Primary Hemostasis - Plug The Leak!
Rapid formation of a temporary platelet plug at the site of vascular injury. Involves platelets, vWF, and vessel wall.
- Sequence of Events:
- Endothelial Injury: Exposes subendothelial collagen and releases von Willebrand Factor (vWF).
- Platelet Adhesion: Platelets adhere to exposed collagen. This is mediated by vWF bridging collagen and platelet receptor GPIb.
- Platelet Activation: Adhesion triggers:
- Shape change (discoid → spiny with pseudopods).
- Degranulation: Release of ADP (potent activator & aggregator) and Thromboxane A2 (TXA2 - vasoconstrictor, promotes aggregation).
- Platelet Aggregation: Activated platelets express GPIIb/IIIa receptors. Fibrinogen binds to GPIIb/IIIa on adjacent platelets, linking them together to form the primary hemostatic plug.

⭐ Clinical Correlates: Bernard-Soulier syndrome is a defect in GPIb (impaired adhesion). Glanzmann thrombasthenia is a defect in GPIIb/IIIa (impaired aggregation).
Secondary Hemostasis - Cascade Conundrum
Secondary hemostasis involves a cascade of enzymatic reactions leading to the formation of a stable fibrin clot, reinforcing the primary platelet plug. (118 words)
- Pathways:
- Intrinsic Pathway (Contact Activation): Initiated by Factor XII activation. Involves Factors XI, IX, VIII. Slower, amplifies coagulation.
- Extrinsic Pathway (Tissue Factor): Initiated by Tissue Factor (TF, Factor III) exposure. Involves Factor VII. Rapid, primary initiator in vivo.
- Common Pathway: Convergence point. Factors X, V, II (Prothrombin), I (Fibrinogen).
- $Prothrombin \xrightarrow{Factor\ Xa, Va, Ca^{2+}, PL} Thrombin$
- $Fibrinogen \xrightarrow{Thrombin} Fibrin\ monomers\ (soluble)$
- Fibrin Clot Formation & Stabilization:
- Thrombin converts fibrinogen to fibrin monomers.
- Fibrin monomers polymerize.
- Factor XIIIa (activated by Thrombin) cross-links fibrin polymers, forming a stable, insoluble clot.
- Vitamin K-Dependent Factors: II, VII, IX, X, Protein C, S. Essential for synthesis. 📌 Mnemonic: "1972" (factors 2,7,9,10) + C & S.
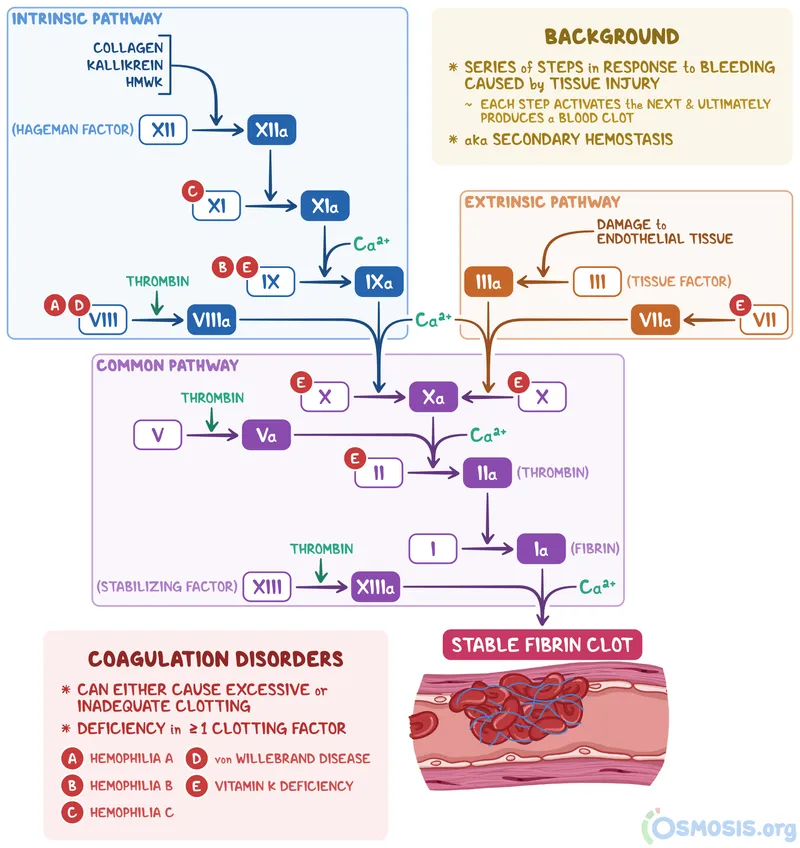
⭐ Tissue Factor (Factor III) is the primary physiological initiator of coagulation.
Clot Regulation - Checks & Balances
Natural Anticoagulants:
- Antithrombin III:
- Potentiated by Heparin.
- Inhibits Thrombin (IIa), IXa, Xa, XIa, XIIa.
- Protein C & S (Vit K-dependent):
- Activated by Thrombin-Thrombomodulin complex.
- Inactivate cofactors Va, VIIIa.
- TFPI (Tissue Factor Pathway Inhibitor):
- Inhibits TF-VIIa complex & Factor Xa.
Fibrinolysis (Clot Dissolution):
- $Plasminogen \xrightarrow{tPA/uPA} Plasmin$
- Plasmin: Degrades fibrin/fibrinogen → FDPs (Fibrin Degradation Products).
- D-dimer: Specific FDP, indicates breakdown of cross-linked fibrin.
- Activators:
- t-PA (tissue Plasminogen Activator): Main physiological activator, from endothelium.
- u-PA (urokinase Plasminogen Activator).
- Inhibitors:
- PAI-1 (Plasminogen Activator Inhibitor-1): Inhibits t-PA/u-PA.
- $\alpha_2$-Antiplasmin: Directly inhibits plasmin.

⭐ Activated Protein C Resistance (e.g., Factor V Leiden mutation) is a common cause of inherited thrombophilia.
Lab Tests & Links - Diagnostic Dots
- PT (Prothrombin Time): Extrinsic & Common pathways (Factors VII, X, V, II, I). Monitors Warfarin. 📌 PT (Play Tennis Outside - Extrinsic).
- aPTT (Activated Partial Thromboplastin Time): Intrinsic & Common pathways (Factors XII, XI, IX, VIII, X, V, II, I). Monitors Heparin. 📌 PTT (Play Table Tennis Inside - Intrinsic).
- TT (Thrombin Time): Assesses Fibrinogen $\rightarrow$ Fibrin conversion.
- Fibrinogen Assay: Quantifies fibrinogen levels.
- D-dimer Assay: Detects fibrin degradation products.
Interpretation Highlights:
| Disorder (Defect) | PT | aPTT | TT |
|---|---|---|---|
| Hemophilia A (↓FVIII) / B (↓FIX) | Normal | ↑ | Normal |
| vWD (↓vWF) | Normal | N/↑¹ | Normal |
| Vit K Def/Warfarin | ↑ | ↑/N | Normal |
⭐ Mixing studies: Correction of PT/aPTT suggests factor deficiency; no correction suggests an inhibitor.
High‑Yield Points - ⚡ Biggest Takeaways
- Primary hemostasis: Platelet adhesion (vWF-GpIb), activation, and aggregation (GpIIb/IIIa-fibrinogen).
- Coagulation cascade (secondary hemostasis): Vitamin K vital for factors II, VII, IX, X, C, S.
- Fibrinolysis: Plasmin (from plasminogen via tPA) degrades fibrin, forming D-dimers.
- Key natural anticoagulants: Antithrombin III (inhibits thrombin, Xa), Protein C/S (inactivate Va/VIIIa).
- PT for extrinsic pathway (Warfarin); aPTT for intrinsic pathway (Heparin).
- Common bleeding disorders: Hemophilias (Factor VIII/IX↓), von Willebrand Disease.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

