Light's Dance - Absorbance Rules
- Spectrophotometry & Colorimetry: Techniques measuring substance concentration based on light absorption/transmission.
- Spectrophotometry: Uses specific wavelengths.
- Colorimetry: Uses visible light; often for colored solutions.
- Principle: Interaction of light with matter. Light absorbed by a solution is proportional to the concentration of the absorbing substance.
- Beer-Lambert Law: $A = \epsilon c l$
- $A$: Absorbance (no units)
- $\epsilon$: Molar absorptivity (L·mol⁻¹·cm⁻¹)
- $c$: Concentration (mol/L)
- $l$: Path length (cm)
- Absorbance & Transmittance: $A = -\log T$ or $A = \log(1/T)$, where $T$ is transmittance.
- Limitations: Deviations at high concentrations, chemical reactions, stray light, non-monochromatic light.

⭐ Beer-Lambert Law is the cornerstone of quantitative analysis using spectrophotometry, directly relating absorbance to concentration.
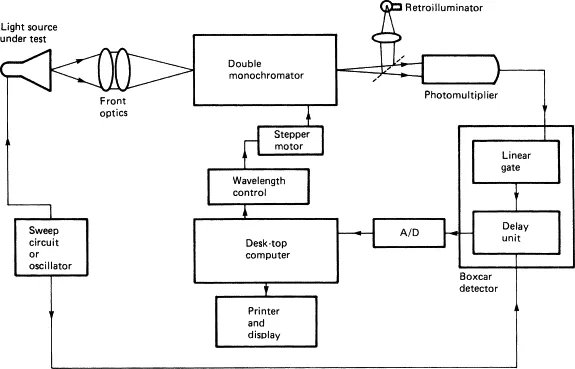
The Machine - Peeking Inside
- Spectrophotometer Components:
- Light Source: UV (Deuterium lamp), Visible (Tungsten lamp).
- Monochromator: Prism or Grating; selects specific wavelength ($\lambda$).
- Sample Holder: Cuvette (path length usually 1 cm).
- Detector: Photomultiplier Tube (PMT) or Photodiode; measures transmitted light intensity.
- Readout Device: Displays absorbance/transmittance.
- Colorimeter Components:
- Light Source: LED or Tungsten lamp.
- Filter: Selects a specific range of wavelengths.
- Sample Holder: Cuvette.
- Detector: Photocell or Photodiode.
- Readout Device.
- Cuvette Types:
- Quartz: For UV light (<340 nm).
- Glass/Plastic: For visible light.
- 📌 Mnemonic: "Quite Useful Glass Visible" (Quartz-UV, Glass-Visible).
- Key Difference: Spectrophotometer uses a monochromator (precise $\lambda$); Colorimeter uses a filter (broader $\lambda$ range).
⭐ Quartz cuvettes are essential for UV spectrophotometry as glass absorbs UV light significantly, especially <340 nm.
Lab Detective - Finding Clues
- Quantitative Analysis (Concentration):
- Proteins: Biuret, Lowry methods.
- Nucleic Acids: Absorbance at $A_{260}$.
- Metabolites: Glucose (GOD-POD), cholesterol.
- Hemoglobin: Cyanmethemoglobin method ($A_{540}$).
- Qualitative Analysis (Identification):
- Absorption spectra identify substances by unique peak absorbance wavelengths.
- Absorption spectra identify substances by unique peak absorbance wavelengths.
- Enzyme Kinetics:
- Measure reaction rates by monitoring changes in absorbance of substrate/product over time.
- Purity Assessment (Nucleic Acids):
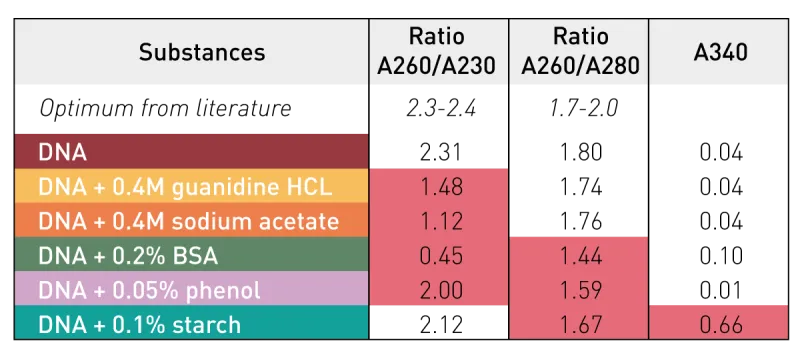
- $A_{260}/A_{280}$ ratio:
- Pure DNA ≈ 1.8.
- Pure RNA ≈ 2.0.
- Ratios < 1.8 (DNA) or < 2.0 (RNA) suggest protein/phenol contamination.
- $A_{260}/A_{280}$ ratio:
⭐ The $A_{260}/A_{280}$ ratio is a widely used, quick method to assess the purity of nucleic acid preparations.
- Clinical Examples:
- Hemoglobin estimation: Drabkin’s (cyanmethemoglobin) method.
- Glucose estimation: GOD-POD (Glucose Oxidase-Peroxidase) method.
Color Power - Simpler & Specific
- Colorimetry:
- Simpler technique; uses filters for broad wavelength bands (visible range).
- Principle: Measures colored solution intensity.
- Follows Beer-Lambert Law: $A = \epsilon c l$.
- Calibration Curve:
- Plot Absorbance vs. known concentrations of standards.
- Essential for quantification.
- Blanks:
- Purpose: Zero instrument; correct for solvent/reagent absorbance.
- Types: Reagent blank, sample blank.
| Feature | Spectrophotometry | Colorimetry |
|---|---|---|
| Wavelength Selector | Monochromator (precise) | Filters (broad bands) |
| Range | UV, Visible, IR | Visible |
| Sensitivity | Higher | Lower |
| Cost | Higher | Lower |
| Complexity | More complex | Simpler |
⭐ Colorimeters use filters to select a range of wavelengths, making them simpler and cheaper but less specific than spectrophotometers which use monochromators.
High‑Yield Points - ⚡ Biggest Takeaways
- Beer-Lambert Law governs: Absorbance proportional to concentration & path length.
- Spectrophotometry measures substance concentration via light absorbance/transmittance at specific wavelengths.
- Colorimetry is a spectrophotometry type limited to the visible spectrum.
- Cuvette choice crucial: quartz for UV, glass/plastic for visible light.
- A blank solution is essential for accuracy, correcting background absorbance.
- Key uses: Quantifying DNA, RNA, proteins; monitoring enzyme kinetics.
- High concentrations can cause deviation from Beer-Lambert's linearity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

