LA Basics - Nerve Numbeners
- Mechanism: Reversibly block voltage-gated Na+ channels (VGSCs) → ↓ Na+ influx → prevent action potential generation & conduction.
- Site of Action: Intracellular side of Na+ channel.
- Active Form: Ionized (cationic) form blocks channel; Unionized (uncharged base) form penetrates nerve sheath.
- LAs are weak bases (pKa 7.5-9.0).
- Infected tissue (acidic pH) → ↑ ionized form → ↓ efficacy.
- Order of Blockade (Sensitivity):
- Small, myelinated (B, Aδ - pain, temp) > Small, unmyelinated (C - pain) > Large, myelinated (Aγ, Aβ, Aα - proprioception, touch, motor).
- 📌 Mnemonic: Before All Clinical Manifestations (B > Aδ > Aγ > Aβ > Aα > C - note: C fiber sensitivity varies)
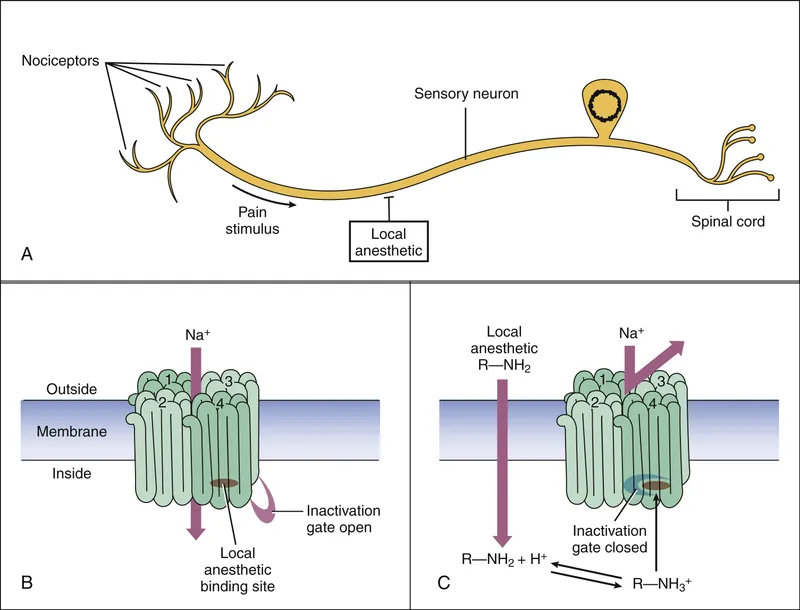
⭐ LAs exhibit use-dependent blockade: higher frequency of nerve stimulation leads to greater blockade because channels are more often in open/inactivated states, which LAs bind with higher affinity than resting state. This is also known as phasic block.
Structure & Types - Chemical Coats
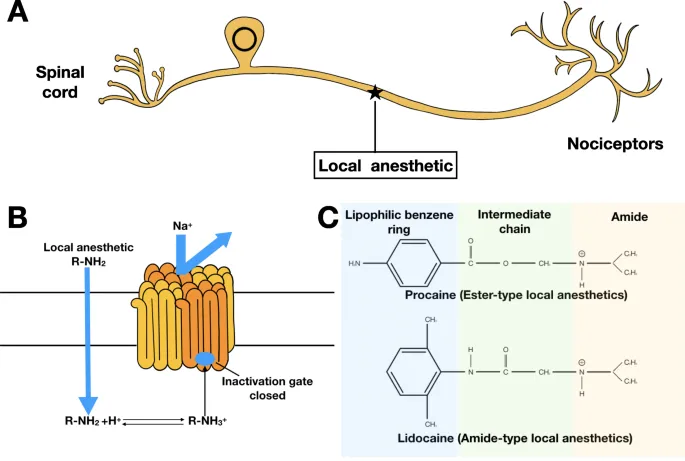
- LA Structure:
- Lipophilic (aromatic) head: ↑Potency, lipid solubility.
- Intermediate chain: Ester/Amide linkage; determines metabolism, allergy.
- Hydrophilic (amine) tail: Ionization, Na+ channel binding.
- Linkage Types:
- Esters ($-COO-$):
- Plasma pseudocholinesterase metabolism.
- ↑ Allergy risk (PABA metabolite).
- E.g., Procaine, Tetracaine, Benzocaine.
- 📌 Esters: PABA → Allergy.
- Amides ($-NHCO-$):
- Hepatic enzyme (CYP450) metabolism.
- ↓ Allergy risk.
- E.g., Lidocaine, Bupivacaine.
- 📌 Amides: two 'i's in name.
- Esters ($-COO-$):
⭐ Cocaine is the only naturally occurring LA; unique vasoconstrictor (inhibits norepinephrine reuptake).
LA Kinetics - Journey Through Body
- Absorption (A):
- Site vascularity dictates speed (e.g., Intercostal > Epidural > Brachial > SubQ).
- Vasoconstrictors (e.g., adrenaline): ↓ absorption, ↑ duration, ↓ toxicity.
- Distribution (D):
- Vessel-rich groups (brain, heart, liver, kidney) first.
- Bound to α1-acid glycoprotein (AAG); free drug is active.
- Metabolism (M):
- Esters (1 'i'): Plasma pseudocholinesterase (fast; PABA → allergy).
- Amides (2 'i's): Liver CYP450 (slow).
- Prilocaine → o-toluidine → Met-Hb (Rx: Methylene Blue).
- Excretion (E):
- Renal (metabolites).
⭐ Ion trapping: LAs (weak bases) accumulate in acidic fetal plasma or inflamed tissue, ↑ local concentration.
Clinical Applications - Numbing Targets
- Topical Anesthesia: Mucous membranes (cornea, oral cavity, GU tract).
- Infiltration Anesthesia: Direct tissue injection for minor procedures; numbs local sensory nerve endings.
- Nerve Blocks: Injection near specific nerves/plexuses.
- Dental procedures (e.g., Inferior alveolar nerve).
- Limb surgery (e.g., Brachial plexus, Femoral nerve).
- Spinal (Intrathecal) Anesthesia: Into CSF (subarachnoid space); targets nerve roots for lower abdominal/limb surgery.
- Epidural Anesthesia: Into epidural space; targets nerve roots (e.g., labor analgesia, postoperative pain).
- Intravenous Regional Anesthesia (Bier Block): IV into exsanguinated, tourniquet-isolated limb.
⭐ Spinal anesthesia is typically administered into the subarachnoid space at the L3-L4 or L4-L5 intervertebral level in adults to avoid puncturing the spinal cord (conus medullaris usually ends at L1-L2).
Toxicity & Management - Danger Zones
- Systemic Toxicity (LAST): Dose-dependent.
- CNS: Early (circumoral numbness, tinnitus, metallic taste, lightheadedness). Late (muscle twitching, tremors, generalized seizures, unconsciousness, coma, respiratory arrest).
- CVS: Initially hypertension/tachycardia, then ↓BP, bradycardia, arrhythmias (esp. Bupivacaine - ventricular), asystole.
- Allergic Reactions: Rare with amides; esters (PABA metabolite) more common.
- Methemoglobinemia: Prilocaine, Benzocaine. Treat with Methylene Blue 1-2 mg/kg IV.
- Danger Zones (Systemic Absorption Rate): 📌 Intercostal > Caudal > Epidural > Brachial Plexus > Sciatic/Femoral > Subcutaneous ("ICE-BS").
⭐ Bupivacaine: high cardiotoxicity. For severe LAST: IV Lipid Emulsion (20%) is key. Bolus 1.5 mL/kg, then 0.25 mL/kg/min infusion.
High‑Yield Points - ⚡ Biggest Takeaways
- Mechanism: Block voltage-gated Na+ channels, preventing action potentials.
- Sensitivity: Small, myelinated fibers (pain, temp) blocked first; sensory > motor.
- Inflamed (acidic) tissue ↓ efficacy due to ↑ ionization.
- Toxicity: CNS effects (seizures) usually precede cardiac toxicity. Bupivacaine is highly cardiotoxic.
- Metabolism: Amides (two 'i's - Lidocaine) in liver; Esters (one 'i' - Procaine) by plasma pseudocholinesterase (PABA allergy).
- Vasoconstrictors (epinephrine) prolong action, ↓ systemic toxicity.
- Cocaine: Ester LA with intrinsic vasoconstrictor activity (NET blockade).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

