MS Fundamentals - What's the Charge?
- Core principle: Measures mass-to-charge ratio ($m/z$) of ions. Neutral molecules are invisible.
- Sample molecules are ionized (charged) in the gas phase.
- Common charges: $\mathbf{+1}, \mathbf{+2}, \mathbf{-1}$.
- Ions are accelerated and separated by electric/magnetic fields based on their $m/z$.
- The $m/z$ value is key: for positive ions, $m/z = (M_{molecule} + n \cdot M_{H^+})/n$.
- Output: Mass spectrum (ion abundance vs. $m/z$).
⭐ Multiply charged ions (e.g., proteins, $z > \mathbf{1}$) appear at lower $m/z$ values, extending the instrument's effective mass range.
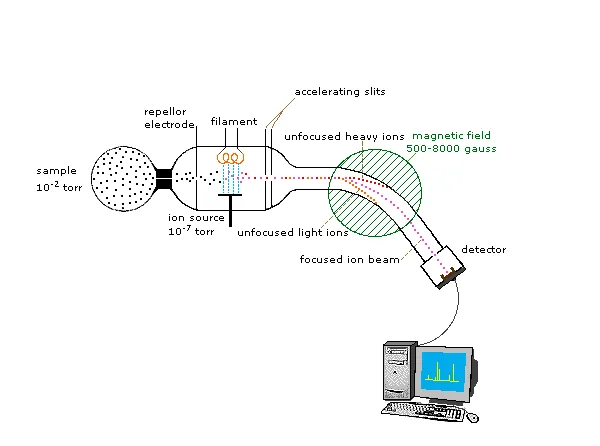
The MS Toolkit - Parts & Players
- Ion Source: Generates gas-phase ions from analyte.
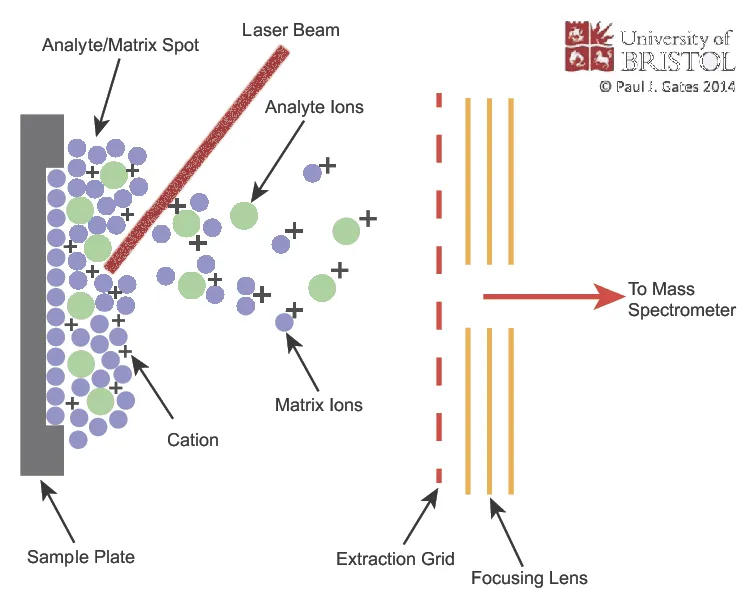
- MALDI: Sample + matrix + laser. For peptides, proteins. Soft ionization.
- ESI: Charged capillary, liquid sample. For polar, labile molecules. Forms multiply charged ions.
- Mass Analyzer: Separates ions by $m/z$ ratio.
- TOF: Measures ion flight time. Lighter ions are faster.
- Quadrupole: Electric fields filter ions by $m/z$.
- Orbitrap: Traps ions; offers high resolution & mass accuracy.
- Detector: Counts ions; generates mass spectrum (intensity vs. $m/z$).

⭐ ESI is a "soft" ionization method, vital for analyzing intact large biomolecules (e.g., proteins) with minimal fragmentation, often producing multiply charged ions.
Key Techniques - Ionize & Analyze!
- Ionization Methods:
- MALDI (Matrix-Assisted Laser Desorption/Ionization):
- Large biomolecules (proteins, peptides); solid sample + matrix.
- Laser desorption/ionization; primarily singly charged ions $[M+H]^+$.
- 📌 MALDI: "Mighty Molecules, Laser Driven Ionization".
- ESI (Electrospray Ionization):
- Peptides, proteins from liquid solutions.
- High voltage → charged droplets → multiply charged ions $[M+nH]^{n+}$.
- 📌 ESI: "Existing Solutions/Samples, Electrically Sprayed Ions".
⭐ ESI's multiple charging allows analysis of very large molecules (e.g., proteins > 100 kDa) on analyzers with limited $m/z$ ranges.
- EI (Electron Ionization):
- Small, volatile molecules; extensive fragmentation (structural info).
- MALDI (Matrix-Assisted Laser Desorption/Ionization):
- Mass Analyzers:
- TOF (Time-of-Flight):
- Ions drift; lighter ions faster. $t \propto \sqrt{m/z}$.
- High resolution; common with MALDI.
- Quadrupole (Q):
- Mass filter using DC/RF fields; stable ion paths for selected $m/z$.
- Ion Trap (IT):
- Traps ions; sequential ejection by $m/z$. Good for MSn.
- Orbitrap:
- High resolution/accuracy; ions orbit central electrode.
- TOF (Time-of-Flight):
Biochem Applications - MS Solves It!
Mass Spectrometry (MS) is a powerhouse in biochemical analysis, offering diverse applications:
- Protein Characterization:
- Identification: Peptide Mass Fingerprinting (PMF) matches peptide masses to database proteins.
- Sequencing: Tandem MS (MS/MS) fragments peptides for de novo sequencing.
- PTM Analysis: Detects phosphorylation, glycosylation, ubiquitination.
- Proteomics:
- Quantitative analysis (e.g., SILAC, iTRAQ) to compare protein levels.
- Mapping protein interaction networks.
- Metabolomics:
- Profiling endogenous small molecules (metabolites).
- Identifying disease biomarkers.
- Drug Discovery:
- Pharmacokinetics (ADME: Absorption, Distribution, Metabolism, Excretion).
- Metabolite identification of drugs.
- Clinical Diagnostics:
- Newborn screening for metabolic disorders (e.g., PKU, MCADD).
- Rapid microbial identification.
- Cancer biomarker detection.
⭐ MS is pivotal in newborn screening, detecting inborn errors of metabolism like Phenylketonuria (PKU) by analyzing amino acid and acylcarnitine profiles from dried blood spots.
High‑Yield Points - ⚡ Biggest Takeaways
- MS Principle: Measures mass-to-charge ratio (m/z) of ions for identification/quantification.
- Core Components: Ion Source (MALDI, ESI), Mass Analyzer (TOF, Quadrupole), Detector.
- MALDI: For large biomolecules (proteins, peptides); ESI: For polar molecules, often with LC.
- Tandem MS (MS/MS): Enables structural elucidation and peptide sequencing via ion fragmentation.
- Key Applications: Proteomics (protein ID), metabolomics, drug analysis, newborn screening.
- Clinical Utility: Microorganism ID, biomarker discovery, therapeutic drug monitoring.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

