Thorax

On this page
🏗️ The Thoracic Fortress: Architectural Mastery of the Central Body Vault
You'll master the thorax as an integrated command center where skeletal architecture, pressure gradients, and cardiovascular mechanics converge to sustain life. This lesson builds your understanding from the bony vault and pleural dynamics through airway navigation and heart-vessel relationships, then synthesizes these systems into clinical decision frameworks. You'll learn why negative pressure drives ventilation, how anatomical landmarks guide procedures, and which hemodynamic principles optimize cardiac performance. By connecting structure to function and physiology to practice, you'll gain the systematic thinking required for confident thoracic assessment and intervention.
Thoracic Boundaries: The Protective Framework
The thoracic cavity extends from the thoracic inlet (T1 vertebral level) to the diaphragm (T12 level), creating a 30cm vertical chamber that houses critical organs. This space demonstrates remarkable engineering efficiency, maximizing protection while enabling dynamic volume changes during respiration.
- Superior boundary: Thoracic inlet
- Bounded by T1 vertebra, first ribs, and manubrium
- Diameter: 12cm transverse × 5cm anteroposterior
- Critical structures traverse this narrow aperture
- Inferior boundary: Diaphragm
- Dome-shaped muscle separating thorax from abdomen
- Central tendon at T8-T9 level during expiration
- 3 major openings at T8, T10, and T12 levels
- Anterior boundary: Sternum and costal cartilages
- 17cm long sternum with 3 distinct parts
- Sternal angle at T4-T5 level (critical landmark)
- Posterior boundary: Thoracic vertebrae and ribs
- 12 thoracic vertebrae with increasing size caudally
- Costovertebral joints enable rib mobility
📌 Remember: STOP for thoracic inlet contents - Subclavian vessels, Trachea, Oesophagus, Phrenic/vagus nerves. These structures must navigate the 5cm anteroposterior diameter without compression.
| Thoracic Level | Anterior Landmark | Posterior Landmark | Clinical Significance | Normal Measurements |
|---|---|---|---|---|
| T2 | Sternal angle | T4-T5 disc | Aortic arch, tracheal bifurcation | 5cm from skin |
| T4 | Nipple line | T6 vertebra | Right atrium border | 8cm from midline |
| T6 | Xiphoid process | T8-T9 disc | Heart apex, diaphragm | 10cm from sternum |
| T8 | Costal margin | T10 vertebra | IVC through diaphragm | 12cm intercostal space |
| T10 | Epigastrium | T12 vertebra | Esophageal hiatus | 15cm from xiphoid |
Mediastinal Architecture: The Central Command Center
The mediastinum functions as the thorax's central corridor, containing all thoracic organs except the lungs. This 15cm wide × 25cm tall space demonstrates sophisticated compartmentalization that enables surgical access while protecting vital structures.
- Superior mediastinum (above sternal angle)
- Contains great vessels, trachea, esophagus
- Thymus occupies anterior portion in children
- Brachiocephalic veins form SVC at T3 level
- Right brachiocephalic: 2.5cm length
- Left brachiocephalic: 6cm length (crosses midline)
- Anterior mediastinum (between sternum and pericardium)
- Thymic remnants and mediastinal fat
- Internal mammary vessels descend 1cm from sternal edge
- Potential space for pneumomediastinum accumulation
- Middle mediastinum (pericardial contents)
- Heart and pericardium occupy 60% of space
- Phrenic nerves course along lateral pericardium
- Pulmonary vessels and main bronchi
- Posterior mediastinum (behind pericardium)
- Descending aorta shifts leftward from T4 level
- Azygos system provides venous drainage
- Thoracic duct ascends from T12 to T5 levels
💡 Master This: Mediastinal shift indicates pressure imbalance between pleural cavities. Tension pneumothorax causes >2cm tracheal deviation away from affected side, while massive pleural effusion shifts mediastinum >1.5cm toward unaffected side.
⭐ Clinical Pearl: Mediastinal widening >8cm on chest X-ray suggests aortic dissection, lymphadenopathy, or mediastinal hematoma. Normal mediastinal width measures 6-8cm at the level of the aortic arch.
The thoracic framework establishes the foundation for understanding complex cardiopulmonary interactions. Connect this architectural knowledge through pleural mechanics to understand respiratory physiology patterns.
🏗️ The Thoracic Fortress: Architectural Mastery of the Central Body Vault
⚙️ The Pleural Engine: Pressure Dynamics and Respiratory Mechanics
Pleural Space Architecture: The Frictionless Interface
The pleural cavity represents a potential space containing 10-20ml of pleural fluid that enables frictionless lung movement during respiration. This microscopic interface demonstrates remarkable engineering efficiency, reducing friction to <0.01 coefficient while maintaining structural integrity.
- Visceral pleura (lung surface)
- Mesothelial cells produce surfactant-like proteins
- Subpleural capillaries maintain 25 mmHg pressure
- Lymphatic drainage removes excess fluid at 20ml/day
- Parietal pleura (chest wall surface)
- Systemic blood supply from intercostal arteries
- Lymphatic stomata (2-12 μm diameter) enable fluid absorption
- Sensory innervation from intercostal and phrenic nerves
- Pleural fluid dynamics
- Protein concentration: 1.5-2.0 g/dL (serum: 6-8 g/dL)
- Cell count: <1000 cells/μL (<50% neutrophils)
- pH: 7.60-7.64 (slightly alkaline)
- Glucose: 60-100 mg/dL (serum ratio: 0.6-1.0)
📌 Remember: LIGHT criteria distinguish exudate from transudate - LDH ratio >0.6, Increased protein ratio >0.5, Glucose <60 mg/dL, High cell count >1000, Turbid appearance.
Respiratory Pressure Mechanics: The Ventilation Engine
Breathing occurs through coordinated pressure changes that create pressure gradients driving airflow. During quiet inspiration, diaphragmatic contraction generates -1 to -3 mmHg alveolar pressure, while pleural pressure decreases to -8 mmHg.
- Inspiration mechanics
- Diaphragm descent: 1.5cm during quiet breathing
- Rib elevation: 2-5cm increase in anteroposterior diameter
- Pleural pressure: Decreases from -5 to -8 mmHg
- Alveolar pressure: Drops -1 to -3 mmHg below atmospheric
- Pressure gradient drives 500ml tidal volume
- Airflow rate: 0.5 L/sec during peak inspiration
- Expiration mechanics
- Passive recoil of elastic tissues
- Pleural pressure: Returns to -5 mmHg
- Alveolar pressure: Rises +1 to +3 mmHg above atmospheric
- Airflow reversal empties lungs at 0.4 L/sec
⭐ Clinical Pearl: Pneumothorax eliminates negative pleural pressure, causing immediate lung collapse. Tension pneumothorax creates positive pleural pressure >+10 mmHg, compressing mediastinal structures and requiring immediate decompression.
| Respiratory Phase | Pleural Pressure | Alveolar Pressure | Transpulmonary Pressure | Airflow Direction |
|---|---|---|---|---|
| Quiet expiration | -5 mmHg | 0 mmHg | +5 mmHg | None |
| Early inspiration | -6 mmHg | -1 mmHg | +5 mmHg | Inward |
| Peak inspiration | -8 mmHg | 0 mmHg | +8 mmHg | None |
| Early expiration | -6 mmHg | +1 mmHg | +7 mmHg | Outward |
| Forced expiration | +5 mmHg | +10 mmHg | +5 mmHg | Rapid outward |
Lung compliance measures the volume change per unit pressure change, normally 200ml/cmH2O in healthy adults. This parameter reflects the lung's ability to expand during inspiration and determines the work of breathing.
- Static compliance (no airflow)
- Normal value: 200ml/cmH2O
- Decreased in: Fibrosis (<100ml/cmH2O), pneumonia
- Increased in: Emphysema (>300ml/cmH2O), aging
- Dynamic compliance (during breathing)
- Frequency-dependent decrease in disease
- Small airway obstruction reduces dynamic compliance
- Normal ratio: Dynamic/static = 0.8-1.0
💡 Master This: Compliance = ΔVolume/ΔPressure. Restrictive disease decreases compliance (stiff lungs), requiring higher pressures for same volume. Obstructive disease may increase compliance but impairs elastic recoil.
⭐ Clinical Pearl: Pressure-volume loops reveal respiratory mechanics. Hysteresis (different inflation/deflation curves) indicates surfactant function. Flattened loops suggest reduced compliance in restrictive disease.
Understanding pleural pressure dynamics provides the foundation for interpreting respiratory pathophysiology. Connect these mechanical principles through airway anatomy to understand ventilation-perfusion relationships.
⚙️ The Pleural Engine: Pressure Dynamics and Respiratory Mechanics
🎯 The Airway Command Center: Navigation and Clinical Recognition Patterns
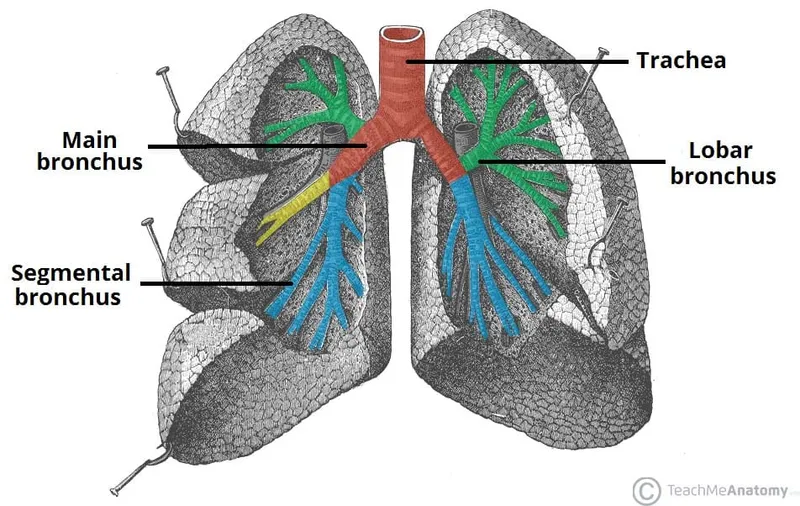
Tracheobronchial Architecture: The Branching Highway System
The trachea begins at the C6 vertebral level and extends 10-12cm to the carina at T5 level. This central airway demonstrates sophisticated engineering with C-shaped cartilage rings providing structural support while maintaining flexibility for swallowing and neck movement.
- Tracheal specifications
- Length: 10-12cm (adult), 4-5cm (infant)
- Diameter: 15-20mm (adult), 4-5mm (newborn)
- Cartilage rings: 16-20 incomplete rings
- Posterior membrane: Trachealis muscle enables diameter changes
- Cough reflex: Diameter reduces to 2-3mm
- Deep inspiration: Diameter increases to 25mm
- Carina anatomy (critical landmark)
- T5 vertebral level (sternal angle externally)
- Sharp ridge between main bronchi
- Highly sensitive to mechanical stimulation
- Cough reflex threshold: <1mm displacement
📌 Remember: CARINA landmarks - C5-T5 level, Aortic arch relation, Ridge between bronchi, Intubation landmark, Nerve-rich (vagal), Angle of 60-70 degrees.
Main Bronchi: The Primary Distribution Channels
The main bronchi demonstrate asymmetric anatomy that directly impacts clinical practice. The right main bronchus measures 2.5cm length with a 25-degree angle, while the left main bronchus extends 5cm with a 45-degree angle from the trachea.
- Right main bronchus characteristics
- Shorter (2.5cm), wider (15mm), more vertical
- 25-degree angle from tracheal axis
- First branch: Right upper lobe bronchus (1cm from carina)
- Aspiration risk: 85% of foreign bodies enter right side
- Left main bronchus characteristics
- Longer (5cm), narrower (13mm), more horizontal
- 45-degree angle from tracheal axis
- Crosses anterior to esophagus and descending aorta
- More resistant to foreign body aspiration
| Bronchial Feature | Right Main | Left Main | Clinical Significance |
|---|---|---|---|
| Length | 2.5cm | 5cm | Intubation depth |
| Angle | 25 degrees | 45 degrees | Aspiration risk |
| Diameter | 15mm | 13mm | Bronchoscope size |
| First branch | 1cm from carina | 5cm from carina | Surgical landmarks |
| Relations | Azygos vein | Aortic arch | Compression sites |
Each lung demonstrates distinct bronchial patterns that enable precise anatomical localization. The right lung contains 3 lobes with 10 bronchopulmonary segments, while the left lung has 2 lobes with 8-10 segments (variable lingular anatomy).
- Right lung bronchial pattern
- Upper lobe: 3 segments (apical, posterior, anterior)
- Middle lobe: 2 segments (lateral, medial)
- Lower lobe: 5 segments (superior, medial basal, anterior basal, lateral basal, posterior basal)
- Superior segment branches posteriorly from lower lobe
- Basal segments arise from common basal trunk
- Left lung bronchial pattern
- Upper lobe: 4-5 segments (apicoposterior, anterior, superior lingular, inferior lingular)
- Lower lobe: 4-5 segments (superior, anteromedial basal, lateral basal, posterior basal)
- Lingula represents middle lobe equivalent
- Anteromedial basal may be single or divided
⭐ Clinical Pearl: Segmental anatomy enables precise localization of pathology. Right middle lobe syndrome involves medial and lateral segments, while lingular pneumonia affects superior and inferior lingular segments.
Clinical Recognition Patterns: "See This, Think That"
Bronchoscopic navigation requires systematic pattern recognition based on anatomical landmarks and branching angles. Successful bronchoscopy depends on recognizing carina morphology, bronchial angles, and segmental takeoffs.
- Carina assessment patterns
- Sharp, thin ridge: Normal anatomy
- Widened, blunted: Subcarinal lymphadenopathy
- Fixed, immobile: Malignant invasion
- Erythematous: Inflammatory process
- Bronchial angle recognition
- Right upper lobe: Anterior takeoff at 45 degrees
- Right middle lobe: Anterior-lateral at 90 degrees
- Left upper lobe: Anterior-superior at 60 degrees
- Lower lobe superior segments: Posterior takeoff bilaterally
💡 Master This: Bronchoscopic landmarks follow the "3-2-1 rule" - 3 segments in RUL, 2 segments in RML, 1 superior segment in each lower lobe branches posteriorly. This pattern guides systematic examination.
⭐ Clinical Pearl: Foreign body aspiration shows 90% right-sided preference due to straighter angle and larger diameter. Peanuts cause severe inflammatory response within 6-12 hours, while plastic objects may remain asymptomatic for weeks.
Understanding airway architecture provides the framework for interpreting respiratory pathophysiology. Connect these anatomical patterns through vascular relationships to understand ventilation-perfusion matching principles.
🎯 The Airway Command Center: Navigation and Clinical Recognition Patterns
🔍 The Cardiovascular Command Matrix: Systematic Heart-Vessel Integration
Cardiac Chamber Architecture: The Four-Room Powerhouse
The heart functions as a dual pump system with right heart handling venous return at low pressures (0-25 mmHg) and left heart managing systemic circulation at high pressures (80-120 mmHg). This pressure differential reflects the 6-fold difference in systemic versus pulmonary vascular resistance.
- Right atrium specifications
- Volume capacity: 100-150ml at end-diastole
- Pressure range: 0-8 mmHg (central venous pressure)
- Wall thickness: 2-3mm (thinnest cardiac chamber)
- Key structures: Tricuspid valve (3 leaflets), coronary sinus opening
- Tricuspid annulus: 11-13cm circumference
- Coronary sinus: 3cm length, 1cm diameter
- Right ventricle characteristics
- Volume: 150-200ml end-diastolic, 50-70ml end-systolic
- Ejection fraction: 55-70% (higher than left ventricle)
- Wall thickness: 3-5mm (one-third of left ventricle)
- Systolic pressure: 15-25 mmHg (pulmonary artery pressure)
- Left atrium specifications
- Volume capacity: 80-120ml at end-diastole
- Pressure range: 8-12 mmHg (pulmonary capillary wedge)
- Wall thickness: 3-4mm (muscular posterior wall)
- Pulmonary veins: 4 openings (2 right, 2 left)
- Left ventricle powerhouse
- Volume: 120-180ml end-diastolic, 40-60ml end-systolic
- Ejection fraction: 55-65% (normal range)
- Wall thickness: 8-12mm (muscular wall)
- Systolic pressure: 100-140 mmHg (aortic pressure)
📌 Remember: RAMP for right heart pressures - RA: 0-8 mmHg, Atrial kick: +2-4 mmHg, Mitral equivalent (tricuspid): 0-5 mmHg, Pulmonary artery: 15-25 mmHg.
| Chamber | End-Diastolic Volume | End-Systolic Volume | Ejection Fraction | Wall Thickness | Normal Pressure |
|---|---|---|---|---|---|
| Right Atrium | 100-150ml | 50-80ml | 45-55% | 2-3mm | 0-8 mmHg |
| Right Ventricle | 150-200ml | 50-70ml | 55-70% | 3-5mm | 15-25 mmHg |
| Left Atrium | 80-120ml | 30-50ml | 60-70% | 3-4mm | 8-12 mmHg |
| Left Ventricle | 120-180ml | 40-60ml | 55-65% | 8-12mm | 100-140 mmHg |
The great vessels demonstrate asymmetric branching patterns that reflect embryological development and functional requirements. The aortic arch curves leftward over the left main bronchus, while pulmonary arteries course under their respective main bronchi.
- Ascending aorta specifications
- Length: 5-6cm from aortic valve to arch
- Diameter: 3.0-3.5cm (normal adult)
- Wall thickness: 2-3mm (elastic fibers)
- Branches: Coronary arteries (RCA and LCA)
- Right coronary ostium: 1cm above aortic valve
- Left coronary ostium: 1.5cm above aortic valve
- Aortic arch configuration
- 3 major branches: Brachiocephalic, left carotid, left subclavian
- Arch diameter: 2.5-3.0cm (decreases with age)
- Ligamentum arteriosum: Connects to left pulmonary artery
- Recurrent laryngeal nerve: Loops under arch
- Pulmonary trunk anatomy
- Length: 3-4cm before bifurcation
- Diameter: 2.5-3.0cm (similar to aorta)
- Bifurcation level: T5-T6 (carina level)
- Pressure: 15-25 mmHg systolic, 8-12 mmHg diastolic
⭐ Clinical Pearl: Aortic root dilatation >4.0cm indicates aneurysm risk. Bicuspid aortic valve accelerates dilatation, requiring surgical intervention at 4.5cm versus 5.5cm for tricuspid valves.
Coronary Circulation: The Heart's Private Blood Supply
The coronary circulation delivers 250ml/min (5% of cardiac output) to the 300g heart muscle, demonstrating the highest oxygen extraction (70-80%) of any organ system. This high-demand circulation operates primarily during diastole when aortic pressure exceeds ventricular pressure.
- Right coronary artery (RCA) territory
- Origin: Right aortic sinus at 90-degree angle
- Length: 10-15cm total course
- Major branches: Acute marginal, posterior descending (PDA)
- Territory: Right ventricle, inferior wall, posterior wall
- Dominance: 85% of population (RCA gives PDA)
- Blood flow: 80ml/min (30% of coronary flow)
- Left coronary artery (LCA) system
- Left main: 1-2cm length, 4-5mm diameter
- LAD territory: Anterior wall, anterior septum
- Circumflex territory: Lateral wall, posterior wall (if dominant)
- Blood flow: 170ml/min (70% of coronary flow)
💡 Master This: Coronary dominance determines inferior wall blood supply. Right dominant (85%): RCA gives PDA and PLV. Left dominant (8%): Circumflex gives PDA. Co-dominant (7%): Both contribute to posterior circulation.
⭐ Clinical Pearl: Coronary steal phenomenon occurs when vasodilators redirect flow from stenotic to normal vessels. Dipyridamole stress testing exploits this principle, revealing >70% stenosis through perfusion defects.
Understanding cardiovascular architecture establishes the foundation for interpreting hemodynamic relationships. Connect these structural principles through pressure-volume relationships to understand cardiac performance optimization.
🔍 The Cardiovascular Command Matrix: Systematic Heart-Vessel Integration
⚖️ The Hemodynamic Optimization Engine: Evidence-Based Cardiac Performance
Cardiac Output Optimization: The Performance Framework
Cardiac output represents the ultimate measure of cardiac performance, calculated as Heart Rate × Stroke Volume. Normal values range 4-8 L/min with cardiac index (CO/BSA) of 2.5-4.0 L/min/m². Understanding the determinants and optimization strategies enables evidence-based management.
- Preload optimization (venous return)
- Optimal CVP: 8-12 mmHg (mechanically ventilated)
- PCWP target: 12-18 mmHg (left heart filling)
- Volume responsiveness: >15% stroke volume increase with 500ml bolus
- Frank-Starling mechanism: Optimal stretch at 2.2μm sarcomere length
- Underfilled: CVP <8 mmHg, flat pressure waveforms
- Overfilled: CVP >18 mmHg, elevated filling pressures
- Afterload management (vascular resistance)
- SVR normal: 800-1200 dynes·sec/cm⁵
- PVR normal: 150-250 dynes·sec/cm⁵
- Afterload reduction: 10-15% increase in stroke volume
- Vasopressor threshold: MAP <65 mmHg requires support
- Contractility enhancement
- Ejection fraction: >55% indicates normal contractility
- dP/dt max: >1200 mmHg/sec (invasive measure)
- Inotropic support: Dobutamine 2.5-10 μg/kg/min
- Calcium sensitizers: Levosimendan for heart failure
📌 Remember: SOAP for cardiac output determinants - Stroke volume (preload, afterload, contractility), Oxygen delivery, Arrhythmia control, Perfusion pressure (MAP >65 mmHg).
| Parameter | Normal Range | Optimization Target | Monitoring Method | Intervention Threshold |
|---|---|---|---|---|
| Cardiac Index | 2.5-4.0 L/min/m² | >2.5 L/min/m² | Thermodilution | <2.2 L/min/m² |
| CVP | 2-8 mmHg | 8-12 mmHg | Central line | <8 or >18 mmHg |
| PCWP | 6-12 mmHg | 12-18 mmHg | Swan-Ganz | <12 or >20 mmHg |
| SVR | 800-1200 | 800-1000 | Calculation | <600 or >1400 |
| SvO2 | 65-75% | >65% | Mixed venous | <60% |
Evidence-based cardiac management follows systematic algorithms that optimize outcomes through protocolized care. Goal-directed therapy reduces mortality by 15-20% compared to empirical management in critically ill patients.
- Volume responsiveness assessment
- Passive leg raise: >10% stroke volume increase predicts fluid responsiveness
- Pulse pressure variation: >13% indicates volume responsiveness (mechanical ventilation)
- Stroke volume variation: >10% suggests preload dependence
- IVC collapsibility: >50% collapse indicates volume depletion
- Vasopressor selection criteria
- Norepinephrine: First-line for distributive shock (0.1-3.0 μg/kg/min)
- Vasopressin: Second-line at 0.03-0.04 units/min (fixed dose)
- Epinephrine: Refractory shock or cardiac arrest (0.1-1.0 μg/kg/min)
- Phenylephrine: Pure vasoconstrictor for hypotension without cardiac dysfunction
- Inotropic therapy protocols
- Dobutamine: Beta-1 agonist, 2.5-10 μg/kg/min, increases contractility
- Milrinone: PDE-3 inhibitor, 0.375-0.75 μg/kg/min, inodilator effect
- Levosimendan: Calcium sensitizer, 0.1-0.2 μg/kg/min, 24-hour half-life
- Digoxin: Na-K ATPase inhibitor, 0.25mg daily, chronic heart failure
⭐ Clinical Pearl: Early goal-directed therapy within 6 hours reduces septic shock mortality from 46% to 30%. ScvO2 >70%, MAP >65 mmHg, and urine output >0.5ml/kg/hr indicate adequate resuscitation.
Monitoring and Outcomes: The Performance Dashboard
Advanced hemodynamic monitoring enables real-time optimization and outcome prediction. Pulmonary artery catheters provide comprehensive data but require expertise for appropriate interpretation and clinical application.
- Invasive monitoring parameters
- Cardiac output: Thermodilution or Fick method
- Mixed venous saturation: SvO2 >65% indicates adequate oxygen delivery
- Pulmonary vascular resistance: PVR = (MPAP-PCWP)/CO × 80
- Systemic vascular resistance: SVR = (MAP-CVP)/CO × 80
- Non-invasive alternatives
- Echocardiography: Real-time assessment of ventricular function
- Pulse contour analysis: Continuous cardiac output monitoring
- Bioimpedance: Non-invasive stroke volume measurement
- Near-infrared spectroscopy: Tissue oxygenation monitoring
💡 Master This: Hemodynamic optimization requires systematic approach: 1) Ensure adequate preload, 2) Optimize afterload, 3) Enhance contractility if needed, 4) Control heart rate and rhythm. Sequential optimization prevents conflicting interventions.
⭐ Clinical Pearl: Cardiac power output (MAP × CO / 451) provides superior prognostic information compared to ejection fraction alone. CPO <0.6 watts indicates high mortality risk in cardiogenic shock.
Understanding hemodynamic optimization principles provides the foundation for advanced cardiac care. Connect these performance metrics through multi-system integration to understand comprehensive thoracic physiology.
⚖️ The Hemodynamic Optimization Engine: Evidence-Based Cardiac Performance
🔗 The Thoracic Integration Matrix: Multi-System Orchestration Mastery
Cardiopulmonary Integration: The Synchronized Performance Engine
The heart and lungs function as an integrated unit where mechanical, electrical, and chemical interactions optimize oxygen delivery and carbon dioxide removal. Ventricular interdependence demonstrates how right heart function directly affects left heart performance through shared septal mechanics.
- Respiratory-cardiac coupling
- Inspiratory venous return: Increases by 20-30% during inspiration
- Ventricular interdependence: Septal shift affects opposite ventricle
- Pulsus paradoxus: >10 mmHg indicates severe respiratory distress
- Heart-lung interactions: Positive pressure ventilation reduces venous return
- PEEP effects: 5 cmH2O reduces cardiac output by 10-15%
- Preload optimization: Volume loading counteracts PEEP effects
- Ventilation-perfusion matching
- V/Q ratio: 0.8 in healthy lungs (ventilation 4L/min, perfusion 5L/min)
- Zone 1 (apex): PA > Pa > Pv (minimal perfusion)
- Zone 2 (middle): Pa > PA > Pv (waterfall effect)
- Zone 3 (base): Pa > Pv > PA (optimal matching)
- Hypoxic vasoconstriction: Redirects blood from poorly ventilated areas
- Shunt fraction: <5% normal, >20% indicates severe disease
- Integrated oxygen transport
- Oxygen delivery: DO2 = CO × CaO2 × 10 (normal 900-1200 ml/min)
- Oxygen consumption: VO2 = CO × (CaO2-CvO2) × 10 (normal 200-300 ml/min)
- Extraction ratio: O2ER = VO2/DO2 (normal 20-30%)
- Critical DO2: <400 ml/min causes supply-dependent consumption
📌 Remember: VIPER for V/Q relationships - Ventilation 4L/min, Ideal ratio 0.8, Perfusion 5L/min, Exchange optimization, Redistribution with disease.
| Integration Parameter | Normal Value | Pathological Threshold | Clinical Significance | Monitoring Method |
|---|---|---|---|---|
| V/Q Ratio | 0.8 | <0.6 or >1.2 | Mismatch severity | ABG analysis |
| Shunt Fraction | <5% | >20% | Severe disease | 100% O2 test |
| DO2 | 900-1200 ml/min | <400 ml/min | Critical threshold | CO × CaO2 |
| O2ER | 20-30% | >40% | Inadequate delivery | SvO2 monitoring |
| Pulsus Paradoxus | <10 mmHg | >20 mmHg | Severe obstruction | Blood pressure |
The autonomic nervous system provides real-time regulation of cardiopulmonary function through sympathetic and parasympathetic pathways that respond to chemical, mechanical, and emotional stimuli within milliseconds.
- Sympathetic cardiopulmonary control
- Cardiac sympathetics: T1-T5 segments increase heart rate and contractility
- Pulmonary sympathetics: Bronchodilation and pulmonary vasoconstriction
- Stress response: Epinephrine increases cardiac output by 50-100%
- Baroreceptor reflex: Aortic arch and carotid sinus detect pressure changes
- Response time: 1-3 seconds for heart rate adjustment
- Sensitivity: 1 mmHg pressure change triggers reflex
- Parasympathetic regulation
- Vagal control: Heart rate reduction via muscarinic receptors
- Respiratory sinus arrhythmia: Heart rate varies with breathing
- Diving reflex: Bradycardia and peripheral vasoconstriction
- Vagal tone: High variability indicates healthy autonomic function
- Chemical control integration
- Central chemoreceptors: Medulla responds to CSF pH changes
- Peripheral chemoreceptors: Carotid bodies detect PO2 <60 mmHg
- Hypercapnic drive: Primary respiratory stimulus (responds to CO2)
- Hypoxic drive: Secondary stimulus, becomes primary in COPD
Lymphatic-Immune Integration: The Thoracic Defense Network
The thoracic lymphatic system processes 2-4 liters of lymph daily, filtering antigens and maintaining fluid balance. The thoracic duct represents the largest lymphatic vessel, collecting 75% of body lymph and returning it to venous circulation.
- Thoracic duct specifications
- Length: 38-45cm from cisterna chyli to left subclavian vein
- Diameter: 2-5mm (varies with location)
- Flow rate: 1-3 ml/min (1.5-4.3 L/day)
- Pressure: 5-10 mmHg (higher than venous pressure)
- Valves: Prevent backflow every 2-3cm
- Smooth muscle: Intrinsic contractions propel lymph
- Mediastinal lymph nodes
- Paratracheal nodes: 2R, 4R (right), 2L, 4L (left)
- Subcarinal nodes: Station 7 (critical for lung cancer staging)
- Hilar nodes: Station 10 (primary lung drainage)
- Drainage patterns: Upper lobes → hilar → mediastinal → supraclavicular
- Immune surveillance integration
- Antigen presentation: Dendritic cells migrate to lymph nodes
- T-cell activation: Requires antigen presentation in lymphoid tissue
- Antibody production: B-cells differentiate in germinal centers
- Memory formation: Long-lived immune cells provide protection
⭐ Clinical Pearl: Thoracic duct injury during esophageal surgery causes chylothorax with >1000ml/day milky drainage. Conservative management succeeds in 70% of cases with medium-chain triglyceride diet.
💡 Master This: Multi-system failure follows predictable patterns. Respiratory failure leads to hypoxemia, triggering sympathetic activation, pulmonary vasoconstriction, right heart strain, and eventual cardiovascular collapse. Early intervention breaks this cascade.
Understanding thoracic integration principles reveals how system interactions create clinical syndromes. Connect these multi-system relationships through rapid assessment tools to develop clinical mastery frameworks.
🔗 The Thoracic Integration Matrix: Multi-System Orchestration Mastery
🎯 The Thoracic Mastery Arsenal: Rapid Clinical Command Tools
The Essential Numbers Arsenal: Critical Thresholds for Instant Recall
Master clinicians operate with quantitative precision, knowing critical thresholds that determine immediate interventions. These evidence-based values guide emergency decisions and optimize outcomes through systematic application.
- Respiratory emergency thresholds
- Tension pneumothorax: >2cm tracheal deviation, immediate decompression
- Massive hemothorax: >1500ml initial drainage or >200ml/hr ongoing
- Respiratory failure: PaO2 <60 mmHg or PaCO2 >50 mmHg with pH <7.30
- Pleural effusion: >500ml causes dyspnea, >1500ml requires drainage
- Light's criteria: Protein ratio >0.5, LDH ratio >0.6 = exudate
- Empyema: pH <7.30, glucose <60 mg/dL, LDH >1000 U/L
- Cardiovascular critical values
- Cardiogenic shock: Cardiac index <2.2 L/min/m², PCWP >18 mmHg
- Tamponade: Pulsus paradoxus >20 mmHg, equalized pressures
- Aortic dissection: Mediastinal width >8cm, blood pressure differential >20 mmHg
- Pulmonary embolism: RV/LV ratio >0.9, PA pressure >40 mmHg
- Procedural safety limits
- Chest tube insertion: 5th intercostal space, mid-axillary line
- Central line depth: 15-17cm right IJ, 18-20cm left IJ
- Mechanical ventilation: Plateau pressure <30 cmH2O, PEEP 5-15 cmH2O
- Thoracentesis: <1500ml single session, <1000ml if no manometry
📌 Remember: THORAX emergency numbers - Tension pneumo >2cm shift, Hemothorax >1500ml, Oxygen <60 mmHg, Respiratory failure pH <7.30, Aortic dissection >8cm width, X-ray >20 mmHg pulsus paradoxus.
| Emergency Condition | Critical Threshold | Immediate Action | Time Window | Success Rate |
|---|---|---|---|---|
| Tension Pneumothorax | >2cm tracheal shift | Needle decompression | <5 minutes | >95% |
| Massive Hemothorax | >1500ml drainage | Thoracotomy | <30 minutes | 70-85% |
| Cardiac Tamponade | Pulsus >20 mmHg | Pericardiocentesis | <15 minutes | 85-95% |
| Aortic Dissection | Mediastinal >8cm | BP control <140 | <10 minutes | 80-90% |
| Massive PE | RV/LV >1.0 | Thrombolysis | <60 minutes | 60-80% |
Clinical excellence requires systematic approaches that ensure comprehensive evaluation while minimizing errors. The THORAX protocol provides structured assessment for thoracic emergencies and routine evaluations.
- T - Tracheal position and patency
- Midline position: Equidistant from sternal notch to clavicles
- Deviation significance: >1cm suggests mediastinal shift
- Palpable fremitus: Transmitted vibrations indicate patent airways
- Stridor assessment: Inspiratory = upper airway, expiratory = lower airway
- H - Heart sounds and hemodynamics
- S1/S2 intensity: Muffled suggests pericardial effusion
- S3 gallop: Ventricular dysfunction (sensitivity 85%)
- Murmur timing: Systolic vs diastolic significance
- Blood pressure: Both arms for aortic dissection screening
- O - Oxygenation and ventilation
- Pulse oximetry: >95% normal, <90% requires intervention
- Respiratory rate: 12-20/min normal, >30/min indicates distress
- Work of breathing: Accessory muscles, retractions, paradox
- Capnography: 35-45 mmHg normal ETCO2
- R - Respiratory mechanics
- Chest expansion: Symmetric 3-5cm excursion
- Percussion: Resonant normal, dull = fluid, hyperresonant = air
- Auscultation: Vesicular normal, absent = pneumothorax/effusion
- Tactile fremitus: Increased = consolidation, decreased = effusion
- A - Arterial blood gas interpretation
- pH: 7.35-7.45 (acidosis <7.35, alkalosis >7.45)
- PaCO2: 35-45 mmHg (respiratory component)
- HCO3: 22-26 mEq/L (metabolic component)
- A-a gradient: <15 mmHg normal, >25 mmHg abnormal
- X - X-ray systematic review
- ABCDE approach: Airway, Bones, Cardiac, Diaphragm, Everything else
- Cardiac silhouette: <50% thoracic width normal
- Hilum assessment: Left higher than right normally
- Costophrenic angles: Sharp normally, blunted = effusion
Rapid Intervention Decision Trees: The Action Framework
Clinical mastery requires immediate decision-making based on pattern recognition and evidence-based protocols. These decision trees guide rapid interventions that optimize outcomes through systematic approaches.
- Dyspnea evaluation pathway
- Acute onset (<24 hours): PE, pneumothorax, MI, pneumonia
- Subacute (days-weeks): Heart failure, pleural effusion, anemia
- Chronic (months): COPD, ILD, pulmonary hypertension
- Positional: Orthopnea = heart failure, platypnea = liver disease
- Chest pain differentiation
- Pleuritic (sharp, positional): PE, pneumothorax, pericarditis
- Crushing (pressure): MI, aortic dissection, esophageal rupture
- Tearing (sudden, severe): Aortic dissection, pneumothorax
- Burning (substernal): GERD, esophagitis, peptic ulcer
⭐ Clinical Pearl: The 1-3-5 rule for chest tube management: 1cm above rib, 3 clamps maximum, 5cm water seal depth. Persistent air leak >5 days requires surgical evaluation.
💡 Master This: Clinical pattern recognition develops through systematic practice. See pneumothorax, think tension if hemodynamically unstable. See mediastinal widening, think dissection if chest pain. See elevated JVP, think tamponade if hypotensive.
⭐ Clinical Pearl: Thoracic emergencies follow the "Golden Hour" principle. Tension pneumothorax requires decompression within 5 minutes, massive hemothorax needs surgery within 30 minutes, and aortic dissection demands blood pressure control within 10 minutes for optimal outcomes.
🎯 The Thoracic Mastery Arsenal: Rapid Clinical Command Tools
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















