
- Time-dependent killing (β-lactams): time above MIC >40-50%
- Continuous infusion: meropenem, piperacillin-tazobactam
- Extended infusion: 3-4 hour infusions for critical infections
- Frequent dosing: q6h instead of q8h for severe infections
- Concentration-dependent killing (aminoglycosides, fluoroquinolones)
- Peak/MIC ratio >8-10 for aminoglycosides
- AUC/MIC ratio >125 for fluoroquinolones vs Gram-negatives
- AUC-dependent efficacy (vancomycin, linezolid)
- Vancomycin AUC/MIC >400 for serious MRSA infections
- Trough levels 15-20 mg/L for pneumonia, endocarditis
- Time-dependent killing (β-lactams): time above MIC >40-50%
-
Therapeutic Drug Monitoring (TDM)
- Vancomycin: AUC-based dosing preferred over trough-only
- Target AUC 400-600 mg·h/L for most infections
- Bayesian software for AUC estimation
- Nephrotoxicity risk with AUC >600 or trough >20 mg/L
- Aminoglycosides: once-daily dosing with extended-interval monitoring
- Hartford nomogram for dosing adjustment
- Peak levels 5-10× MIC, trough <1-2 mg/L
- β-lactams: emerging TDM for critically ill patients
- Target free drug concentration >4× MIC
- Continuous infusion monitoring
- Vancomycin: AUC-based dosing preferred over trough-only
📌 Remember: COMBO-POWER - Combination synergy (β-lactam + aminoglycoside), Optimal timing (simultaneous start), Mechanism different (dual targets), Biofilm penetration (enhanced), Outcome improved (mortality reduction), Prevention resistance (mutant selection), Organ penetration (CNS, bone), Widened spectrum (empiric coverage), Early bactericidal (rapid killing), Resistance barrier (high genetic)
| Treatment Strategy | Clinical Indication | Outcome Benefit | Monitoring Required | Duration Optimization |
|---|---|---|---|---|
| Combination Therapy | Severe sepsis | Mortality ↓15-20% | Synergy testing | 3-5 days |
| Extended Infusion | Critical illness | Clinical cure ↑10-15% | Drug levels | Standard duration |
| High-Dose Therapy | CNS infections | Penetration ↑2-3× | Toxicity monitoring | Pathogen-dependent |
| Oral Switch | Clinical stability | Cost ↓50-70% | Bioavailability | Biomarker-guided |
| Outpatient IV | Complex infections | LOS ↓3-5 days | Safety protocols | Weekly assessment |
- Timing critical: <12 hours for optimal outcomes
- Drainage procedures: percutaneous vs surgical approach
- Device removal: infected prosthetics, catheters
- Debridement: necrotizing infections (emergency surgery)
-
Duration Optimization Strategies
- Biomarker-guided: PCT decrease >80% or <0.25 ng/mL
- Clinical stability: fever resolution, WBC normalization
- Pathogen-specific: S. aureus bacteremia 2-6 weeks, Gram-negative bacteremia 7-14 days
- Site-specific: pneumonia 5-7 days, UTI 3-7 days, skin/soft tissue 5-10 days
-
Resistance Prevention Protocols
- Combination therapy for high-resistance risk (P. aeruginosa, Acinetobacter)
- Optimal dosing to exceed mutant prevention concentration
- De-escalation within 48-72 hours based on culture results
- Cycling strategies for ICU empiric therapy
⭐ Clinical Pearl: Combination therapy for Gram-negative bacteremia reduces mortality by 15-20% when started within 24 hours. β-lactam plus aminoglycoside or fluoroquinolone shows synergy against >80% of Enterobacteriaceae. Duration 3-5 days prevents resistance while maintaining efficacy.
💡 Master This: Precision dosing using population pharmacokinetics and Bayesian estimation improves target attainment by 30-40% compared to standard dosing. Model-informed precision dosing (MIPD) platforms integrate patient covariates (weight, renal function, albumin) with real-time drug levels for optimal therapy.
Understanding these treatment optimization principles provides the foundation for mastering infectious disease management across all clinical scenarios, from routine infections to complex multidrug-resistant pathogens requiring innovative therapeutic approaches.
🎯 Treatment Optimization: The Precision Medicine Protocol
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app






















