Iron Absorption and Transport - Rusty Essentials
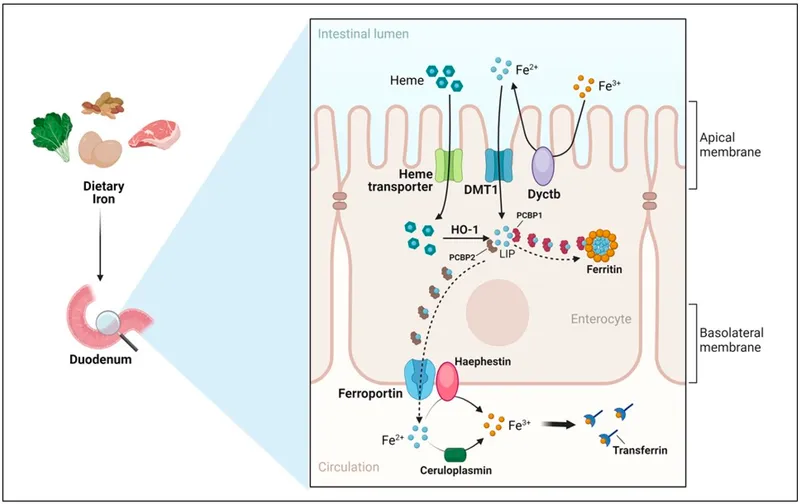
- Absorption: Duodenum (Fe²⁺ form). Non-heme Fe³⁺ reduced by DcytB.
- Enterocyte Uptake: Heme (HCP1); Fe²⁺ (DMT1).
- Basolateral Export: Ferroportin (Fe²⁺). 📌 Ferroportin = For Passage Out.
- Oxidation: Hephaestin (enterocyte) / Ceruloplasmin (plasma) convert Fe²⁺ → Fe³⁺.
- Blood Transport: Transferrin carries Fe³⁺.
⭐ Hepcidin, a liver hormone, is the main negative regulator, blocking iron export by degrading ferroportin.

Iron Absorption and Transport - The Gut Gate
- Dietary Iron:
- Heme (meat): Absorbed by HCP1 (Heme Carrier Protein 1).
- Non-heme ($Fe^{3+}$, plants): Needs reduction to $Fe^{2+}$ (ferrous).
- Duodenal Enterocyte Uptake (Apical):
- Non-heme $Fe^{3+}$ $\xrightarrow{DcytB \ (reductase)}$ $Fe^{2+}$.
- $Fe^{2+}$ enters via DMT1 (Divalent Metal Transporter 1).
- Heme enters via HCP1; Heme Oxygenase releases $Fe^{2+}$ intracellularly.
- Intracellular $Fe^{2+}$ & Basolateral Export:
- Storage: As ferritin (stores $Fe^{3+}$).
- Export: $Fe^{2+}$ via ferroportin (FPN1).
- $Fe^{2+}$ $\xrightarrow{Hephaestin \ (Cu-ferroxidase)}$ $Fe^{3+}$.
- $Fe^{3+}$ binds plasma transferrin.
⭐ DMT1 is the key apical transporter for non-heme ferrous iron; ferric iron is not its substrate.
Iron Absorption and Transport - Bloodstream Bound
- Basolateral Export from Enterocyte:
- $Fe^{2+}$ exits via Ferroportin (FPN1).
- Hephaestin: Membrane-bound ferroxidase (Cu$^{2+}$-dependent); oxidizes $Fe^{2+}$ to $Fe^{3+}$ at basolateral membrane for plasma loading.
- Ceruloplasmin: Plasma ferroxidase (Cu$^{2+}$-dependent); also oxidizes $Fe^{2+}$ to $Fe^{3+}$.
- Plasma Transport (Bound to Transferrin):
- Transferrin (Tf): Main iron transport protein; binds 2 $Fe^{3+}$ ions per molecule.
- Synthesized in liver.
- Delivers iron to cells with Transferrin Receptors (TfR1), especially erythroid precursors.
- TIBC (Total Iron Binding Capacity): Indirect measure of circulating transferrin. ↑ in iron deficiency, ↓ in iron overload/inflammation.
- Transferrin (Tf): Main iron transport protein; binds 2 $Fe^{3+}$ ions per molecule.
⭐ Normal transferrin saturation (Serum Iron / TIBC × 100) is 20-50% (average ~33%). Values <16% suggest iron deficiency.
- Regulation:
- Hepcidin: Key regulator; binds ferroportin, causing its internalization & degradation, thus ↓ iron release into blood from enterocytes and macrophages.
Iron Absorption and Transport - Hepcidin's Command
- Hepcidin: Key peptide hormone from liver; master regulator of systemic iron.
- Mechanism: Binds ferroportin (FPN1) → internalization & degradation of FPN1 → ↓ iron export from enterocytes/macrophages into blood.
- Regulation of Hepcidin Synthesis:
- Upregulation (↑ Hepcidin):
- High iron stores (sensing via HFE, TfR2, HJV)
- Inflammation (IL-6 via STAT3 pathway)
- Downregulation (↓ Hepcidin):
- Iron deficiency
- Hypoxia (HIF-dependent pathways)
- Increased erythropoietic activity (Erythroferrone - ERFE)
- Upregulation (↑ Hepcidin):
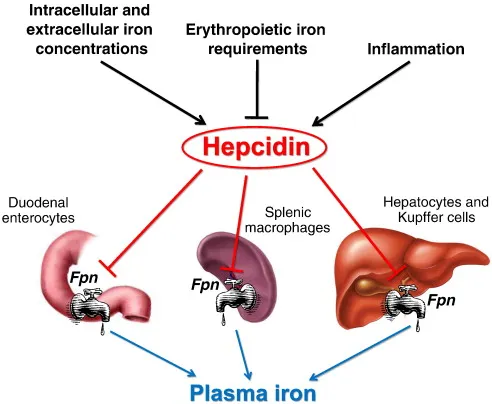
⭐ Hepcidin dysregulation is central to iron disorders: low hepcidin in hemochromatosis, high hepcidin in anemia of chronic disease (ACD).
High‑Yield Points - ⚡ Biggest Takeaways
- Iron absorption predominantly occurs in the duodenum and proximal jejunum.
- Dietary iron: Heme iron (Fe2+) is better absorbed than non-heme iron (Fe3+).
- Apical uptake: DMT1 transports Fe2+; DcytB reduces Fe3+ to Fe2+.
- Basolateral export: Ferroportin transports iron out, aided by hephaestin (oxidation).
- Plasma transport: Transferrin binds and transports Fe3+ in blood.
- Key regulator: Hepcidin inhibits iron absorption by degrading ferroportin.
- Storage: Iron is stored intracellularly as ferritin (Fe3+ form).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

