Media Fundamentals - Broth & Beyond
-
Core Constituents:
- Peptone: Source of nitrogen, amino acids.
- Meat/Yeast Extract: Vitamins, growth factors.
- NaCl: Maintains osmotic balance.
- Water: Solvent.
- Agar: Solidifying agent (seaweed polysaccharide).
-
Physical States (Consistency):
- Liquid (Broth): No agar. Uses: Blood culture, inoculum preparation.
- Semi-solid: 0.2-0.5% agar. Uses: Motility testing, microaerophilic culture.
- Solid: ~2% agar. Uses: Colony isolation, obtaining pure culture.
-
Sterilization Methods for Media:
| Method | Temperature/Details | Common Media Examples |
|---|---|---|
| Autoclave | 121°C, 15 psi, 15-20 min | Most routine media (Nutrient Agar/Broth) |
| Filtration | Membrane filter (0.22 µm or 0.45 µm) | Heat-labile solutions (sera, antibiotics, sugars) |
| Inspissation | 80-85°C, 30 min, 3 successive days | Egg-based media (Lowenstein-Jensen, Dorset Egg) |
| Tyndallization | 100°C (steam), 20-30 min, 3 successive days | Gelatin media, some sugar media |
Media Types - Picky Eaters' Paradise

| Media Type | Purpose | Key Ingredients/Inhibitors/Indicators | Examples (Organisms) |
|---|---|---|---|
| Basal | Non-fastidious growth | Peptone, meat extract, NaCl | Nutrient Agar (Staph., non-fast. Enterobacteriaceae) |
| Enriched | Fastidious growth | Basal + Blood, serum, egg | Blood Agar (Strep.), Choc. Agar (H. influ., Neisseria), Loeffler's (C. diph.) |
| Selective | Favors specific MOs, inhibits others | Dyes, bile, antibiotics, ↑salt | MacConkey (Gram-neg), LJ (MTB), TCBS (Vibrio), MSA (S. aureus) |
| Differential | Distinguishes MOs by biochem rxns | Indicators, fermentable carbs | MacConkey (LF/NLF), BA (hemolysis), CLED (LF), TSI |
| Transport | Maintains viability, no overgrowth | Buffers, reducers, charcoal | Cary-Blair (Shigella), Stuart's, Amies (Neisseria) |
| Anaerobic | Obligate anaerobe growth | Reducing agents, hemin, Vit K | RCM Broth (Clostr.), Thioglycollate, Anaerobic BA (Bacteroides) |
| Enrichment | ↑ desired MOs (liquid) | Specific nutrients/inhibitors | Selenite F (Salmonella, Shigella), APW (Vibrio) |
Culture Techniques - Streaks & Stabs
- Inoculation Methods:
- Streak Plate: Isolates colonies (quadrant method).
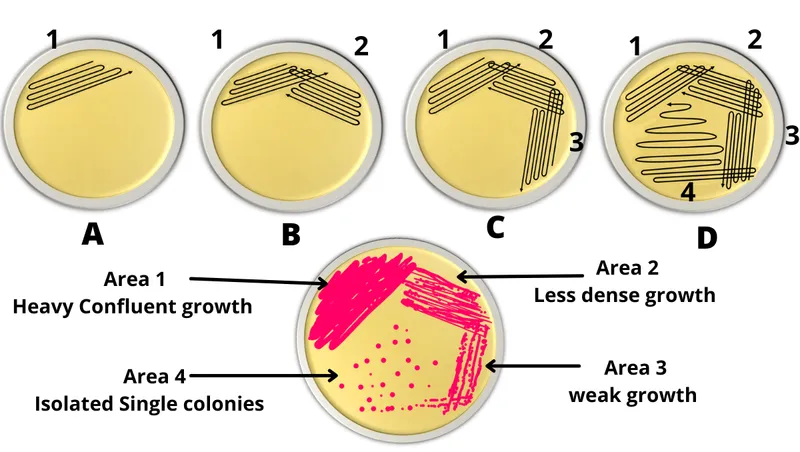
- Lawn Culture: Uniform growth (e.g., antibiotic sensitivity).
- Stab Culture: Motility, O2 needs, enzymes (e.g., TSI).
- Pour Plate: Quantifies bacteria (CFU/mL).
- Liquid Culture (Broth): Enrichment, blood/sterility tests.
- Streak Plate: Isolates colonies (quadrant method).
- Incubation Conditions:
- Temperature: Pathogens 35-37°C; Fungi 25-30°C or 37°C (dimorphic).
- O2: Aerobic, Anaerobic, Facultative, Microaerophilic (low O2).
- CO2: Capnophilic organisms require ↑CO2 (5-10%) (e.g., Neisseria, Haemophilus).
- Colony Morphology (Macroscopic): Size, shape, margin, elevation, color, opacity, consistency, odor.
⭐ The primary goal of the streak plate method is to obtain well-isolated colonies, each arising from a single bacterial cell, allowing for identification and purity assessment.
- Simplified Blood Culture Workflow:
High‑Yield Points - ⚡ Biggest Takeaways
- Blood Agar: Shows hemolysis (α, β, γ); supports fastidious bacteria like Streptococcus.
- MacConkey Agar: Selective (Gram-negatives), differential (lactose fermenters turn pink).
- Chocolate Agar: Lysed blood provides X & V factors for Haemophilus, Neisseria.
- Lowenstein-Jensen (LJ) Medium: For culturing slow-growing Mycobacterium tuberculosis.
- Sabouraud Dextrose Agar (SDA): Acidic pH, for fungal isolation and culture.
- Transport Media (e.g., Cary-Blair): Maintain pathogen viability, prevent overgrowth during specimen transit.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

