Head

On this page
🏗️ Cranial Architecture: The Skull's Engineering Marvel
The skull is far more than a protective helmet-it's a precisely engineered vault where bone architecture, growth mechanisms, and strategic openings orchestrate brain protection, sensory function, and vascular supply. You'll discover how sutures enable cranial expansion, how dozens of foramina channel critical nerves and vessels, how three distinct fossae compartmentalize brain regions, and how dural layers create both barriers and potential spaces for pathology. Mastering this anatomy transforms abstract bone landmarks into a clinical roadmap for diagnosing fractures, bleeds, and nerve injuries with confidence and precision.
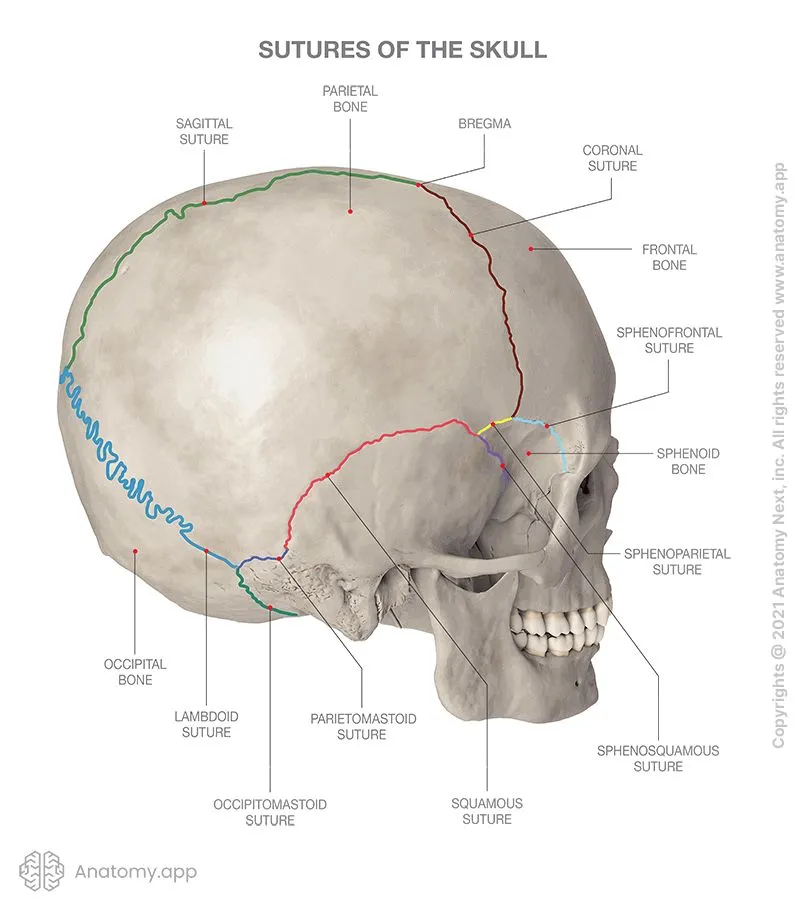
Neurocranium: The Protective Vault
The neurocranium houses the brain within 8 primary bones forming a 1,400-1,500 mL capacity vault. The frontal bone contributes 40% of anterior protection, while the parietal bones provide 60% of lateral coverage. Temporal bones contain the middle ear apparatus and house 85% of skull base foramina.
-
Frontal Bone Architecture
- Frontal squama: vertical protective wall with 6-8mm thickness
- Orbital plates: horizontal shelves forming 75% of orbital roof
- Frontal sinuses: air-filled spaces reducing weight by 15%
- Adult capacity: 4-7 mL per sinus
- Drainage angle: 45-60 degrees via frontonasal duct
-
Parietal Bone Engineering
- Maximum thickness: 7mm at parietal eminence
- Minimum thickness: 4mm at temporal lines
- Growth pattern: centrifugal from ossification centers
- Anterior center appears at week 8 of gestation
- Posterior center appears at week 16 of gestation
-
Temporal Bone Complexity
- Squamous part: 2-4mm thick, most vulnerable to fracture
- Petrous part: 8-12mm thick, hardest bone in body
- Mastoid process: 20-25 air cells reducing weight
- Pneumatization completes by age 6-8 years
- Total air volume: 6-14 mL per temporal bone
📌 Remember: STOP for temporal bone parts - Squamous, Tympanic, Occipital (mastoid), Petrous. Each part has distinct thickness: Squamous (2-4mm), Petrous (8-12mm), Mastoid (pneumatized), Tympanic (1-2mm).
Viscerocranium: The Facial Framework
The facial skeleton comprises 14 bones creating the respiratory, digestive, and sensory apparatus framework. The maxilla contributes 70% of midface structure, while the mandible provides 100% of lower facial support with 2.5x the bite force capacity of other facial bones.
-
Maxillary Architecture
- Body dimensions: 35mm height × 25mm width × 30mm depth
- Sinus capacity: 15-20 mL reducing facial weight by 25%
- Alveolar process: supports 8 teeth with 12-15mm bone height
- Cortical thickness: 1-3mm buccal, 0.5-2mm palatal
- Cancellous density: 0.5-0.8 g/cm³ varying by region
-
Mandibular Engineering
- Body height: 25-35mm at mental foramen level
- Ramus height: 50-70mm from angle to condyle
- Bite force generation: 150-200 pounds at molars
- Cortical thickness: 2-4mm at body, 1-2mm at ramus
- Trabecular pattern: horizontal in body, vertical in ramus
⭐ Clinical Pearl: Mandibular fractures occur at predictable stress points - 36% at angle, 20% at body, 20% at condyle, 14% at symphysis, 10% at ramus. Le Fort fracture patterns follow horizontal weakness planes at specific anatomical levels.
| Bone Component | Thickness (mm) | Key Function | Clinical Significance | Fracture Risk |
|---|---|---|---|---|
| Frontal squama | 6-8 | Brain protection | Frontal sinus involvement | Moderate |
| Parietal eminence | 7 | Maximum protection | Temporal muscle attachment | Low |
| Temporal squamous | 2-4 | Lateral coverage | Thinnest cranial area | High |
| Occipital squama | 10-15 | Posterior protection | Venous sinus proximity | Low |
| Maxillary body | 1-3 | Midface support | Sinus communication | High |
The cranial vault's engineering principles establish the foundation for understanding intracranial pressure dynamics and surgical approach planning. Connect these architectural concepts through suture biomechanics to understand growth patterns and pathological expansion mechanisms.
🏗️ Cranial Architecture: The Skull's Engineering Marvel
⚡ Suture Dynamics: The Skull's Growth Engines
Primary Suture Architecture
The 5 major sutures create interlocking seams allowing controlled expansion while maintaining protective integrity. Coronal sutures contribute 40% of anteroposterior growth, while sagittal sutures enable 60% of biparietal expansion. Lambdoid sutures provide posterior growth accommodation with 15-20 degrees of angular flexibility.
-
Coronal Suture Engineering
- Location: frontal-parietal junction at bregma level
- Fusion timeline: 24-26 years from anterior to posterior
- Growth contribution: 6-8mm annually until age 7
- Peak expansion: birth to 2 years (12mm/year)
- Maintenance phase: 2-7 years (4-6mm/year)
- Consolidation: 7-26 years (1-2mm/year)
-
Sagittal Suture Dynamics
- Length: 12-15cm from bregma to lambda
- Fusion pattern: posterior to anterior over 20-30 years
- Biparietal expansion: 8-10mm annually in first 2 years
- Maximum width: birth (9-10cm) to adult (14-15cm)
- Growth rate: anterior 1/3 fastest (60% of expansion)
- Posterior 1/3: 40% of total biparietal growth
-
Lambdoid Suture Mechanics
- Configuration: inverted V-shape with 120-130 degree angle
- Fusion timeline: 26-30 years bilateral progression
- Occipital growth: posterior expansion 4-6mm annually
- Wormian bones: 60-80% of normal skulls
- Inca bones: 5-15% frequency in lambdoid region
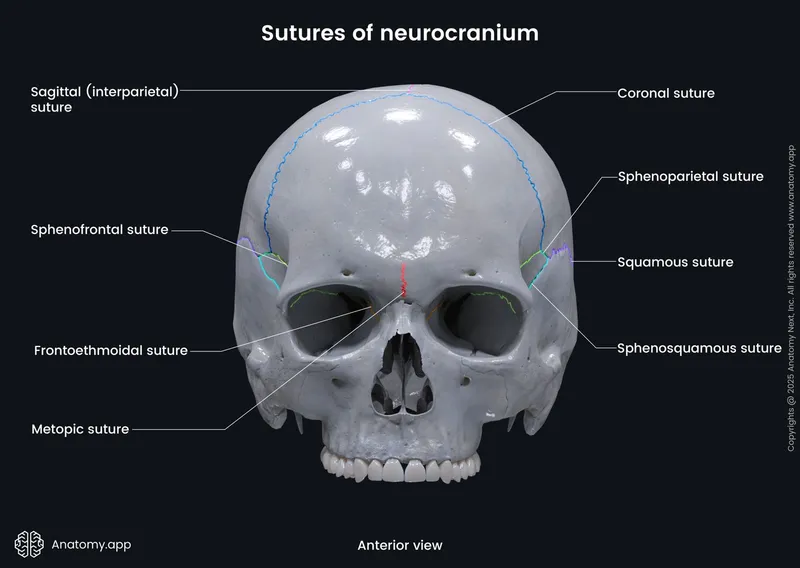
📌 Remember: CALM for suture fusion timing - Coronal (24-26 years), Anterior fontanelle (12-18 months), Lambdoid (26-30 years), Metopic (6-8 months). Early fusion creates craniosynostosis with predictable deformity patterns.
Fontanelle Function and Clinical Significance
Fontanelles represent membranous spaces allowing rapid brain expansion during critical growth periods. The anterior fontanelle accommodates 75% of first-year brain growth, expanding from 3-4cm at birth to peak size at 6 months before gradual closure.
-
Anterior Fontanelle Dynamics
- Dimensions: diamond-shaped 3-4cm × 2-3cm at birth
- Peak expansion: 4-5cm × 3-4cm at 6 months
- Closure timeline: 12-18 months with centripetal pattern
- Normal pressure: 5-15 mmHg in upright position
- Bulging threshold: >20 mmHg indicates increased ICP
- Depression significance: <0 mmHg suggests dehydration
-
Posterior Fontanelle Architecture
- Shape: triangular with 1-2cm base dimension
- Closure: 6-8 weeks postnatal (earliest fontanelle)
- Clinical access: limited due to small size and early closure
- Palpation window: birth to 6 weeks only
- Pressure correlation: less reliable than anterior fontanelle
⭐ Clinical Pearl: Fontanelle tension correlates with intracranial pressure in 85% of cases. Bulging fontanelle with >20 mmHg pressure indicates increased ICP requiring immediate evaluation. Sunken fontanelle suggests dehydration with >5% fluid loss.
| Suture/Fontanelle | Fusion Age | Growth Contribution | Clinical Significance | Pathology Risk |
|---|---|---|---|---|
| Metopic | 6-8 months | Frontal width expansion | Trigonocephaly if early | High |
| Coronal | 24-26 years | Anteroposterior growth | Brachycephaly if early | Moderate |
| Sagittal | 20-30 years | Biparietal expansion | Scaphocephaly if early | High |
| Lambdoid | 26-30 years | Posterior growth | Plagiocephaly if unilateral | Low |
| Anterior fontanelle | 12-18 months | Rapid brain expansion | ICP monitoring window | Critical |
Understanding suture biomechanics provides the framework for recognizing abnormal cranial development patterns. Connect these growth dynamics through foraminal anatomy to understand neurovascular pathway development and surgical landmark identification.
⚡ Suture Dynamics: The Skull's Growth Engines
🚪 Foraminal Highways: The Skull's Neurovascular Gateways
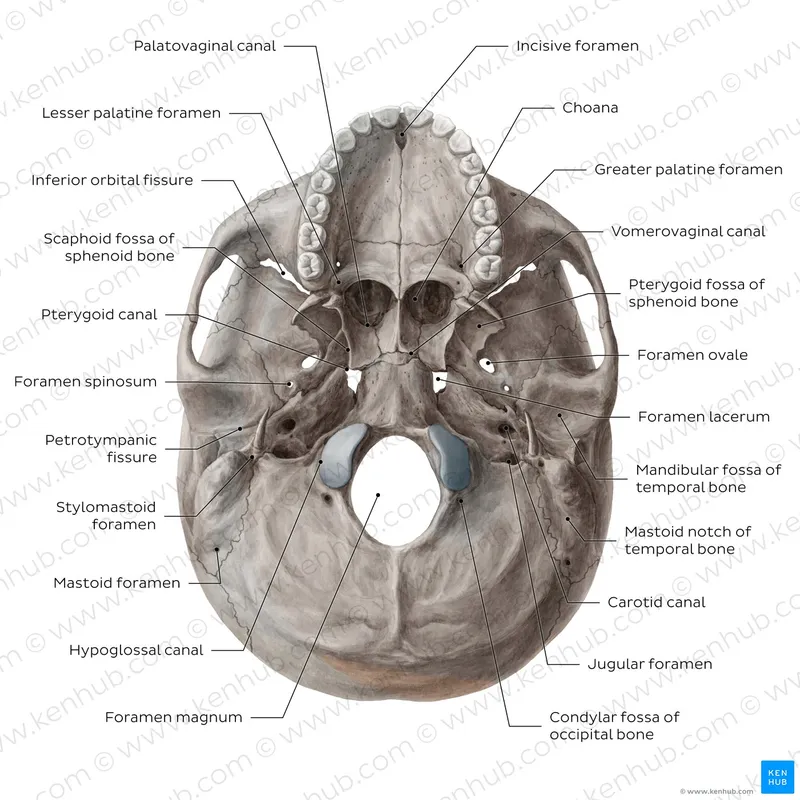
Anterior Cranial Fossa Openings
The anterior fossa contains 15+ foramina primarily serving olfactory and optic pathways. The cribriform plate houses 20-25 olfactory foramina allowing CN I passage, while the optic canal provides protected transit for CN II and ophthalmic artery through 4-6mm diameter openings.
-
Cribriform Plate Architecture
- Olfactory foramina: 20-25 openings per side
- Foramen diameter: 0.5-1mm each opening
- Clinical vulnerability: CSF leak risk with 15-20% incidence in frontal trauma
- Anosmia correlation: damage to >75% of olfactory fibers
- Meningitis risk: direct CNS access via nasal cavity
- Repair success: 85-90% with early intervention
-
Optic Canal Dynamics
- Dimensions: 4-6mm diameter × 8-10mm length
- Contents: optic nerve (3mm diameter) + ophthalmic artery (1-2mm)
- Angulation: 45-degree medial inclination toward sella
- Compression threshold: >20% diameter reduction causes visual deficits
- Surgical access: lateral orbitotomy or transcranial approaches
- Decompression success: 70-80% if performed within 24-48 hours
📌 Remember: SOME for anterior fossa contents - Sphenoidal sinus, Olfactory (CN I), Meningeal vessels, Ethmoid air cells. Trauma here causes anosmia (CN I), visual loss (CN II), and CSF rhinorrhea (cribriform fractures).
Middle Cranial Fossa Pathways
The middle fossa houses the most clinically significant foramina with 8 major openings serving CN II-VI and critical vascular structures. The foramen ovale transmits CN V3 through 7-8mm opening, while the foramen rotundum provides CN V2 passage through 3-4mm diameter.
-
Foramen Ovale Engineering
- Dimensions: oval-shaped 7-8mm × 4-5mm
- Contents: mandibular nerve (CN V3) + accessory meningeal artery
- Clinical access: percutaneous approach for trigeminal neuralgia
- Success rate: 85-90% for glycerol rhizotomy
- Complication rate: <5% with experienced operators
- Anatomical variation: 15-20% show foramen Vesalius proximity
-
Foramen Rotundum Architecture
- Shape: round opening 3-4mm diameter
- Location: greater sphenoid wing at pterygopalatine fossa junction
- Contents: maxillary nerve (CN V2) exclusively
- Surgical approach: transpterygoid or transantral
- Nerve block success: 90-95% for maxillary anesthesia
- Anatomical consistency: >95% reliable landmark
-
Superior Orbital Fissure Complexity
- Dimensions: 22-25mm length × 2-6mm width (variable)
- Contents: CN III, IV, V1, VI + ophthalmic vessels
- Compartmentalization: lateral and medial divisions
- Lateral compartment: CN IV, V1 (lacrimal, frontal)
- Central compartment: CN III (superior, inferior divisions)
- Medial compartment: CN VI, V1 (nasociliary)
⭐ Clinical Pearl: Superior orbital fissure syndrome affects CN III, IV, V1, VI causing complete ophthalmoplegia + V1 anesthesia. Foramen ovale provides percutaneous access for trigeminal neuralgia treatment with 85-90% success rates using glycerol rhizotomy or radiofrequency ablation.
Posterior Cranial Fossa Gateways
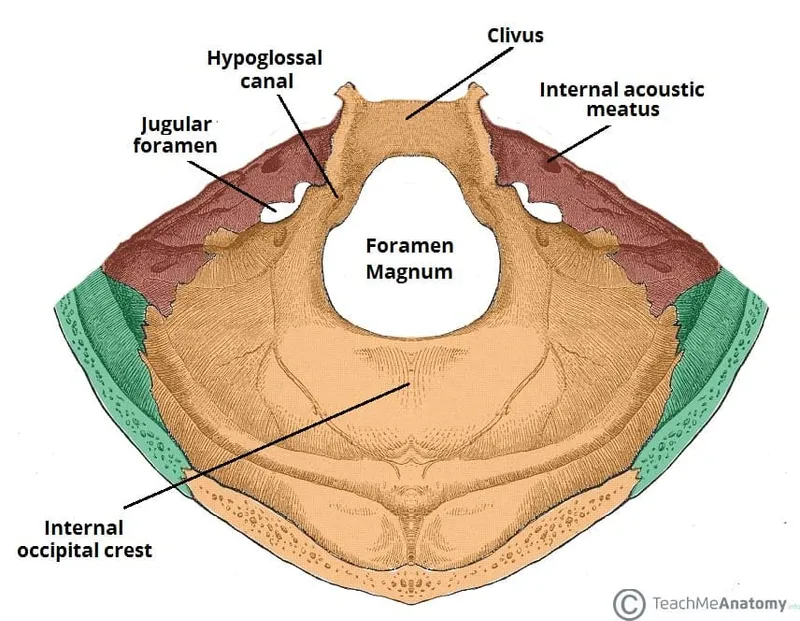
The posterior fossa contains the largest foramina including foramen magnum (35mm × 30mm) and jugular foramina (10-15mm diameter). These openings accommodate major neural and vascular structures including brainstem, vertebral arteries, and CN IX-XII.
-
Foramen Magnum Architecture
- Dimensions: oval 35mm anteroposterior × 30mm transverse
- Contents: medulla oblongata, vertebral arteries, spinal accessory nerves
- Clinical significance: tonsillar herniation threshold at 5mm descent
- Normal CSF flow: 500-750 mL/day through opening
- Compression effects: >20% reduction causes syringomyelia
- Surgical decompression: suboccipital craniectomy with C1 laminectomy
-
Jugular Foramen Complexity
- Compartments: anterior (pars nervosa) + posterior (pars vascularis)
- Anterior contents: CN IX + tympanic branch
- Posterior contents: CN X, XI + jugular bulb + sigmoid sinus
- Surgical approach: retrosigmoid or transcondylar
- Tumor involvement: paragangliomas in 60-70% of cases
- Functional preservation: >80% with careful dissection
| Foramen | Diameter (mm) | Primary Contents | Clinical Access | Pathology Risk |
|---|---|---|---|---|
| Cribriform | 0.5-1 each | CN I (olfactory) | Endoscopic | CSF leak (15-20%) |
| Optic canal | 4-6 | CN II + ophthalmic artery | Orbitotomy | Compression (20%) |
| Superior orbital fissure | 2-6 width | CN III,IV,V1,VI | Orbital approach | Syndrome (5-10%) |
| Foramen rotundum | 3-4 | CN V2 (maxillary) | Transpterygoid | Block failure (5%) |
| Foramen ovale | 7-8 × 4-5 | CN V3 (mandibular) | Percutaneous | Variation (15-20%) |
Foraminal mastery establishes the foundation for cranial nerve examination interpretation and surgical approach selection. Connect these neurovascular pathways through cranial fossa anatomy to understand three-dimensional relationships and clinical correlation patterns.
🚪 Foraminal Highways: The Skull's Neurovascular Gateways
🏛️ Cranial Fossa Territories: The Brain's Three-Level Penthouse
Anterior Cranial Fossa: The Frontal Command Center
The anterior fossa houses frontal lobes and olfactory apparatus within a shallow depression spanning 8-10cm anteroposteriorly. This level accommodates executive function and smell processing with specialized bony architecture supporting 1,400-1,600g of frontal brain tissue.
-
Architectural Boundaries
- Anterior: frontal bone inner table with 6-8mm thickness
- Posterior: lesser sphenoid wing and anterior clinoid processes
- Floor: orbital plates + cribriform plate + ethmoid roof
- Total surface area: 45-50 cm² per hemisphere
- Maximum depth: 15-20mm below Frankfurt horizontal
- Load distribution: frontal bone bears 60% of anterior brain weight
-
Functional Specialization
- Brain contents: frontal lobes (prefrontal cortex, motor areas)
- Vascular supply: anterior cerebral arteries (A1-A2 segments)
- Venous drainage: superior sagittal sinus (anterior 1/3)
- CSF production: anterior horns contribute 15-20% of total
- Perfusion pressure: 60-80 mmHg in anterior circulation
- Metabolic demand: 20-25% of total brain glucose consumption
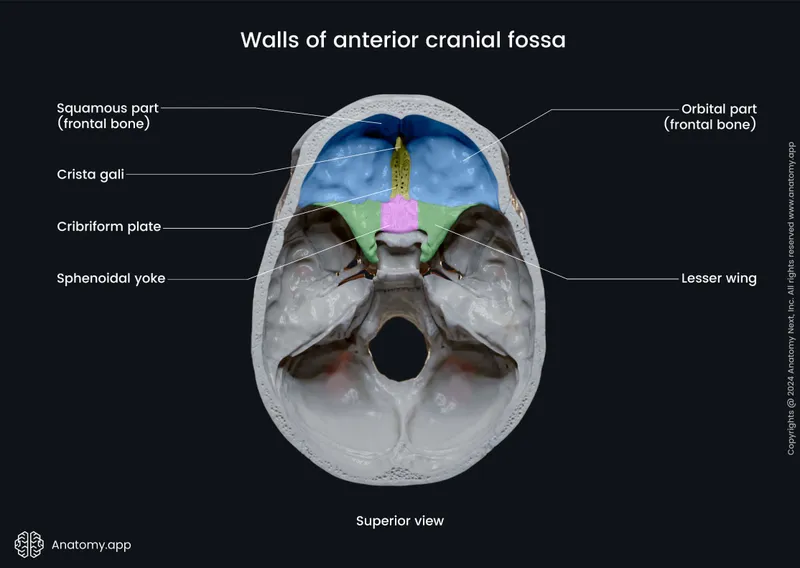
📌 Remember: FACE for anterior fossa contents - Frontal lobes, Anterior cerebral arteries, Cribriform plate (CN I), Ethmoid air cells. Trauma here causes personality changes, anosmia, and CSF rhinorrhea.
Middle Cranial Fossa: The Temporal Processing Hub
The middle fossa represents the deepest and most complex cranial level, housing temporal lobes and critical neurovascular structures within butterfly-shaped depressions. Each temporal fossa accommodates 800-1,000g of brain tissue with maximum depth of 55-65mm below the temporal squama.
-
Geometric Architecture
- Shape: butterfly configuration with central sphenoid body
- Lateral walls: temporal squama (2-4mm thickness - weakest point)
- Floor: greater sphenoid wing + temporal bone (petrous pyramid)
- Surface area: 65-75 cm² per side (largest fossa)
- Volume capacity: 180-220 mL per temporal fossa
- Structural support: sphenoid bears 70% of middle fossa load
-
Neurovascular Complexity
- Brain contents: temporal lobes (hippocampus, amygdala, auditory cortex)
- Major vessels: middle cerebral arteries (M1-M4 segments)
- Cranial nerves: CN II-VI traverse 8 major foramina
- Perfusion territory: MCA supplies 60% of cerebral cortex
- Venous drainage: sphenoparietal + cavernous sinuses
- CSF circulation: temporal horns + basal cisterns
-
Clinical Vulnerability Zones
- Temporal squama: thinnest bone (2-4mm) - epidural hematoma risk
- Cavernous sinus: CN III-VI + ICA - thrombosis complications
- Petrous apex: CN V-VIII - petrosal syndrome potential
- Fracture incidence: temporal bone in 40% of skull trauma
- Surgical access: pterional, subtemporal, transpetrosal approaches
- Complication rates: CN deficits in 15-25% of middle fossa surgery
⭐ Clinical Pearl: Middle cranial fossa contains 60% of clinically significant foramina and the weakest skull bone (temporal squama). Epidural hematomas occur in 85% of temporal bone fractures due to middle meningeal artery laceration. Cavernous sinus thrombosis affects CN III-VI causing complete ophthalmoplegia.
Posterior Cranial Fossa: The Brainstem Sanctuary
The posterior fossa houses brainstem, cerebellum, and CN VIII-XII within the largest and deepest cranial compartment. This level accommodates 150-180g of brainstem tissue and 140-150g of cerebellar tissue with maximum depth of 70-80mm below the external occipital protuberance.
-
Protective Architecture
- Boundaries: occipital bone (10-15mm thickness - strongest)
- Floor: foramen magnum (35mm × 30mm) - largest opening
- Roof: tentorium cerebelli - dural partition separating supra/infratentorial
- Volume capacity: 280-320 mL (largest fossa)
- Bone thickness: maximum protection for vital centers
- Structural integrity: occipital withstands >3,000 pounds force
-
Vital Function Centers
- Brainstem: medulla, pons, midbrain - cardiorespiratory control
- Cerebellum: motor coordination + cognitive processing
- Cranial nerves: CN VIII-XII - special senses + autonomic functions
- Blood supply: vertebrobasilar system (20% of cardiac output)
- CSF production: 4th ventricle (25% of total CSF)
- Perfusion pressure: 80-100 mmHg (posterior circulation)
| Cranial Fossa | Depth (mm) | Volume (mL) | Primary Contents | Bone Thickness | Clinical Risk |
|---|---|---|---|---|---|
| Anterior | 15-20 | 120-150 | Frontal lobes, CN I | 6-8mm | CSF leak, anosmia |
| Middle | 55-65 | 180-220 | Temporal lobes, CN II-VI | 2-4mm | Epidural hematoma |
| Posterior | 70-80 | 280-320 | Brainstem, cerebellum, CN VIII-XII | 10-15mm | Herniation, death |
Cranial fossa mastery provides the three-dimensional framework for understanding intracranial relationships and surgical approaches. Connect these territorial concepts through dural anatomy to understand protective membrane systems and venous drainage patterns.
🏛️ Cranial Fossa Territories: The Brain's Three-Level Penthouse
🛡️ Dural Defense Systems: The Brain's Protective Membrane Network
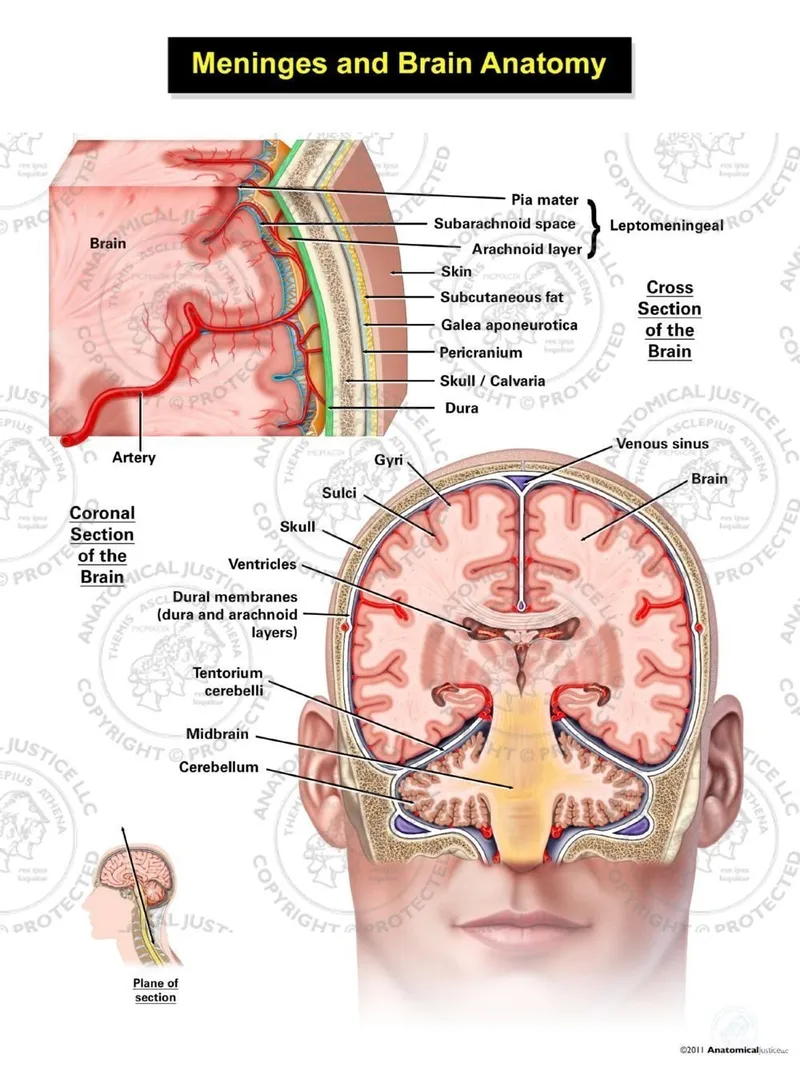
Dural Architecture and Compartmentalization
The dura mater creates 4 major compartments through fibrous partitions that limit mass effect and herniation patterns. The falx cerebri divides cerebral hemispheres, while the tentorium cerebelli separates supratentorial and infratentorial spaces with critical pressure thresholds.
-
Falx Cerebri Engineering
- Dimensions: sickle-shaped 15-18cm length × 6-8cm maximum height
- Attachments: crista galli to internal occipital protuberance
- Venous integration: superior and inferior sagittal sinuses
- Thickness variation: anterior (2-3mm) to posterior (8-10mm)
- Herniation prevention: limits subfalcine displacement to <5mm
- Surgical landmark: midline reference for hemispheric procedures
-
Tentorium Cerebelli Dynamics
- Configuration: tent-shaped with tentorial notch (25-30mm diameter)
- Function: separates cerebrum from cerebellum + brainstem
- Critical opening: tentorial hiatus for brainstem passage
- Herniation threshold: >5mm uncal displacement causes CN III palsy
- Pressure gradient: supratentorial vs infratentorial >10 mmHg = danger
- Surgical access: subtentorial vs supratentorial approaches
-
Falx Cerebelli Architecture
- Size: small triangular partition 2-3cm height
- Location: between cerebellar hemispheres in posterior fossa
- Clinical significance: minimal due to small size
- Venous drainage: occipital sinus (often absent)
- Herniation: rare due to limited compartmentalization
📌 Remember: FIT for dural partitions - Falx cerebri (hemispheric separation), Incisura tentorialis (brainstem passage), Tentorium cerebelli (supra/infratentorial division). Each creates herniation syndromes with specific clinical patterns.
Venous Sinus Networks and Drainage Patterns
Dural venous sinuses represent endothelial-lined channels within dural layers providing cerebral venous drainage for 500-750 mL/min of cerebral blood flow. The superior sagittal sinus drains 60% of cerebral cortex, while transverse sinuses handle 40% through deep venous systems.
-
Superior Sagittal Sinus Dynamics
- Course: anterior (falx attachment) to confluence (15-18cm)
- Drainage pattern: cortical veins enter at acute angles
- Flow characteristics: laminar with minimal turbulence
- Diameter progression: 2-3mm (anterior) to 8-12mm (posterior)
- Flow velocity: 15-25 cm/sec with cardiac pulsatility
- Pressure: 5-12 mmHg (slightly above venous pressure)
-
Transverse-Sigmoid Sinus Complex
- Configuration: transverse (horizontal) to sigmoid (S-shaped)
- Drainage territory: temporal, parietal, occipital regions
- Jugular connection: sigmoid continues as internal jugular vein
- Diameter: 8-15mm at transverse portion
- Flow rate: 200-300 mL/min per side
- Dominance: right side in 65%, left in 25%, codominant 10%
-
Cavernous Sinus Architecture
- Location: lateral to sella turcica (sphenoid body)
- Contents: ICA + CN III, IV, V1, V2, VI
- Venous connections: superior and inferior ophthalmic veins
- Dimensions: 20mm length × 10mm width × 8mm height
- Clinical significance: thrombosis affects multiple CNs
- Surgical access: high risk due to neurovascular density
⭐ Clinical Pearl: Venous sinus thrombosis presents with increased ICP (headache, papilledema) + focal deficits depending on location. Superior sagittal sinus thrombosis causes bilateral leg weakness, cavernous sinus thrombosis causes ophthalmoplegia + facial numbness. D-dimer elevation occurs in >90% of cases.
Cerebrospinal Fluid Dynamics and Pressure Regulation
CSF circulation through dural-arachnoid interfaces maintains intracranial pressure homeostasis with production (500-750 mL/day) balanced by absorption through arachnoid granulations. Normal ICP ranges 5-15 mmHg with pathological thresholds at >20 mmHg.
-
CSF Production Centers
- Choroid plexus: 70% of CSF production (350-525 mL/day)
- Ependymal surfaces: 20% of production (100-150 mL/day)
- Brain parenchyma: 10% of production (50-75 mL/day)
- Total volume: 120-150 mL in adults
- Turnover rate: 3-4 times daily (complete replacement)
- Production rate: 0.35-0.4 mL/min (constant)
-
Absorption Mechanisms
- Arachnoid granulations: 85% of CSF absorption
- Spinal nerve roots: 10% of absorption
- Lymphatic drainage: 5% of absorption
- Pressure threshold: >5 mmHg CSF pressure for absorption
- Flow rate: 0.35-0.4 mL/min (matches production)
- Pathological backup: >20 mmHg causes papilledema
| Dural Structure | Function | Clinical Significance | Pathology | Pressure Threshold |
|---|---|---|---|---|
| Falx cerebri | Hemispheric separation | Subfalcine herniation | Mass effect | >5mm displacement |
| Tentorium cerebelli | Supra/infratentorial division | Uncal herniation | Temporal lobe mass | >5mm uncal shift |
| Superior sagittal sinus | Cortical venous drainage | Thrombosis, hemorrhage | Bilateral leg weakness | >20 mmHg ICP |
| Cavernous sinus | Parasellar drainage | CN III-VI deficits | Ophthalmoplegia | Variable |
| Arachnoid granulations | CSF absorption | Hydrocephalus | Absorption failure | >20 mmHg CSF |
Dural mastery establishes the foundation for understanding intracranial pressure dynamics and neurosurgical approaches. Connect these protective systems through clinical correlation patterns to understand traumatic injury mechanisms and surgical intervention strategies.
🛡️ Dural Defense Systems: The Brain's Protective Membrane Network
🎯 Clinical Mastery Arsenal: Rapid Head Assessment Protocols
Rapid Trauma Assessment Protocol
Primary survey identifies immediately life-threatening injuries within 60-90 seconds using systematic ABCDE approach with head-specific modifications. Glasgow Coma Scale provides standardized neurological assessment with prognostic accuracy of 85-90% for outcome prediction.
-
Airway Assessment with C-Spine Protection
- Cervical immobilization: mandatory until cleared (15% C-spine injury rate)
- Airway patency: blood, vomit, debris clearance
- Intubation criteria: GCS ≤8 or declining by ≥2 points
- RSI protocol: etomidate (0.3 mg/kg) + succinylcholine (1.5 mg/kg)
- C-spine precautions: manual inline stabilization during intubation
- Backup plan: surgical airway if failed intubation (<1% incidence)
-
Breathing and Ventilation
- Respiratory pattern: Cheyne-Stokes, central neurogenic hyperventilation
- Oxygen saturation: maintain >95% to prevent secondary injury
- Ventilation targets: PaCO2 35-40 mmHg (avoid hyperventilation)
- Hyperventilation risk: cerebral vasoconstriction → ischemia
- Hypoventilation risk: increased ICP via vasodilation
- Monitoring: end-tidal CO2 35-40 mmHg target
-
Circulation and Hemorrhage Control
- Blood pressure: maintain SBP >90 mmHg (prevent hypotension)
- Scalp bleeding: direct pressure + hemostatic agents
- Shock evaluation: rarely from isolated head trauma in adults
- Hypotension causes: spinal shock, other injuries, medications
- Resuscitation target: SBP 100-110 mmHg (avoid fluid overload)
- Blood products: early if hemorrhagic shock + head trauma
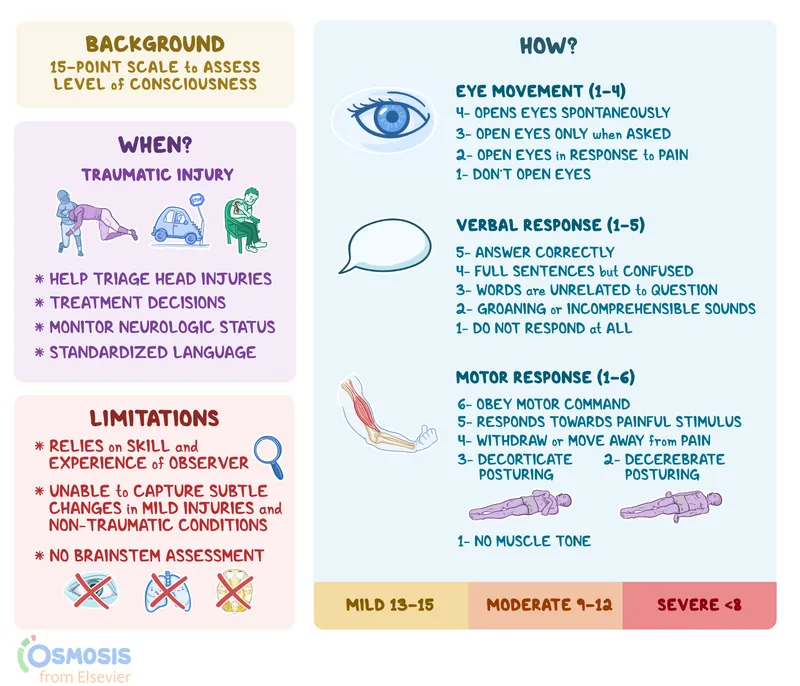
📌 Remember: GCS components - Eye opening (1-4), Verbal response (1-5), Motor response (1-6). Total 15 = normal, 13-15 = mild, 9-12 = moderate, 3-8 = severe TBI. Intubation indicated for GCS ≤8.
Neurological Examination Essentials
Focused neurological assessment identifies localizing signs and herniation syndromes within 2-3 minutes using standardized techniques. Pupillary examination provides 85% accuracy for increased ICP detection when combined with motor responses.
-
Pupillary Assessment Protocol
- Size measurement: 2-8mm normal range (document in mm)
- Light reactivity: direct and consensual responses
- Shape evaluation: round vs oval (early herniation sign)
- Anisocoria >1mm: abnormal requiring investigation
- Fixed dilated pupil: CN III compression (uncal herniation)
- Bilateral fixed pupils: brainstem injury (poor prognosis)
-
Motor Function Evaluation
- Best motor response: follows commands vs localizes vs withdraws
- Lateralizing signs: hemiparesis suggests focal lesion
- Posturing patterns: decorticate vs decerebrate
- Decorticate: flexor posturing (cortical injury)
- Decerebrate: extensor posturing (brainstem injury)
- Flaccid: no response (severe brainstem damage)
-
Cranial Nerve Screening
- CN II: visual fields, fundoscopy (papilledema)
- CN III, IV, VI: extraocular movements, ptosis
- CN V, VII: facial sensation, facial movement
- Battle's sign: mastoid ecchymosis (basilar skull fracture)
- Raccoon eyes: periorbital ecchymosis (anterior fossa fracture)
- CSF rhinorrhea/otorrhea: dural tear (15-20% of basilar fractures)
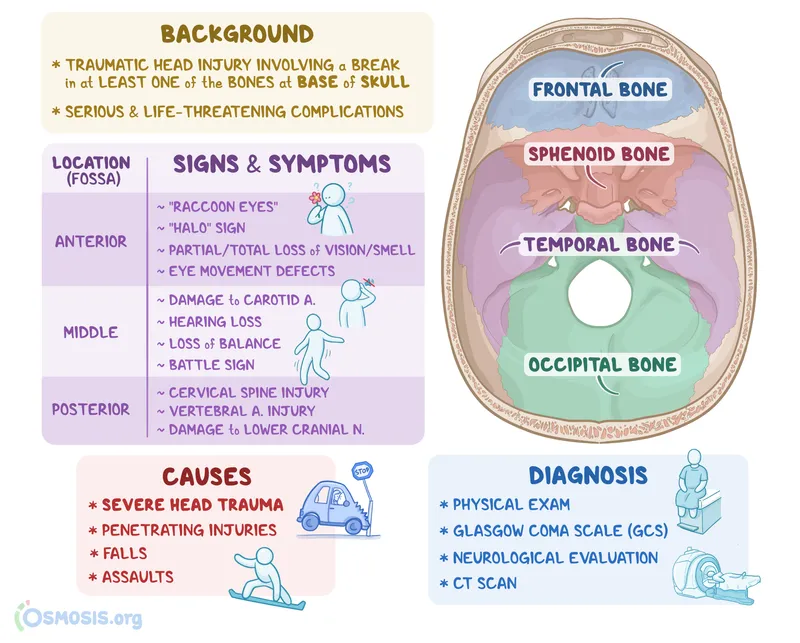
⭐ Clinical Pearl: Lucid interval occurs in 30-50% of epidural hematomas - initial loss of consciousness, recovery period (minutes to hours), then rapid deterioration. Temporal bone fractures cause epidural bleeding in 85% of cases via middle meningeal artery laceration.
Imaging Interpretation and Decision Making
CT head remains first-line imaging with 95% sensitivity for acute hemorrhage and surgical lesions. Decision rules optimize imaging utilization while maintaining >99% sensitivity for clinically significant injuries.
-
CT Indications (Canadian CT Head Rule)
- High risk (100% require CT**): GCS <15 at 2 hours, suspected skull fracture, vomiting ≥2 episodes
- Medium risk (selective CT): age >65, amnesia >30 minutes, dangerous mechanism
- Low risk (observation): none of above criteria
- Sensitivity: 100% for neurosurgical intervention
- Specificity: 50-60% (reduces unnecessary CTs)
- Cost savings: 30-40% reduction in imaging
-
CT Findings Requiring Neurosurgical Consultation
- Epidural hematoma: lens-shaped hyperdensity (emergency surgery)
- Subdural hematoma: crescent-shaped (surgery if >10mm or shift >5mm)
- Intracerebral hemorrhage: surgery if >30 mL or deterioration
- Midline shift >5mm: increased ICP (decompression indicated)
- Cisternal compression: herniation risk (urgent intervention)
- Hydrocephalus: ventricular dilation (EVD placement)
| Assessment Component | Normal Finding | Abnormal Threshold | Clinical Significance | Intervention Required |
|---|---|---|---|---|
| GCS | 15 | ≤13 | Moderate-severe TBI | Intubation if ≤8 |
| Pupils | 2-8mm, reactive | >1mm difference | Herniation syndrome | Urgent decompression |
| Motor response | Follows commands | Localizes only | Focal brain injury | Neurosurgical evaluation |
| CT midline shift | 0mm | >5mm | Increased ICP | Surgical decompression |
| Hematoma volume | None | >30 mL ICH, >10mm SDH | Mass effect | Emergency surgery |
🎯 Clinical Mastery Arsenal: Rapid Head Assessment Protocols
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app

















