Foundations of Genetic Disease - Gene Glitches 101
- Mutation: Permanent DNA alteration; frequency <1%.
- Polymorphism: Common DNA variation; frequency >1%.
- Penetrance: Proportion of individuals with a specific genotype who express the expected phenotype. All-or-none.
- Expressivity: Variation in phenotypic expression (severity/type) among individuals with the same genotype. Variable.
- Classifications:
- Monogenic: Single gene defect (e.g., Cystic Fibrosis).
- Polygenic: Multiple genes + environment (e.g., Type 2 Diabetes).
- Chromosomal: Numerical/structural anomalies (e.g., Trisomy 21).
- Mitochondrial: Maternally inherited (e.g., LHON).

⭐ Pleiotropy refers to a single gene influencing multiple, often seemingly unrelated, phenotypic traits (e.g., Marfan syndrome).
Mutation Mayhem - DNA's Dark Side
- Point Mutations (Single Base Change):
- Silent: Different codon, same amino acid (AA).
- Missense: Different codon, different AA (e.g., Sickle cell: HbS, $GAG \rightarrow GTG$, Glu→Val).
- Nonsense: Codon becomes STOP (UAA, UAG, UGA). 📌 "U Are Away, U Go Away, U Are Gone". Yields truncated protein.
- Frameshift Mutations (Insertions/Deletions not multiple of 3 bases):
- Alters reading frame downstream; often premature STOP.
- Functional Consequences:
- Loss-of-Function (LOF): Reduced or absent protein function (typically recessive).
- Gain-of-Function (GOF): Increased or novel protein function (typically dominant).
- Dominant Negative: Mutant interferes with wild-type function.
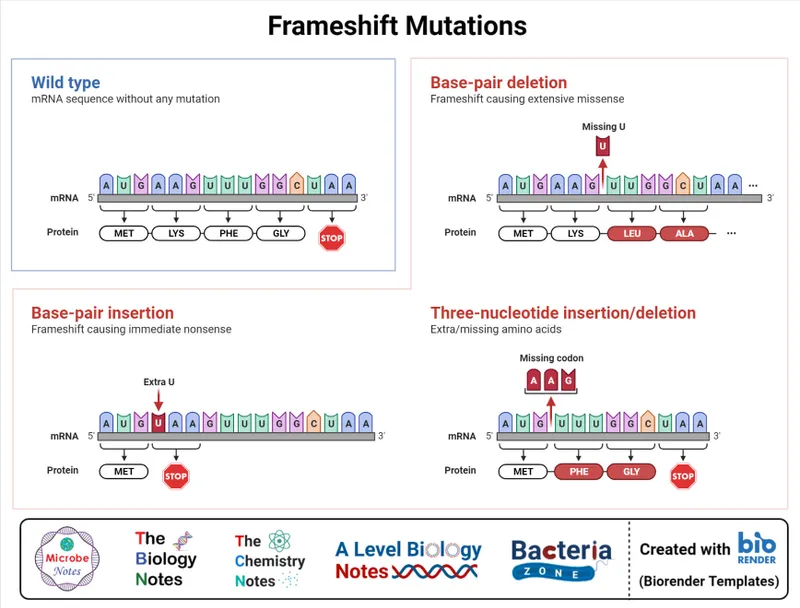
⭐ Nonsense mutations often trigger nonsense-mediated mRNA decay (NMD), a quality control degrading faulty mRNAs.
Inheritance Patterns & Exemplars - Family Fault Lines
- Pedigree Analysis: Visualizes gene transmission.

- Key Distinctions:
| Pattern | Transmission | Risk (Typical) | Sexes | Notes | E.g. |
|---|---|---|---|---|---|
| AD | Vertical | 50% (1 affected parent) | M=F | Often structural proteins; Variable expressivity, Incomplete penetrance | Huntington's, Marfan, Achondroplasia, Neurofibromatosis T1 |
| AR | Horizontal | 25% (carrier parents) | M=F | Enzyme deficiencies; Consanguinity ↑ risk | Cystic Fibrosis ($\Delta$F508), Sickle Cell, PKU, Tay-Sachs, Thalassemia |
| XLR | No male-to-male | 50% sons (carrier mom) | M >> F | Skips generations; Carrier females usually unaffected | Duchenne MD, Hemophilia A/B, G6PD deficiency, Lesch-Nyhan |
| XLD | No male-to-male | Dad→all daughters; Affected mom → 50% offspring | F > M | Often severe/lethal in males | Fragile X, Rett Syndrome, Hypophosphatemic rickets (Vit D-resist) |
| Mito. | Maternal only | All (affected mom) | M=F | Heteroplasmy; Affects high-energy tissues (CNS, muscle) | LHON, MELAS, MERRF |
⭐ Anticipation: Disease worsens or appears earlier in successive generations. Common in trinucleotide repeat disorders (e.g., Huntington's [AD], Myotonic Dystrophy [AD], Fragile X [XLD]).
📌 Mnemonic AD/AR: "Dominant diseases Destroy Generations (Vertical). Recessive diseases Run in sibs (Horizontal)."
Chromosomal Chaos & Diagnostics - Blueprint Blunders
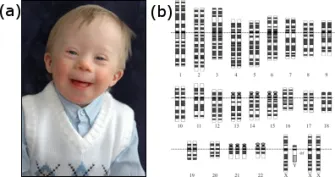
- Numerical Abnormalities (Aneuploidy): Gain/loss of chromosomes.
- Down Syndrome: Trisomy 21. 📌 Mnemonic: Voting age 21 (Down for voting).
- Turner Syndrome: 45,XO (females, short stature).
- Klinefelter Syndrome: 47,XXY (males, gynecomastia).
- Structural Abnormalities: Altered chromosome structure.
- Translocations: e.g., Philadelphia Chromosome t(9;22) in CML.
- Deletions: e.g., Cri-du-chat syndrome (del 5p).
- Key Diagnostics:
- Karyotyping: Visualizes chromosome number & structure.
- FISH: Detects specific DNA sequences/regions.
- PCR: Amplifies DNA for mutation detection.
⭐ The Philadelphia chromosome, t(9;22)(q34;q11.2), creating the BCR-ABL1 fusion gene, is a hallmark of Chronic Myeloid Leukemia (CML).
High‑Yield Points - ⚡ Biggest Takeaways
- Single-gene disorders follow Mendelian inheritance (AD, AR, X-linked).
- Trinucleotide repeat expansions cause Huntington's (CAG) & Fragile X syndrome (CGG).
- Mitochondrial diseases: maternal inheritance, affect high-energy tissues.
- Genomic imprinting: differential expression by parental origin (e.g., Prader-Willi, Angelman).
- Loss-of-function mutations: common in recessive diseases; gain-of-function in dominant ones.
- Epigenetic changes (e.g., methylation) alter gene expression, causing disease without DNA sequence change.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

