Interorgan Metabolite Exchange - Metabolic Symphony
- Organs communicate via circulating metabolites to meet energy demands.
- Liver: "Metabolic Maestro". Exports glucose (gluconeogenesis, glycogenolysis), ketone bodies, VLDL. Processes lactate, alanine, glycerol.
- Skeletal Muscle: Uses glucose, fatty acids, ketone bodies. Stores glycogen. Exports lactate (Cori cycle), alanine (glucose-alanine cycle).
- Adipose Tissue: Stores TAGs. Releases fatty acids & glycerol during fasting.
- Brain: Obligate glucose user (normally ~120g/day); adapts to ketone bodies in prolonged starvation.
- Kidney: Gluconeogenesis (e.g., from glutamine) during prolonged fasting/acidosis. Excretes nitrogenous waste.
- Intestine: Uses glutamine as primary fuel. Exports citrulline.
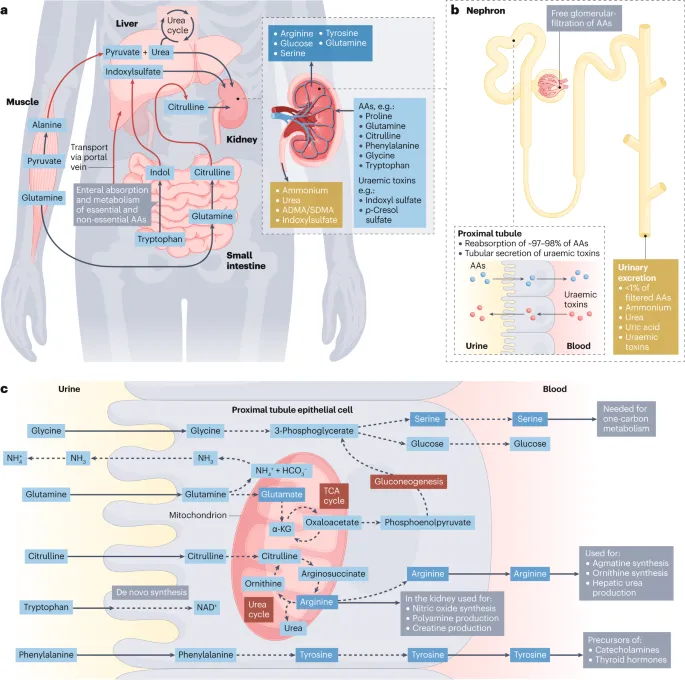
⭐ During prolonged starvation, the kidney can contribute up to 50% of glucose production via gluconeogenesis.
Fed State Exchange - Postprandial Powwow
- Hormones: ↑ Insulin, ↓ Glucagon.
- Goal: Anabolism (storage of glucose, AAs, fats).
- 📌 Insulin: "INto cells" for glucose (muscle/adipose).
- Gut: Absorbs glucose, AAs (portal vein → Liver); CMs (lymph → blood).
- Liver: Central hub.
- Glucose → Glycogen; excess → FAs → VLDL.
- AAs → Protein synthesis; BCAAs → Muscle.
- CM remnants cleared; FAs → VLDL.
- Muscle:
- Glucose (GLUT4, insulin) → Glycogen, ATP.
- BCAAs → Protein synthesis.
- Adipose:
- Glucose (GLUT4, insulin) → Glycerol-3-P.
- FAs (CMs, VLDL via LPL) + Glycerol-3-P → TAG storage.
- Brain/RBCs: Glucose uptake (insulin-independent).
⭐ > Insulin is key: promotes glucose uptake in muscle & adipose tissue by translocating GLUT4 transporters to the cell membrane.
Fasting State Exchange - Famine Fortitude
Glucagon ↑, Insulin ↓. Goal: Maintain blood glucose for brain & RBCs; spare protein.
-
Initial Phase (Post-absorptive to ~24h)
- Liver: Glycogenolysis (main glucose source, depletes by ~18-24h). Gluconeogenesis (GNG) begins (lactate, alanine, glycerol).
- Adipose: Lipolysis ↑ (Hormone Sensitive Lipase - HSL) → FFAs (energy for muscle/liver) + Glycerol (GNG substrate).
- Muscle: Protein breakdown → Alanine, Glutamine (GNG substrates).
-
Prolonged Phase (Starvation >24-48h)
- Liver: GNG (primary glucose source). Ketogenesis ↑↑ (from FFAs) → $\beta$-hydroxybutyrate, acetoacetate.
- Adipose: Sustained lipolysis.
- Muscle: ↓ Glucose uptake. Uses FFAs, ketone bodies. Protein catabolism ↓ (sparing).
- Brain: Adapts to use ketone bodies (up to 70% energy).
- Kidney: Significant GNG (up to 50% of total GNG in late starvation).

⭐ During prolonged starvation, the kidney can contribute up to 50% of total gluconeogenesis, highlighting its crucial role beyond excretion.
Key Metabolic Cycles - Shuttles & Switches
- Cori Cycle (Lactic Acid Cycle):
- Muscle (anaerobic): Glucose $\rightarrow$ Lactate $\rightarrow$ Liver.
- Liver: Lactate $\rightarrow$ Glucose (gluconeogenesis) $\rightarrow$ Muscle.
- Function: Sustains muscle glycolysis; prevents acidosis. Net cost: 4 ATP.
- Glucose-Alanine Cycle (Cahill Cycle):
- Muscle: Glucose $\rightarrow$ Pyruvate; Pyruvate + NH2 $\rightarrow$ Alanine $\rightarrow$ Liver.
- Liver: Alanine $\rightarrow$ Pyruvate (gluconeogenesis) + NH2 (urea cycle).
- Function: N-transport from muscle; glucose for muscle.
- Glycerol-3-Phosphate Shuttle:
- Cytosolic NADH e- $\rightarrow$ Mitochondrial FADH2 (to Complex II).
- Yield: ~1.5 ATP / NADH. Sites: Skeletal muscle, brain.
- Malate-Aspartate Shuttle:
- Cytosolic NADH e- $\rightarrow$ Mitochondrial NADH (to Complex I).
- Yield: ~2.5 ATP / NADH. Sites: Liver, heart, kidney.
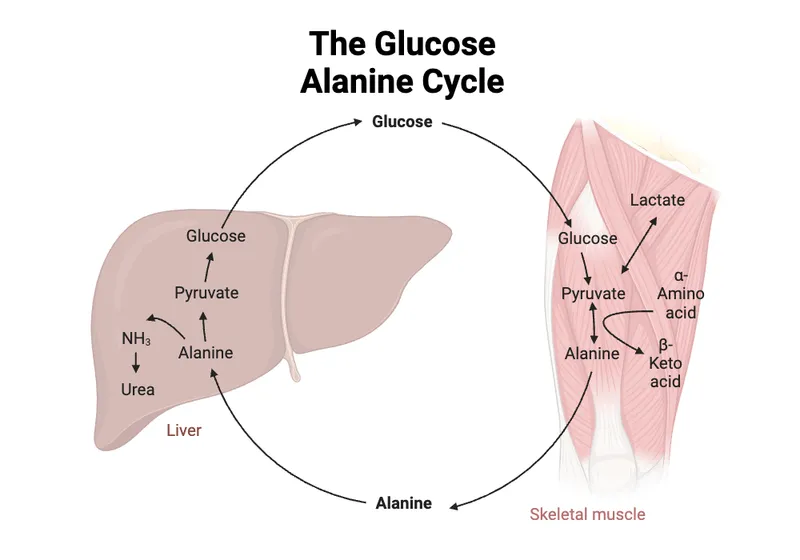
⭐ The Cori Cycle recycles lactate from anaerobic muscle glycolysis to glucose in the liver, costing a net 4 ATP, thus shifting metabolic burden.
High‑Yield Points - ⚡ Biggest Takeaways
- Liver: Central metabolic hub for gluconeogenesis, urea cycle, ketogenesis.
- Muscle: Uses glucose, fatty acids, ketones; releases alanine, lactate.
- Brain: Primarily glucose-dependent; adapts to ketone bodies in starvation.
- Adipose Tissue: Stores triglycerides; mobilizes fatty acids, glycerol.
- Kidney: Contributes to gluconeogenesis (prolonged fast); ammonia excretion.
- RBCs: Solely glucose-dependent (anaerobic); produce lactate for Cori cycle.
- Glucose-Alanine cycle: Key for nitrogen transport from muscle to liver.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

