Developmental Anatomy

On this page
🧬 The Developmental Blueprint: Embryonic Architecture Mastery
You'll master how a single fertilized cell orchestrates the precise assembly of the human body, exploring the molecular signals that sculpt tissues, the architectural principles that build organs, and the critical windows when disruptions cause congenital anomalies. This lesson connects embryonic blueprints to clinical reality-you'll recognize malformation patterns, understand their developmental origins, and apply evidence-based interventions that change outcomes. By integrating molecular command centers with multi-system networks, you'll think like both architect and detective, transforming developmental biology into diagnostic power at the bedside.
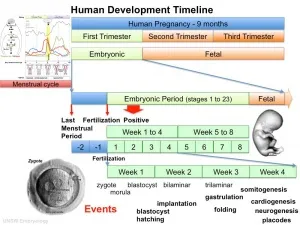
📌 Remember: FOLD - Fertilization (day 1), Organogenesis (weeks 3-8), Later fetal period (weeks 9-38), Delivery. Critical periods: 85% of major anomalies occur during organogenesis when cell division rates reach 100,000 cells/hour.
The developmental timeline operates with surgical precision - teratogenic exposure during days 15-60 causes 60-fold higher anomaly rates compared to later periods. Master clinicians recognize that neural tube closure completes by day 28, cardiac septation by day 45, and palatal fusion by week 10 - knowledge that predicts which anomalies cluster together.
⭐ Clinical Pearl: Folic acid supplementation reduces neural tube defects by 72% when started 4 weeks pre-conception. Maternal diabetes increases congenital heart defect risk by 3-5 fold, with HbA1c >8.5% showing 15% anomaly rates versus 2-3% in controlled diabetes.
| Developmental Period | Timeline | Key Events | Teratogen Sensitivity | Major Anomalies |
|---|---|---|---|---|
| Pre-embryonic | Days 1-14 | Fertilization, implantation | All-or-none effect | Embryonic death |
| Embryonic | Days 15-56 | Organogenesis | Maximum (85% anomalies) | Structural defects |
| Early fetal | Weeks 9-16 | Organ maturation | Moderate | Growth restriction |
| Late fetal | Weeks 17-38 | Growth, refinement | Minimal | Functional defects |
| Postnatal | Birth onward | Continued development | Variable | Growth disorders |
- Morphogenetic Movements
- Cell proliferation: exponential growth from 1 cell to 2.6 trillion at birth
- Cell migration: neural crest cells travel >50 cell diameters to form facial structures
- Cell differentiation: pluripotent stem cells commit to 220+ cell types
- Apoptosis: programmed death of 50-70% of neurons during development
- Cell adhesion: CAM molecules guide tissue boundary formation
Connect these foundational principles through molecular signaling cascades to understand how single gene mutations can disrupt entire developmental programs.
🧬 The Developmental Blueprint: Embryonic Architecture Mastery
⚙️ The Molecular Command Center: Signaling Pathway Mastery
📌 Remember: WISH - Wnt (axis formation), Insulin-like growth factors (growth), Sonic hedgehog (patterning), Hox genes (segmentation). These 4 pathway families control >90% of developmental decisions with cross-regulatory feedback loops.
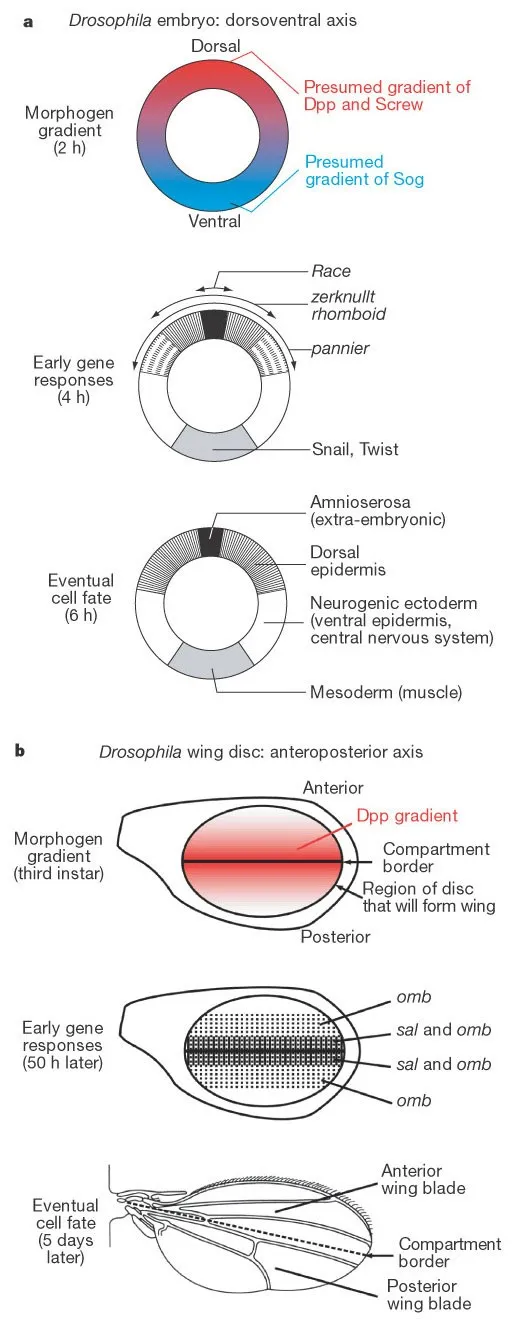
⭐ Clinical Pearl: Holoprosencephaly results from Shh pathway disruption - mutations in SHH, PTCH1, or GLI2 cause cyclopia in severe cases (1:16,000 births) and single central incisor in mild forms (1:50 people). Cholesterol synthesis defects also disrupt Shh signaling since the pathway requires cholesterol modification.
| Signaling Pathway | Key Molecules | Primary Functions | Clinical Relevance | Mutation Frequency |
|---|---|---|---|---|
| Wnt | β-catenin, TCF/LEF | Axis formation, proliferation | Colorectal cancer (80% APC mutations) | 1:10,000 |
| Hedgehog | Shh, Ptch, Gli | Patterning, growth | Holoprosencephaly, basal cell carcinoma | 1:16,000 |
| TGF-β/BMP | Smad proteins | Cell fate, apoptosis | Marfan syndrome, PAH | 1:5,000 |
| FGF | FGFR1-4, ERK | Proliferation, migration | Achondroplasia (99% FGFR3) | 1:25,000 |
| Notch | Delta, Jagged, Hes | Lateral inhibition | Alagille syndrome, CADASIL | 1:70,000 |
- Hox genes: 39 genes in 4 clusters specify anterior-posterior identity
- Master regulators: PAX6 (eye), NKX2.5 (heart), SOX9 (cartilage)
- Chromatin remodeling: >200 histone modifications control gene accessibility
- DNA methylation: 5-methylcytosine silences tissue-specific genes
- Enhancer elements: cis-regulatory sequences active across >1 Mb distances
💡 Master This: Epigenetic inheritance explains how environmental factors during critical windows cause permanent phenotypic changes without DNA sequence alterations. Maternal nutrition during periconceptional period affects offspring metabolism for decades through DNA methylation patterns.
These molecular control mechanisms integrate through tissue morphogenesis to create three-dimensional organ architecture with predictable failure patterns.
⚙️ The Molecular Command Center: Signaling Pathway Mastery
🏗️ The Morphogenetic Machinery: Tissue Architecture Assembly
Gastrulation represents the most critical morphogenetic event - 3 germ layers form through coordinated cell movements involving >50,000 cells migrating specific distances within 24-hour windows. Primitive streak formation at day 15 establishes bilateral symmetry and anterior-posterior polarity that determines final body plan.
📌 Remember: GAME - Gastrulation (3 layers), Axis formation (symmetry), Morphogenetic movements (folding), Epithelial-mesenchymal transition (cell migration). Neural tube closure requires >95% accuracy in cellular adhesion - 1 in 1,000 failures causes spina bifida.
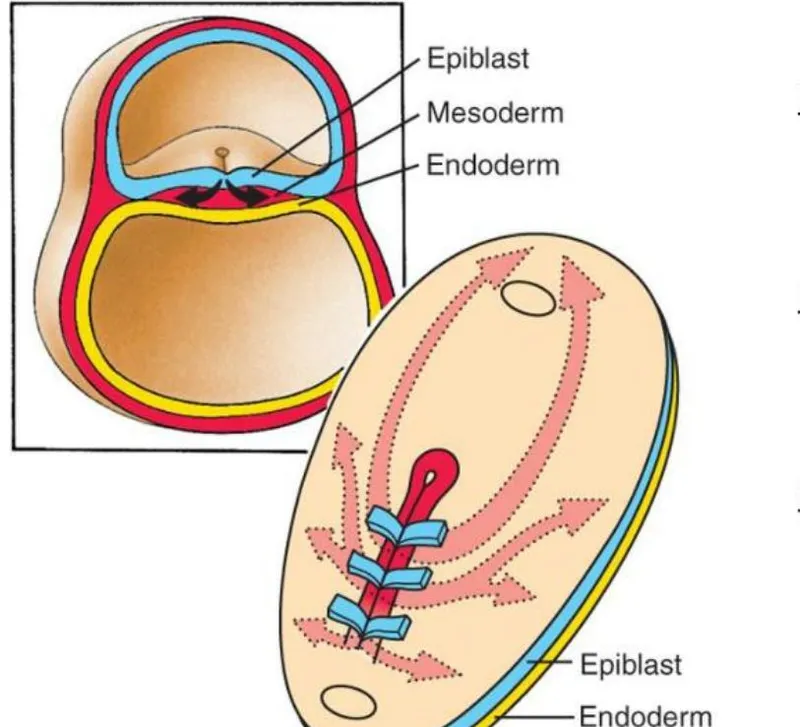
- Epithelial-Mesenchymal Transition (EMT)
- Neural crest delamination: E-cadherin downregulation by >80% enables cell migration
- Mesenchymal condensation: N-CAM and N-cadherin upregulation creates tissue boundaries
- Basement membrane breakdown: MMP2/9 activity increases 10-fold during EMT
- Snail/Slug transcription factors: repress epithelial genes within 2-4 hours
- TGF-β signaling: induces EMT in >15 developmental contexts
⭐ Clinical Pearl: Cleft lip/palate results from failed epithelial fusion during weeks 6-10. Bilateral cleft lip occurs in 1:1,000 births with 80% heritability. Folic acid supplementation reduces risk by 25%, while maternal smoking increases risk 2-fold through oxidative stress affecting cell adhesion molecules.
| Morphogenetic Process | Timeline | Key Mechanisms | Failure Consequences | Prevention Strategies |
|---|---|---|---|---|
| Neural tube closure | Days 22-28 | Apical constriction, convergent extension | Spina bifida (1:1,000) | Folic acid 400μg daily |
| Cardiac looping | Days 23-28 | Left-right asymmetry, cell migration | Dextrocardia, CHD | Avoid teratogens |
| Limb bud formation | Days 26-32 | FGF signaling, Hox expression | Limb defects (1:2,000) | Genetic counseling |
| Palatal fusion | Weeks 6-10 | Epithelial apoptosis, mesenchymal proliferation | Cleft palate (1:700) | Folic acid, avoid smoking |
| Gut rotation | Weeks 6-10 | Differential growth, mesenteric attachment | Malrotation (1:500) | Prenatal diagnosis |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
Start["🧬 Epithelial Sheet
• Cell-cell adhesion• Polarized layer"]
Signals{"⚡ EMT Signals
• Induction phase• Growth factors"}
Ecad["⬇️ E-cadherin Loss
• Loss of adhesion• Junction breakdown"]
Bcat["🧬 Beta-catenin
• Nuclear entry• Gene activation"]
Migrat["🏃 Cell Migration
• Motile phenotype• Invasive ability"]
Condense["🧫 Mesenchymal
• Condensation step• Tissue remodeling"]
Organ["🫀 Organ Formation
• Morphogenesis• Final structure"]
Start --> Signals Signals -->|TGF-beta| Ecad Signals -->|Wnt| Bcat Ecad --> Migrat Bcat --> Migrat Migrat --> Condense Condense --> Organ
style Start fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Signals fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Ecad fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Bcat fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style Migrat fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style Condense fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style Organ fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252
> 💡 **Master This**: **Mechanical forces** during **morphogenesis** generate **tissue tensions** that guide **further development**. **Hydrocephalus** can result from **altered CSF flow** during **brain folding**, while **oligohydramnios** causes **limb contractures** through **reduced fetal movement** - demonstrating how **physical constraints** shape **anatomical outcomes**.
Understanding **morphogenetic principles** enables **pattern recognition** of **developmental anomaly clusters** that share **common mechanistic origins**.
---
🏗️ The Morphogenetic Machinery: Tissue Architecture Assembly
🔍 The Anomaly Detective: Pattern Recognition Mastery
VACTERL association demonstrates non-random clustering - Vertebral, Anal, Cardiac, Tracheoesophageal, Renal, Limb defects occur together in 1:10,000-40,000 births with >3 features required for diagnosis. No single gene causes VACTERL, suggesting disrupted morphogenetic field affecting multiple organ systems during weeks 4-6.
📌 Remember: CATCH-22 - Cardiac defects, Abnormal facies, Thymic hypoplasia, Cleft palate, Hypocalcemia from 22q11.2 deletion. Affects 1:4,000 births with >180 clinical features due to neural crest cell migration defects during weeks 3-4.
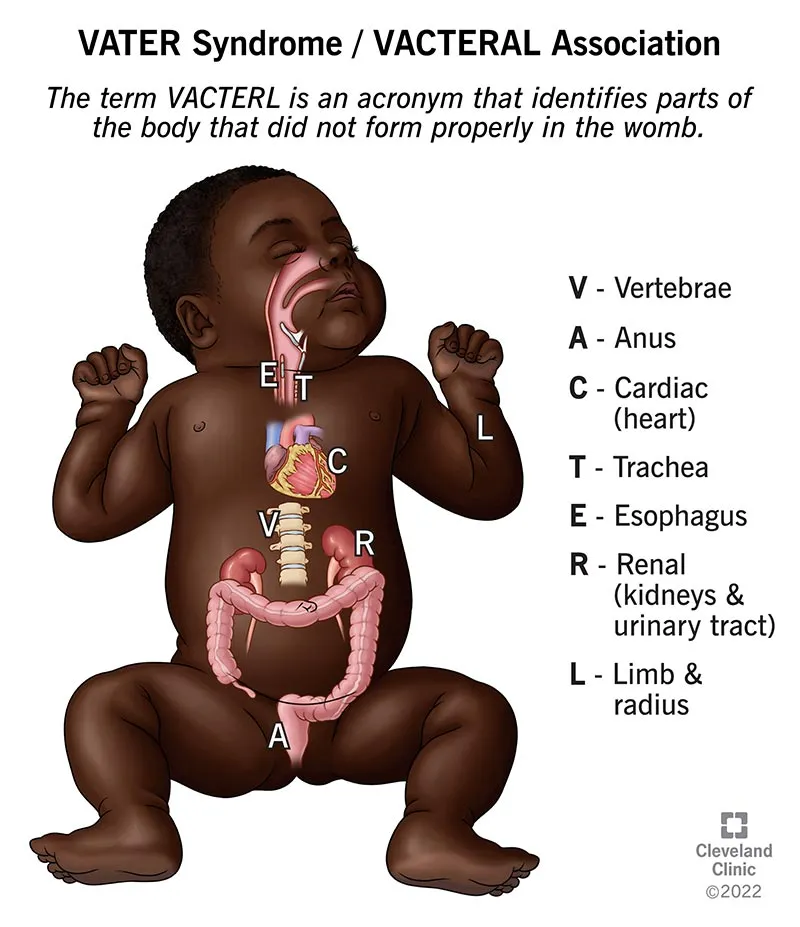
- Sequence versus Syndrome Recognition
- Potter sequence: oligohydramnios → fetal compression → growth restriction, limb deformities, pulmonary hypoplasia
- DiGeorge syndrome: 22q11.2 deletion → neural crest defects → cardiac, facial, thymic, parathyroid anomalies
- CHARGE syndrome: CHD7 mutations → chromatin remodeling defects → multiple organ dysgenesis
- Holoprosencephaly sequence: forebrain cleavage failure → facial dysmorphism, endocrine defects
- Sirenomelia sequence: vascular steal → caudal regression, renal agenesis
⭐ Clinical Pearl: Maternal diabetes increases congenital anomaly risk by 2-5 fold with characteristic pattern - caudal regression (200-fold increased), cardiac defects (3-fold), neural tube defects (2-fold). HbA1c >8.5% during organogenesis shows 15% anomaly rate versus 2-3% in well-controlled diabetes.
| Anomaly Pattern | Underlying Mechanism | Key Features | Recurrence Risk | Diagnostic Clues |
|---|---|---|---|---|
| VACTERL | Morphogenetic field defect | ≥3 of 7 features | 1% (sporadic) | Normal karyotype |
| CHARGE | CHD7 chromatin remodeling | Coloboma, heart, atresia | 50% (AD) | Semicircular canal dysplasia |
| 22q11.2 deletion | Neural crest migration | Cardiac, facial, immune | 50% (AD) | Hypocalcemia, infections |
| Diabetic embryopathy | Hyperglycemia/ketones | Caudal, cardiac, neural | 2-5% (maternal) | Maternal HbA1c >7% |
| Fetal alcohol | CNS toxicity, growth | Facial, growth, cognitive | Variable | Maternal history |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
Start["⚠️ Teratogenic Exposure
• Environmental risk• Fetal impact"]
Critical["📋 Critical Period
• Timing of insult• Gestational age"]
Neural["🩺 Neural Crest
• Migratory cell risk• Early development"]
Charge["🩺 CHARGE/22q11.2
• Facial anomalies• DiGeorge pattern"]
Cardiac["🩺 Cardiac Defects
• Heart formation• Mid-organogenesis"]
Vacterl["🩺 VACTERL Pattern
• Vertebral defects• Renal/Limb risk"]
Limb["🩺 Limb Defects
• Extremity growth• Late organogenesis"]
Thal["🩺 Thalidomide
• Phocomelia link• Shortened limbs"]
Start --> Critical Critical -->|Weeks 3-4| Neural Neural --> Charge Critical -->|Weeks 4-6| Cardiac Cardiac --> Vacterl Critical -->|Weeks 6-8| Limb Limb --> Thal
style Start fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Critical fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Neural fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Charge fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Cardiac fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Vacterl fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Limb fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Thal fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8
> 💡 **Master This**: **Timing-specific teratogenic patterns** enable **retrospective exposure dating**. **Thalidomide** exposure during **days 20-36** causes **specific limb defects** - **upper limb phocomelia** (days 24-30), **lower limb defects** (days 28-36). **Isotretinoin** during **weeks 3-5** causes **neural crest defects** identical to **22q11.2 deletion syndrome**.
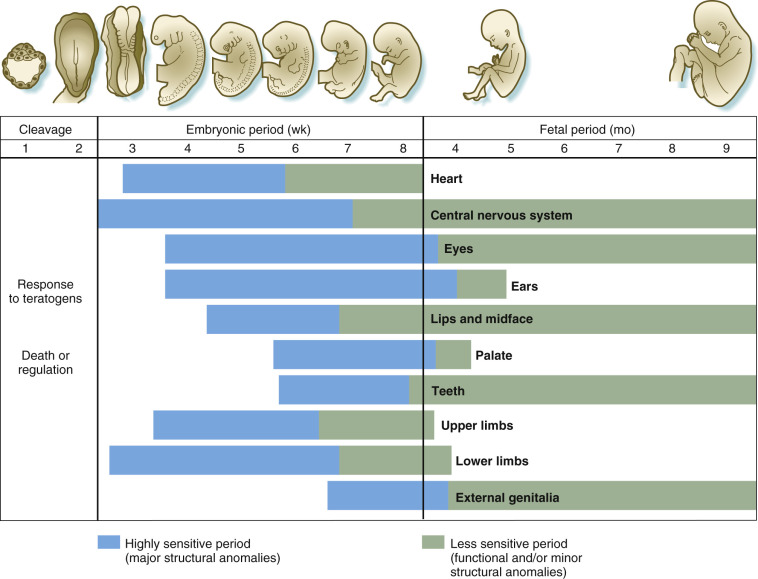
These **pattern recognition skills** guide **systematic evaluation approaches** for **efficient anomaly workup** and **accurate recurrence counseling**.
---
🔍 The Anomaly Detective: Pattern Recognition Mastery
⚖️ The Treatment Algorithm: Evidence-Based Intervention Mastery
Neural tube defect management exemplifies evidence-based algorithms - fetal surgery for myelomeningocele before 26 weeks reduces hindbrain herniation by 36% and shunt requirement by 40%, but increases preterm delivery risk to 79% versus 15% in postnatal repair. Selection criteria include lesion level ≤S1, hindbrain herniation, and absence of kyphosis >30°.
📌 Remember: FETAL - Fetal surgery criteria: Early gestation (<26 weeks), Termination counseling completed, Anatomical eligibility confirmed, Low maternal risk. Success rates: 40% reduction in VP shunt needs, improved motor function in 42% at 30 months.
- Congenital Heart Disease Algorithms
- Critical CHD screening: pulse oximetry at 24-48 hours detects 76% of critical lesions
- Balloon atrial septostomy: emergency intervention for TGA within hours of birth
- Norwood procedure: 3-stage palliation for HLHS with 85% survival to stage 3
- PGE infusion: maintains ductal patency in ductal-dependent lesions
- Hybrid procedures: PA banding + ductal stenting for high-risk neonates
⭐ Clinical Pearl: Congenital diaphragmatic hernia outcomes correlate with lung-to-head ratio (LHR) - LHR >1.4 predicts >90% survival, LHR <1.0 indicates <50% survival. ECMO improves survival in severe cases from 50% to 75%, but requires reversible pulmonary hypertension.
| Condition | Intervention Window | Success Criteria | Survival Rates | Long-term Outcomes |
|---|---|---|---|---|
| Myelomeningocele | <26 weeks GA | Lesion ≤S1, hindbrain herniation | 95% survival | 40% ↓ shunt needs |
| Critical CHD | First 48 hours | Pulse ox <95% | 85-95% survival | Normal development 80% |
| CDH | Immediate | LHR >1.0 | 75% with ECMO | Pulmonary function 60% normal |
| Gastroschisis | Immediate | Primary closure if possible | >95% survival | Normal GI function 90% |
| Cleft lip/palate | 3-6 months/12-18 months | Adequate weight gain | >99% survival | Normal speech 85% |
These evidence-based algorithms integrate with multisystem considerations to optimize long-term functional outcomes across developmental trajectories.
⚖️ The Treatment Algorithm: Evidence-Based Intervention Mastery
🔗 The Integration Matrix: Multi-System Developmental Networks
Ciliopathies demonstrate multi-system integration - primary cilia dysfunction affects >15 organ systems through disrupted signaling pathways. Bardet-Biedl syndrome involves >20 genes encoding ciliary proteins, causing retinal degeneration, polydactyly, renal cysts, obesity, and cognitive impairment through defective Hedgehog, Wnt, and PDGF signaling.
📌 Remember: CILIA - Cardiac defects, Intellectual disability, Liver fibrosis, Infertility, Anosmia. Primary ciliary dyskinesia affects 1:15,000-30,000 with >40 genes involved. Kartagener syndrome (situs inversus + PCD) occurs in 50% of PCD patients.
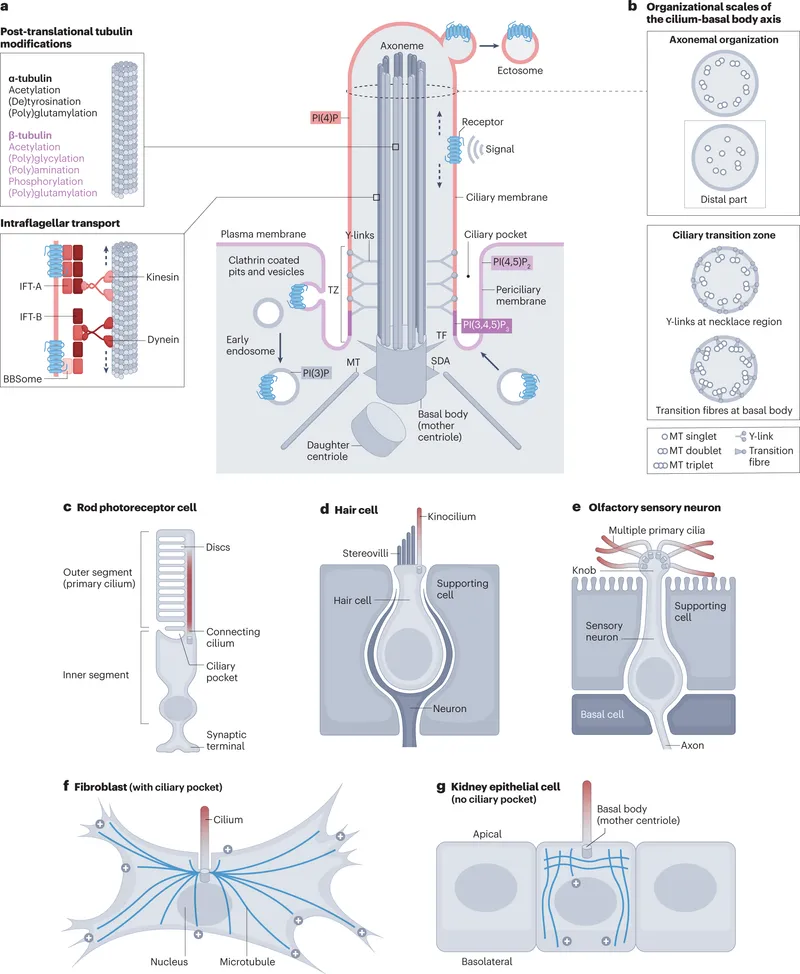
- Neurocristopathies Network
- Neural crest cell derivatives: peripheral nervous system, craniofacial skeleton, cardiac outflow tract, enteric nervous system
- CHARGE syndrome: CHD7 mutations affect neural crest migration → coloboma, heart defects, choanal atresia
- Waardenburg syndrome: PAX3/SOX10 mutations → hearing loss, pigmentation defects, Hirschsprung disease
- 22q11.2 deletion: affects >30 genes → cardiac, facial, immune, psychiatric phenotypes
- Treacher Collins: TCOF1 mutations → mandibulofacial dysostosis through ribosomal dysfunction
⭐ Clinical Pearl: Holoprosencephaly spectrum demonstrates gene-environment interactions - SHH mutations cause severe forms in homozygotes but single central incisor in heterozygotes. Maternal diabetes increases penetrance by 3-fold through glucose-induced oxidative stress affecting Shh signaling.
| Integration Network | Key Pathways | Organ Systems | Clinical Syndromes | Molecular Targets |
|---|---|---|---|---|
| Ciliopathies | Hedgehog, Wnt, PDGF | Kidney, retina, brain, limbs | BBS, Joubert, NPHP | >200 ciliary genes |
| Neurocristopathies | SOX10, PAX3, TFAP2A | Heart, face, ENS, PNS | CHARGE, Waardenburg | Neural crest TFs |
| RASopathies | RAS/MAPK pathway | Heart, face, growth, cognition | Noonan, CFC, Costello | RAS pathway genes |
| Chromatinopathies | Chromatin remodeling | Multiple systems | Kabuki, Cornelia de Lange | SWI/SNF complex |
| Ribosomopathies | Ribosome biogenesis | Craniofacial, hematopoietic | Treacher Collins, DBA | Ribosomal proteins |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
NCC["🧠 Neural Crest Cells
• Multipotent origin• Ectoderm derivation"]
SIG["🚦 Migration Signals
• Molecular cues• Directed movement"]
TAR["🎯 Target Tissues
• Site destination• Tissue induction"]
OFT["🫀 Outflow Tract
• Heart septation• Great vessels"]
FSK["💀 Facial Skeleton
• Cranial bones• Pharyngeal arches"]
PNS["🔌 PNS/ENS
• Peripheral nerves• Enteric system"]
CHD["🩺 CHD Phenotypes
• Cardiac defects• Clinical disease"]
NCC --> SIG SIG --> TAR TAR -->|Cardiac| OFT TAR -->|Craniofacial| FSK TAR -->|Neural| PNS
OFT --> CHD FSK --> CHD PNS --> CHD
style NCC fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style SIG fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style TAR fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style OFT fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style FSK fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style PNS fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style CHD fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252
> 💡 **Master This**: **Phenotypic variability** within **syndrome families** reflects **modifier genes**, **environmental factors**, and **stochastic developmental events**. **22q11.2 deletion** shows **>95%** penetrance for **some features** (thymic hypoplasia) but **<30%** for **others** (cleft palate), explaining **intrafamilial variation** even with **identical deletions**.
These **integration networks** provide **frameworks** for **systematic clinical assessment** and **targeted therapeutic interventions** based on **pathway-specific mechanisms**.
---
🔗 The Integration Matrix: Multi-System Developmental Networks
🎯 The Clinical Arsenal: Rapid Assessment & Management Tools
Rapid Anomaly Assessment Protocol follows systematic evaluation - external examination identifies >80% of major anomalies within first 24 hours. Dysmorphology examination includes >50 specific measurements and pattern recognition for >7,000 genetic syndromes with diagnostic algorithms achieving >90% accuracy for common conditions.
📌 Remember: RAPID - Recognize patterns, Assess severity, Predict associations, Identify urgency, Direct management. Critical anomalies requiring immediate intervention: choanal atresia (airway), gastroschisis (fluid/heat loss), critical CHD (circulation).
- Essential Clinical Measurements
- Growth parameters: head circumference (<3rd percentile = microcephaly), length/weight ratios
- Facial measurements: interpupillary distance, palpebral fissure length, philtrum length
- Limb proportions: upper/lower segment ratio, arm span/height ratio
- Dermatoglyphic analysis: palmar creases, fingerprint patterns, ridge counts
- Organ-specific screening: cardiac echo (CHD), renal ultrasound (anomalies), ophthalmologic exam (colobomas)
⭐ Clinical Pearl: Three or more minor anomalies predict major anomaly presence in >90% of cases. Single minor anomaly occurs in 14% of normal newborns, two minor anomalies in 0.8%, three or more in <0.5% - making multiple minor anomalies highly predictive of underlying syndrome.
| Assessment Priority | Time Frame | Key Features | Immediate Actions | Specialist Referral |
|---|---|---|---|---|
| Airway/Breathing | <5 minutes | Choanal atresia, micrognathia | Oral airway, intubation | ENT, anesthesia |
| Circulation | <10 minutes | Cyanosis, murmur, pulses | Pulse ox, echo, PGE | Cardiology, NICU |
| Neurological | <30 minutes | Tone, reflexes, seizures | Glucose, electrolytes | Neurology, genetics |
| Gastrointestinal | <1 hour | Abdominal wall defects | IV access, NGT, warming | Pediatric surgery |
| Genitourinary | <24 hours | Ambiguous genitalia, masses | Karyotype, electrolytes | Urology, endocrine |
- Management Decision Trees
- Surgical timing: immediate (life-threatening), urgent (<48 hours), elective (optimal timing)
- Multidisciplinary coordination: genetics, surgery, cardiology, neurology involvement
- Family counseling: recurrence risks, prenatal diagnosis options, support resources
- Long-term monitoring: growth, development, organ function surveillance
- Transition planning: adult care preparation for chronic conditions
These systematic assessment tools enable rapid recognition, appropriate triage, and evidence-based management of developmental anomalies across diverse clinical presentations.
🎯 The Clinical Arsenal: Rapid Assessment & Management Tools
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app


















