Embryology and Development

On this page
🧬 Embryonic Architecture: The Master Blueprint
You'll master how a single cell orchestrates the construction of a complete human body by exploring the molecular signals, cellular movements, and genetic switches that transform embryonic potential into organized tissues and organs. This lesson connects the elegant logic of developmental biology-from axis formation to organ patterning-with the clinical reality of congenital anomalies you'll diagnose throughout your career. By understanding what drives normal development, you'll recognize precisely where and why things go wrong, turning embryology from memorization into a powerful diagnostic framework that explains birth defects, guides genetic counseling, and reveals why certain malformations cluster together.
Developmental Timing Mastery
Embryonic development follows precise temporal windows where specific events must occur within narrow timeframes:
- Week 1: Fertilization → Blastocyst formation (6-7 days)
- Week 2: Implantation → Bilaminar disc (8-14 days)
- Week 3: Gastrulation → Trilaminar disc (15-21 days)
- Week 4: Neurulation → Neural tube closure (22-28 days)
- Weeks 3-8: Organogenesis → Critical period (most teratogenic)
- Weeks 9-38: Fetal growth → Maturation phase
📌 Remember: FING - Fertilization (Week 1), Implantation (Week 2), Neurulation (Week 4), Gastrulation (Week 3)
⭐ Clinical Pearl: 95% of major congenital anomalies occur during the embryonic period (weeks 3-8), when organ systems are establishing their basic structure
Germ Layer Derivatives Framework
| Germ Layer | Major Derivatives | Clinical Significance | Malformation Examples | Molecular Signals |
|---|---|---|---|---|
| Ectoderm | CNS, PNS, epidermis, neural crest | 70% of birth defects | Spina bifida, anencephaly | BMP, FGF, Wnt |
| Mesoderm | Musculoskeletal, cardiovascular, urogenital | 20% of anomalies | CHD, limb defects | Shh, Hox genes |
| Endoderm | GI tract, lungs, liver, pancreas | 10% of malformations | TEF, biliary atresia | Sox2, Foxa2 |
The embryonic period represents maximum vulnerability to teratogenic insults:
- Organogenesis sensitivity: Each organ system has a specific critical period
- Dose-response relationship: Threshold effects determine malformation severity
- Genetic susceptibility: 15-25% of birth defects have genetic components
- Environmental factors: 10% directly attributable to teratogens
- Multifactorial inheritance: 65% involve gene-environment interactions
💡 Master This: The all-or-nothing period (weeks 1-2) either causes embryonic death or no effect, while the critical period (weeks 3-8) produces specific organ malformations based on timing
📌 Remember: TORCH infections during critical periods - Toxoplasma, Other (syphilis, VZV), Rubella, CMV, HSV cause characteristic malformation patterns
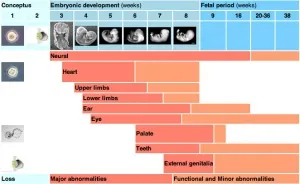
Understanding these temporal relationships predicts malformation patterns and guides genetic counseling approaches for optimal pregnancy outcomes.
🧬 Embryonic Architecture: The Master Blueprint
⚙️ Morphogenetic Machinery: The Cellular Command Center
Cellular Morphogenetic Arsenal
Proliferation Control Mechanisms
Cell division drives tissue growth and organ expansion through tightly regulated cycles:
- G1/S checkpoint: DNA integrity verification (p53 pathway)
- G2/M checkpoint: Chromosome condensation control (cyclin B/CDK1)
- Spindle checkpoint: Proper chromosome attachment (APC/C complex)
- Growth factor dependence: External signal requirements for progression
- Contact inhibition: Density-dependent growth arrest mechanisms
⭐ Clinical Pearl: Cyclopia results from Sonic hedgehog (Shh) signaling disruption, affecting 1 in 16,000 births with holoprosencephaly spectrum disorders
Migration Pattern Recognition
| Migration Type | Mechanism | Examples | Clinical Relevance | Failure Consequences |
|---|---|---|---|---|
| Collective | Sheet movement | Gastrulation, neural crest | 70% of embryonic movements | Spina bifida, cleft palate |
| Individual | Single cell | Primordial germ cells | Gonadal development | Gonadal dysgenesis |
| Chain | Connected cells | Neural crest streams | Craniofacial formation | DiGeorge syndrome |
| Radial | Outward expansion | Limb bud growth | Appendage development | Limb reduction defects |
Apoptosis Sculpting Programs
Programmed cell death refines structures by eliminating excess tissue:
- Interdigital spaces: Webbing removal between fingers/toes (BMP signaling)
- Neural tube: Excess neuron elimination (50% of neurons die normally)
- Reproductive ducts: Müllerian/Wolffian regression (sex-specific)
- Cardiac development: Ventricular septation refinement
- Lens formation: Lens vesicle hollowing (crystallin expression)
💡 Master This: Syndactyly occurs when BMP-mediated apoptosis fails in interdigital regions, affecting 1 in 2,000 births with variable penetrance
📌 Remember: MAPS for morphogenesis - Migration, Apoptosis, Proliferation, Signaling coordinate all developmental processes

These morphogenetic mechanisms integrate through signaling pathway crosstalk to generate the complex cellular choreography underlying organ formation and body plan establishment.
⚙️ Morphogenetic Machinery: The Cellular Command Center
🎯 Pattern Recognition: The Developmental GPS System
Axis Establishment Framework
Body plan organization emerges through coordinate system establishment:
- Anterior-Posterior (A-P): Hox gene clusters specify segmental identity
- Dorsal-Ventral (D-V): BMP/Chordin gradients establish tissue polarity
- Left-Right (L-R): Nodal signaling determines organ asymmetry
- Proximal-Distal (P-D): FGF/Wnt gradients control appendage outgrowth
📌 Remember: ABCD axis patterning - Anterior-posterior, BMP dorsal-ventral, Ciliary left-right, Distal FGF signaling
Morphogen Gradient Interpretation
| Morphogen | Source | Target Genes | Concentration Effects | Clinical Correlations |
|---|---|---|---|---|
| Sonic Hedgehog | Notochord, ZPA | Gli1/2/3, Ptch1 | High: ventral identity | Holoprosencephaly (1:16,000) |
| BMP4 | Lateral mesoderm | Msx1/2, Dlx genes | High: dorsal/lateral | Spina bifida (1:1,000) |
| FGF8 | AER, isthmus | Sprouty, Dusp6 | High: proliferation | Limb defects (1:2,000) |
| Wnt3a | Primitive streak | T-box, Cdx genes | High: posterior identity | Caudal regression (1:25,000) |
Segmentation Hierarchy
Body segmentation follows hierarchical gene cascades:
- Gap genes: Large segment deletions (hunchback, Krüppel)
- Pair-rule genes: Alternate segment defects (even-skipped, fushi tarazu)
- Segment polarity: Within-segment patterning (engrailed, hedgehog)
- Homeotic genes: Segment identity specification (Hox clusters)
⭐ Clinical Pearl: VACTERL association affects 1 in 10,000 births, representing developmental field defects involving vertebral, anal, cardiac, tracheal, esophageal, renal, and limb structures
Hox Gene Collinearity Principle
Hox gene expression follows spatial and temporal collinearity:
- 3' genes: Anterior expression (brain, cervical)
- 5' genes: Posterior expression (lumbar, sacral)
- Early activation: 3' to 5' temporal sequence
- Nested domains: Overlapping expression boundaries
- Functional redundancy: Paralogous group compensation
💡 Master This: Homeotic transformations result from Hox gene mutations, causing cervical ribs (C7→T1 transformation) in 0.5-1% of population
📌 Remember: HOXC collinearity - Head to tail, Ordered expression, X-axis temporal, Clustered organization
These patterning mechanisms establish positional information that guides cell fate specification and organ placement throughout embryonic development, connecting molecular signals to anatomical outcomes.
🎯 Pattern Recognition: The Developmental GPS System
🔬 Signaling Network Analysis: The Molecular Orchestra
Core Signaling Pathway Arsenal
| Pathway | Key Components | Primary Functions | Malformation Spectrum | Therapeutic Targets |
|---|---|---|---|---|
| Wnt | β-catenin, TCF/LEF | Axis formation, proliferation | Sirenomelia, tetra-amelia | GSK3β inhibitors |
| Hedgehog | Shh, Gli, Ptch | Patterning, growth | Holoprosencephaly, polydactyly | Smoothened modulators |
| BMP/TGF-β | Smad2/3, Noggin | D-V patterning, apoptosis | Neural tube defects | Activin inhibitors |
| FGF | FGFR1-4, ERK | Proliferation, migration | Craniosynostosis, limb defects | FGFR antagonists |
| Notch | Delta, Jagged, Hes | Cell fate, boundaries | Alagille syndrome, spondylocostal | γ-secretase inhibitors |
Signal Transduction Mechanisms
Pathway activation follows conserved molecular logic:
- Ligand binding: Receptor conformational changes (nanomolar affinity)
- Signal amplification: Kinase cascades (100-1000x amplification)
- Nuclear translocation: Transcription factor activation (minutes to hours)
- Target gene expression: Effector protein production (hours to days)
- Feedback regulation: Negative/positive loops (homeostatic control)
⭐ Clinical Pearl: Craniosynostosis affects 1 in 2,500 births, with 85% involving FGFR mutations causing premature suture fusion through excessive proliferation signals
Temporal Signal Integration
Developmental timing requires precise signal coordination:
- Competence windows: Limited responsiveness periods (hours to days)
- Signal duration: Transient vs. sustained activation (different outcomes)
- Threshold effects: Concentration-dependent responses (switch-like behavior)
- Sequential activation: Pathway cascades (temporal ordering)
- Oscillatory signals: Periodic activation (segmentation clocks)
💡 Master This: Somitogenesis requires Notch oscillations every 90-120 minutes in humans, with disruptions causing spondylocostal dysostosis affecting 1 in 100,000 births
📌 Remember: SWIFT signaling analysis - Source identification, Window timing, Intensity thresholds, Feedback loops, Target outcomes
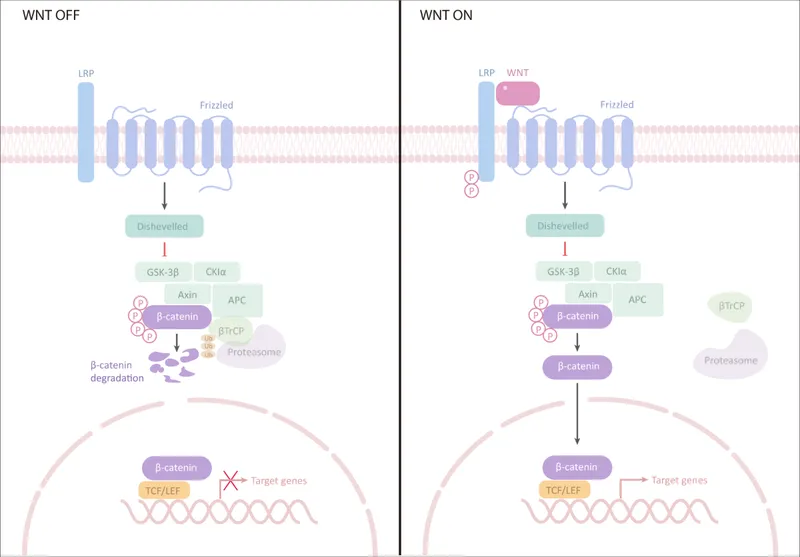
Understanding these signaling network principles enables prediction of phenotypic outcomes from genetic mutations and guides precision medicine approaches for developmental disorders.
🔬 Signaling Network Analysis: The Molecular Orchestra
⚕️ Clinical Integration: The Diagnostic Decision Tree
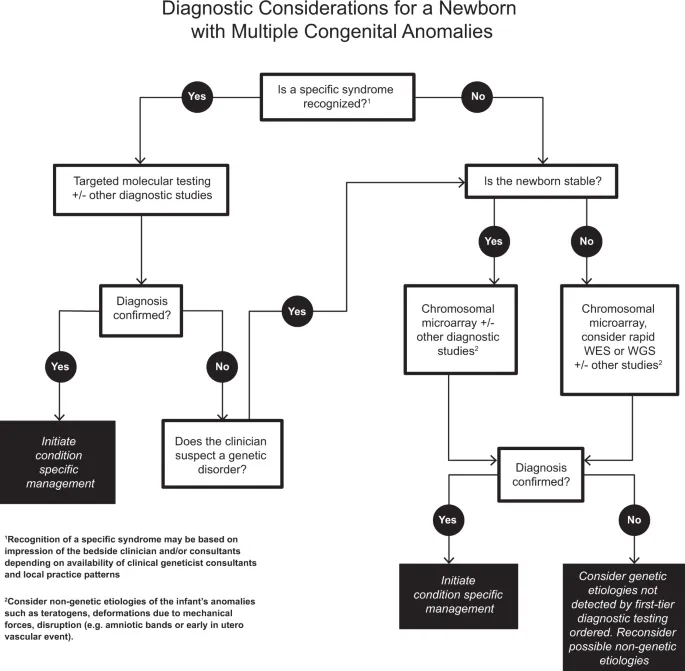
Malformation Classification System
Syndrome Recognition Framework
Pattern-based diagnosis follows systematic evaluation:
- Major malformations: Life-threatening or surgically correctable defects
- Minor anomalies: Cosmetic or functional variants (>4% frequency)
- Sequence: Single primary defect with secondary consequences
- Syndrome: Multiple anomalies with common etiology
- Association: Non-random occurrence without known cause
⭐ Clinical Pearl: CHARGE syndrome affects 1 in 8,500 births with CHD7 mutations in 90% of cases, requiring systematic evaluation of coloboma, heart defects, atresia choanae, retardation, genital anomalies, ear abnormalities
Evidence-Based Risk Assessment
| Risk Category | Recurrence Rate | Genetic Basis | Counseling Approach | Management Strategy |
|---|---|---|---|---|
| Chromosomal | 1-5% (age-dependent) | Aneuploidy, structural | Maternal age risk | CVS/amniocentesis |
| Single gene | 25-50% (inheritance) | Mendelian patterns | Carrier screening | Preimplantation diagnosis |
| Multifactorial | 2-5% (empiric) | Polygenic + environment | Lifestyle modification | Enhanced screening |
| Teratogenic | Variable (exposure) | Environmental agents | Avoidance counseling | Exposure elimination |
Testing strategies maximize diagnostic efficiency:
- Chromosomal microarray: 15-20% yield for multiple anomalies
- Exome sequencing: 25-30% yield for suspected syndromes
- Targeted panels: 40-60% yield for specific phenotypes
- Functional studies: Variant interpretation for uncertain significance
- Family segregation: Inheritance pattern confirmation
💡 Master This: Fetal alcohol spectrum disorders affect 1-5% of births with no safe alcohol threshold, requiring comprehensive evaluation including growth restriction, facial dysmorphism, neurodevelopmental delays
Treatment Algorithm Development
Management protocols integrate severity assessment with intervention timing:
- Immediate: Life-threatening anomalies (cardiac, airway, CNS)
- Urgent: Functional impairment (feeding, breathing, elimination)
- Elective: Cosmetic correction (optimal timing considerations)
- Supportive: Developmental interventions (early intervention services)
- Preventive: Complication monitoring (surveillance protocols)
📌 Remember: TEAMS approach - Timing assessment, Etiology determination, Associated anomalies, Management planning, Support coordination
These evidence-based frameworks enable systematic evaluation of complex presentations while providing families with accurate prognostic information and management expectations.
⚕️ Clinical Integration: The Diagnostic Decision Tree
🌐 Systems Integration: The Developmental Connectome
Multi-System Coordination Networks
Developmental integration occurs through shared molecular pathways:
- Neural crest contributions: Craniofacial, cardiac, enteric systems (common origin)
- Hox gene domains: Axial skeleton, limbs, reproductive tract (positional identity)
- Retinoic acid signaling: CNS, limbs, heart, kidneys (morphogen gradient)
- Sonic hedgehog pathway: CNS, limbs, gut, lungs (pattern formation)
- TBX gene family: Heart, limbs, placenta (T-box transcription factors)
Temporal Cascade Integration
| Developmental Window | Primary Events | System Interactions | Critical Dependencies | Failure Consequences |
|---|---|---|---|---|
| Weeks 3-4 | Gastrulation, neurulation | Axis establishment | Nodal, BMP gradients | VACTERL spectrum |
| Weeks 4-6 | Organogenesis initiation | Field interactions | FGF, Wnt signaling | Multiple anomalies |
| Weeks 6-8 | Organ specification | Inductive cascades | Tissue interactions | Sequence defects |
| Weeks 8-12 | Functional maturation | System coordination | Hormonal signals | Functional deficits |
Recent discoveries reveal novel coordination principles:
- Mechanical forces: Physical constraints shape organ morphology (tensile stress)
- Metabolic coupling: Energy gradients influence cell fate decisions (glycolysis vs. oxidation)
- Epigenetic landscapes: Chromatin modifications coordinate temporal gene expression (histone marks)
- Non-coding RNAs: microRNAs, lncRNAs fine-tune developmental timing (post-transcriptional control)
- Extracellular matrix: Mechanical properties guide cell migration and differentiation (stiffness gradients)
⭐ Clinical Pearl: 22q11.2 deletion syndrome affects 1 in 4,000 births, demonstrating neural crest disruption causing cardiac defects (74%), palatal anomalies (69%), immune deficiency (77%), developmental delays (90%)
Systems Biology Applications
Computational modeling predicts phenotypic outcomes:
- Gene regulatory networks: Boolean logic models predict expression patterns
- Morphogenetic simulations: Physical modeling of tissue deformation
- Pathway perturbation: In silico mutations predict malformation patterns
- Drug target identification: Network analysis reveals therapeutic nodes
- Personalized risk assessment: Polygenic scores for complex traits
💡 Master This: Ciliopathies represent system-wide defects affecting >35 genes in primary cilia function, causing overlapping phenotypes including Bardet-Biedl (1:140,000), Meckel-Gruber (1:13,000), Joubert (1:80,000) syndromes
📌 Remember: LINKS for system integration - Lineage tracing, Inductive signals, Network analysis, Kinetic modeling, System perturbation
Developmental Integration Framework
Comprehensive understanding requires multi-level analysis:
-
Molecular Networks: Signaling pathways, gene regulation, and epigenetic control establish the foundational circuitry for cellular communication and developmental decisions
-
Cellular Interactions: Migration patterns, adhesion systems, and mechanical forces coordinate tissue formation through direct cell-cell contact and matrix-mediated signals
-
Tissue Coordination: Morphogenetic fields, inductive cascades, and temporal synchrony integrate multiple cell types into functional organ systems with precise spatial organization
-
System Outcomes: Functional integration, phenotypic robustness, and evolutionary constraints ensure coordinated development that produces viable organisms capable of environmental adaptation
Understanding these integrated networks enables prediction of complex phenotypes from genetic variants and guides multi-system therapeutic strategies for developmental disorders.
🌐 Systems Integration: The Developmental Connectome
🎯 Clinical Mastery Arsenal: The Embryology Command Center
Essential Clinical Arsenal
High-Yield Recognition Patterns:
- Holoprosencephaly spectrum: Shh pathway → Cyclopia to mild hypotelorism
- VACTERL association: Developmental field → ≥3 of 7 systems affected
- CHARGE syndrome: CHD7 mutations → Coloboma, heart, atresia, retardation, genital, ear
- DiGeorge sequence: 22q11.2 deletion → Cardiac, thymic, parathyroid, facial defects
- Fetal alcohol spectrum: Teratogen exposure → Growth, facial, neurodevelopmental triad
📌 Remember: TORCH-S teratogen timing - Toxoplasma (any trimester), Other/syphilis (2nd-3rd), Rubella (1st), CMV (any), HSV (delivery), Streptococcus (delivery)
Rapid Assessment Framework
| Clinical Presentation | First-Line Evaluation | Diagnostic Yield | Management Priority | Recurrence Risk |
|---|---|---|---|---|
| Multiple anomalies | Chromosomal microarray | 15-20% | Life-threatening first | 1-50% (variable) |
| Isolated heart defect | Echocardiogram + genetics | 8-12% | Surgical planning | 2-4% (empiric) |
| Neural tube defect | MRI + folate levels | 5-10% | Neurosurgical repair | 3-5% (folate responsive) |
| Limb reduction | Targeted gene panel | 25-40% | Functional assessment | 25% (if genetic) |
Master Diagnostic Algorithms
Syndrome Recognition Hierarchy:
- Life-threatening features: Cardiac, airway, CNS anomalies (immediate intervention)
- Dysmorphic features: Facial gestalt recognition (syndrome-specific patterns)
- Growth parameters: Intrauterine vs. postnatal patterns (timing clues)
- Developmental milestones: Global vs. specific delays (system involvement)
- Family history: Inheritance patterns and recurrence risks
💡 Master This: Critical period vulnerability - Week 3-8 organogenesis accounts for 95% of major malformations, with specific timing determining affected systems
📌 Remember: MAGIC embryology mastery - Malformation patterns, Association recognition, Genetic testing, Inheritance counseling, Critical period timing
Essential Numbers for Clinical Practice:
- Neural tube defects: 1-2 per 1,000 births (folate preventable)
- Congenital heart disease: 8 per 1,000 births (most common major anomaly)
- Chromosomal abnormalities: 6 per 1,000 births (maternal age dependent)
- Single gene disorders: 10 per 1,000 births (Mendelian inheritance)
- Teratogen exposure: 2-3% of major malformations (preventable causes)
This clinical mastery framework enables systematic evaluation of complex presentations while providing evidence-based management and accurate family counseling for optimal patient outcomes.
🎯 Clinical Mastery Arsenal: The Embryology Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app










