Hb: Introduction & Types - Kickstart Crew
- RBC protein for $O_2$/$CO_2$ transport & pH buffering. Tetrameric: 4 globin chains + 4 heme groups.
- Heme: Iron (Fe²⁺) in protoporphyrin IX; each binds one $O_2$ molecule.
- Key Physiological Types:
- HbA ($\alpha_2\beta_2$): Major adult form (95-98%).
- HbA2 ($\alpha_2\delta_2$): Minor adult form (2-3.5%).
- HbF ($\alpha_2\gamma_2$): Predominant fetal form (<1% in adults).
⭐ HbF has higher $O_2$ affinity than HbA (binds 2,3-BPG poorly), facilitating placental $O_2$ transfer.
Hb: Structure - Heme Team Assembly
- Overall Structure: Globular protein; tetramer of 4 polypeptide chains (globins).
- Adult HbA: 2 α-chains & 2 β-chains (α₂β₂).
- Each globin chain enfolds a heme moiety.
- Heme Moiety: Prosthetic group; Protoporphyrin IX + central ferrous iron ($Fe^{2+}$).
- $Fe^{2+}$: Forms 6 coordination bonds:
- 4 with Nitrogen atoms of porphyrin ring.
- 1 with Proximal Histidine (F8 residue) of globin.
- 1 reversible bond with $O_2$.
- $Fe^{2+}$: Forms 6 coordination bonds:
- Globin Chains: Synthesized on ribosomes.
- α-globin gene cluster: Chromosome 16.
- β-globin gene cluster: Chromosome 11.
- Key States (Conformational changes upon oxygenation):
- T (Tense) state: Deoxyhemoglobin, low $O_2$ affinity. Stabilized by 2,3-BPG.
- R (Relaxed) state: Oxyhemoglobin, high $O_2$ affinity.

⭐ Iron in heme must be in the ferrous ($Fe^{2+}$) state for $O_2$ binding. Oxidation to ferric ($Fe^{3+}$) forms methemoglobin, which cannot bind $O_2$ effectively.
Hb: Function & ODC - O2 Express
- Primary Function: Transports $O_2$ (lungs → tissues) & $CO_2$ (tissues → lungs).
- Cooperative Binding: Sigmoid $O_2$ dissociation curve (ODC); $O_2$ binding ↑ affinity for more $O_2$.
- Allosteric protein: T (taut, low affinity) ↔ R (relaxed, high affinity) states.
- Oxygen Dissociation Curve (ODC):
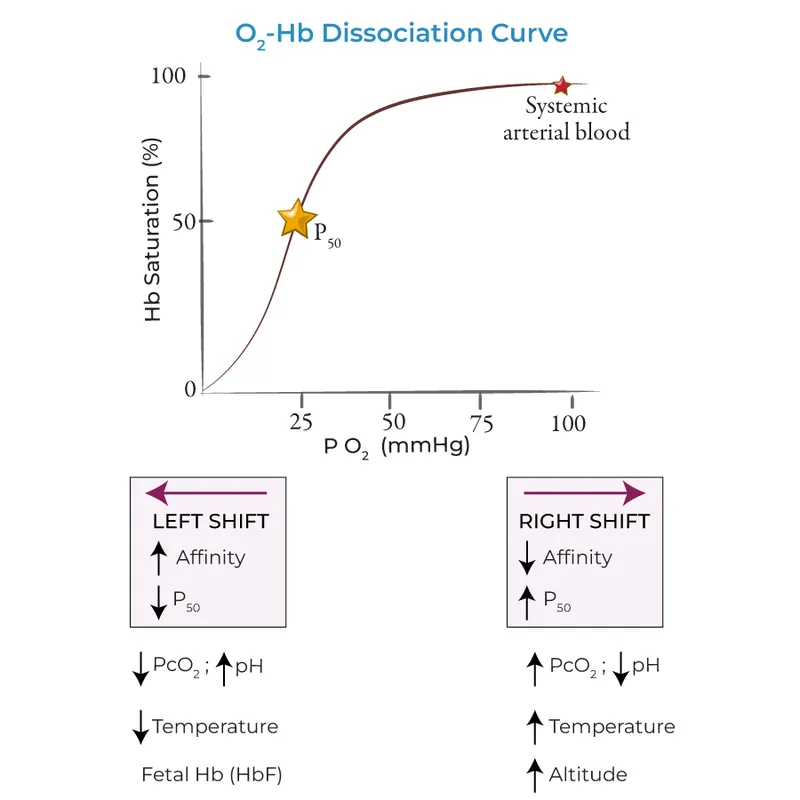
- P50: $O_2$ partial pressure for 50% Hb saturation. Normal ≈ 26.6 mmHg.
- Right Shift (↓ Affinity, ↑ $O_2$ Release):
- Factors: ↑$CO_2$, ↑$H^+$ (↓pH), ↑2,3-BPG, ↑Temperature, ↑Altitude.
- 📌 Mnemonic: "CADET, face Right!" ($CO_2$, Acid, 2,3-DPG, Exercise, Temperature).
- Left Shift (↑ Affinity, ↓ $O_2$ Release):
- Factors: ↓$CO_2$, ↓$H^+$ (↑pH), ↓2,3-BPG, ↓Temperature, HbF, CO, MetHb.
- Key Effects:
- Bohr Effect: $H^+$ & $CO_2$ promote $O_2$ release (right shift).
- Haldane Effect: $O_2$ binding promotes $CO_2$ release in lungs.
- 2,3-BPG: Stabilizes T-state, facilitates $O_2$ unloading.
⭐ Fetal hemoglobin (HbF) has a higher O2 affinity than HbA because it binds 2,3-BPG poorly, resulting in a left-shifted ODC.
Hb: Clinical Variants - Alter Egos
- HbS (Sickle): $\beta$6 Glu $\rightarrow$ Val. Low $O_2$ $\rightarrow$ polymerization, sickling, vaso-occlusion.
- HbC Disease: $\beta$6 Glu $\rightarrow$ Lys. Mild hemolysis, crystals, target cells.
- HbE Disease/Trait: $\beta$26 Glu $\rightarrow$ Lys. Mild microcytic anemia; SE Asia.
- Methemoglobin (MetHb): $Fe^{3+}$ (ferric) state; no $O_2$ binding.
- Causes: Oxidant drugs (nitrites).
- Clinical: Cyanosis, chocolate-brown blood. Rx: Methylene blue.
- Carboxyhemoglobin (COHb): CO binds Hb (affinity >200x $O_2$).
- Clinical: Cherry-red skin, headache, hypoxia. Left shift ODC.
⭐ In Methemoglobinemia, pulse oximetry is falsely reassuring, often reading around 85%.
High‑Yield Points - ⚡ Biggest Takeaways
- Hemoglobin (Hb) is a tetrameric protein (e.g., HbA: α₂β₂), carrying O₂.
- Each heme group contains Fe²⁺ (ferrous iron), binding one O₂ molecule.
- Cooperative O₂ binding yields a sigmoidal oxygen dissociation curve (ODC).
- 2,3-BPG stabilizes Hb's T-state, shifting ODC right (↓O₂ affinity, ↑O₂ release).
- Bohr effect: ↓pH or ↑CO₂ shifts ODC right, enhancing O₂ delivery.
- HbF (α₂γ₂) has higher O₂ affinity than HbA (less 2,3-BPG binding).
- Methemoglobin (Fe³⁺) cannot bind O₂; treat with methylene blue.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

