Alkalosis Overview - pH Up Party
- Alkalosis: Systemic increase in blood $pH > \textbf{7.45}$, indicating decreased hydrogen ion concentration ([H⁺]↓).
- Key physiological disturbances:
- ↑ Neuromuscular excitability: Paresthesias, muscle cramps, carpopedal spasm, tetany.
- Cerebral vasoconstriction: Dizziness, lightheadedness, confusion.
- Cardiac effects: Potential for arrhythmias.
- Classified into Respiratory and Metabolic alkalosis.
⭐ Alkalosis ($pH > \textbf{7.45}$) can precipitate hypocalcemic tetany by decreasing ionized calcium and cause hypokalemia due to intracellular K+ shift.
Respiratory Alkalosis - Huffing & Puffing pH
- Definition: Primary ↓ $PCO_2$ (hypocapnia) leading to ↑ pH.
- Pathophysiology: Alveolar hyperventilation blows off $CO_2$.
- Lab Findings:
- pH > 7.45
- $PCO_2$ < 35 mmHg
- $HCO_3^-$ ↓ (renal compensation: takes hours to days)
- Acute: $HCO_3^-$ ↓ by 2 mEq/L for every 10 mmHg ↓ in $PCO_2$.
- Chronic: $HCO_3^-$ ↓ by 4-5 mEq/L for every 10 mmHg ↓ in $PCO_2$.
- Causes: 📌 CHAMPS
- CNS disorders (stroke, trauma)
- Hypoxia (high altitude, pneumonia, PE)
- Anxiety, pain, fever, ASA (Salicylates)
- Mechanical ventilation (overventilation)
- Progesterone (pregnancy), Pulmonary disease (restrictive)
- Sepsis, Salicylates
- Clinical Features: Lightheadedness, dizziness, paresthesias (perioral, digital), carpopedal spasm, tetany (due to ↓ ionized $Ca^{2+}$), syncope, seizures.
⭐ Salicylate toxicity classically presents with a mixed acid-base disorder: primary respiratory alkalosis (due to direct stimulation of medullary respiratory center) and primary metabolic acidosis.

- Management: Treat underlying cause. Reassurance, rebreathing into a paper bag (for anxiety-induced hyperventilation). Adjust ventilator settings if iatrogenic.
Metabolic Alkalosis - Bicarb Bonanza
-
Pathophysiology: Primary ↑ $[HCO_3^-]$ ($> \textbf{28 mEq/L}$), leading to ↑ pH ($> \textbf{7.45}$).
- Compensation: Respiratory acidosis; $pCO_2$ ↑ by $\textbf{0.7 mmHg}$ for each $\textbf{1 mEq/L}$ ↑ in $[HCO_3^-]$ above 24 mEq/L.
- Maximal compensation: $pCO_2$ rarely exceeds $\textbf{55 mmHg}$.
-
Etiology & Management: Differentiated by urine chloride ($U_{Cl^-}$).
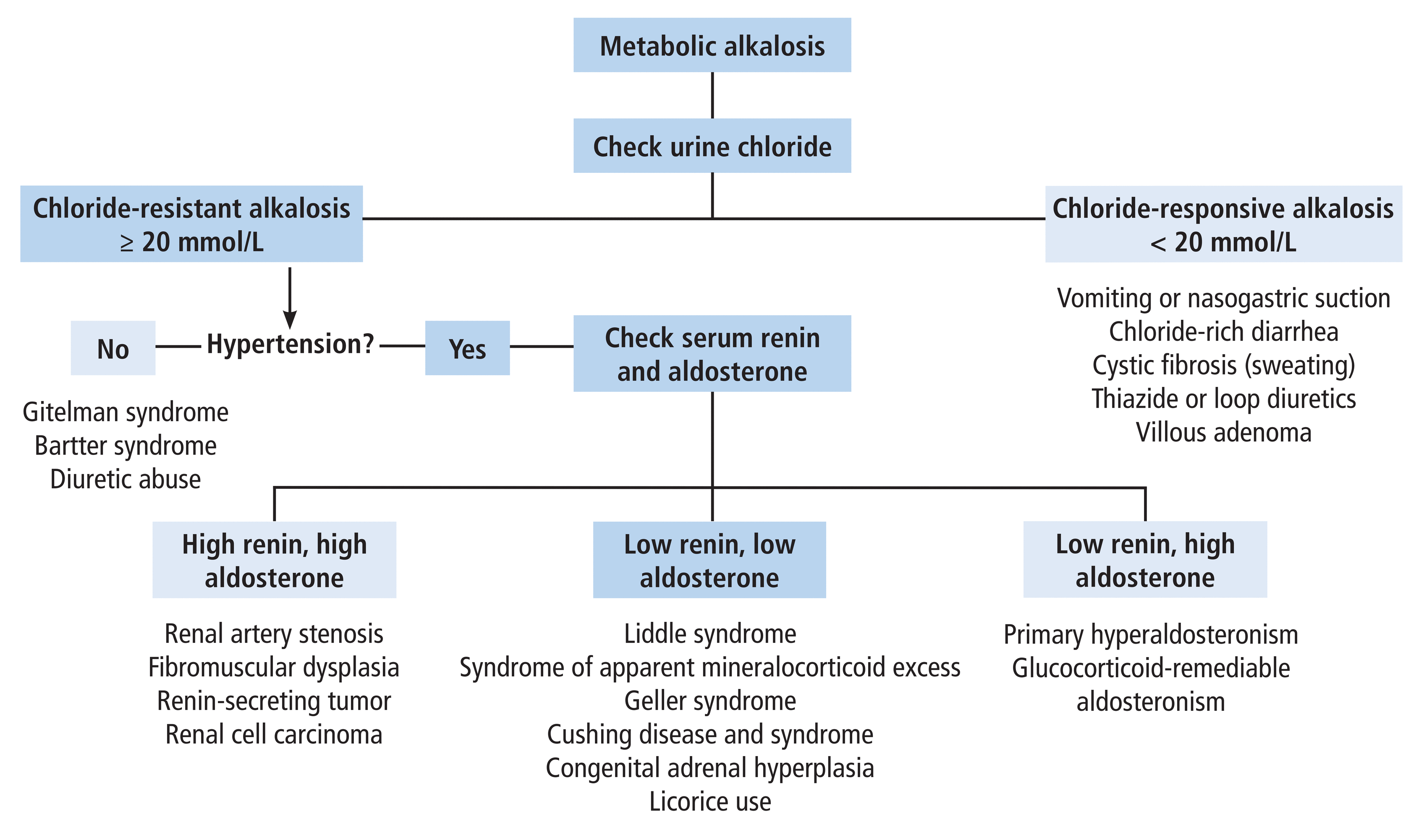
- Clinical Features: Often asymptomatic. Severe:
- Hypokalemia effects: Weakness, arrhythmias.
- ↓ Ionized $Ca^{2+}$: Tetany, paresthesias, seizures.
- Compensatory hypoventilation.
- Diagnosis: ABG, serum electrolytes, urine $Cl^-$, $K^+$, $Na^+$.
⭐ Urine chloride is crucial in differentiating saline-responsive (< 15 mEq/L) from saline-resistant (> 20 mEq/L) metabolic alkalosis.
- Treatment Principles:
- Address underlying cause.
- Correct volume depletion (if present) with isotonic saline.
- Replete $K^+$ if hypokalemic.
- Severe cases ($pH > \textbf{7.6}$): Acetazolamide, HCl infusion (rare), dialysis.
Compensation & Mixed Disorders - Body's Balancing Act
Body attempts to normalize pH. Suspect mixed disorder if compensation is outside expected range.
- Respiratory Alkalosis (Primary ↓PaCO₂):
- Metabolic Comp: Kidneys ↓ plasma [HCO₃⁻].
- Acute: ↓10 PaCO₂ → ↓2 [HCO₃⁻].
- Chronic: ↓10 PaCO₂ → ↓4-5 [HCO₃⁻].
- Metabolic Comp: Kidneys ↓ plasma [HCO₃⁻].
- Metabolic Alkalosis (Primary ↑[HCO₃⁻]):
- Respiratory Comp: Lungs ↑PaCO₂ (hypoventilation).
- Expected PaCO₂: $\text{PaCO}_2 = (0.7 \times [HCO_3^-]) + 20 \text{ mmHg (range } \pm 5)$.
- Respiratory Comp: Lungs ↑PaCO₂ (hypoventilation).
⭐ The PCO₂ in compensatory hypoventilation for metabolic alkalosis rarely rises above 55 mmHg due to the overriding stimulus of hypoxic drive.
High‑Yield Points - ⚡ Biggest Takeaways
- Respiratory alkalosis: Caused by hyperventilation (↓ PCO₂); e.g., hypoxia, anxiety, salicylates. Renal HCO₃⁻ excretion is compensatory.
- Metabolic alkalosis: ↑ HCO₃⁻. Key causes: vomiting/NG suction (saline-responsive, urine Cl⁻ < 10) or mineralocorticoid excess (saline-resistant, urine Cl⁻ > 20).
- Hypoventilation (↑ PCO₂) is the limited respiratory compensation for metabolic alkalosis.
- Hypokalemia often accompanies and worsens metabolic alkalosis.
- Tetany and paresthesias (↓ ionized Ca²⁺) can occur in severe respiratory alkalosis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

