Back
Zymogens Explained: What They Are, How They Work, and High-Yield MCQs for USMLE Step 1 (2026)
Master zymogen activation for USMLE Step 1. Learn why pancreas needs inactive enzyme precursors, high-yield activation sequences, and practice MCQs with detailed explanations.

Zymogens Explained: What They Are, How They Work, and High-Yield MCQs for USMLE Step 1 (2026)
You are probably looking at another enzyme table wondering why pepsinogen exists when pepsin does the actual work. Why does your pancreas store trypsinogen instead of trypsin? USMLE Step 1 loves testing zymogens because most students memorize the names without understanding the logic.
Here is what you need to know: zymogens exist to prevent autodigestion. Your pancreas would literally digest itself if it stored active proteases. The activation cascade isnt just biochemistry trivia — its a safety mechanism that prevents your organs from self-destructing.
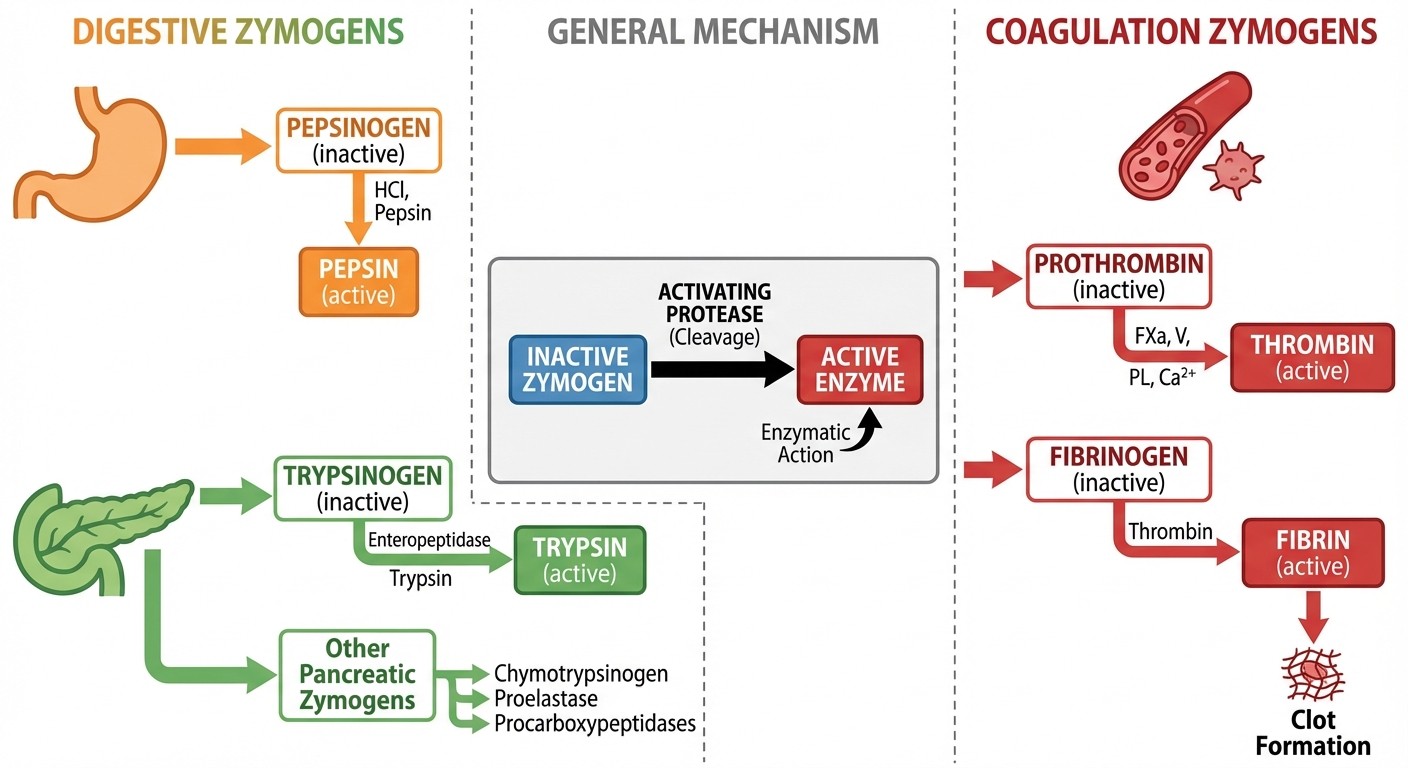
Step 1 tests zymogen activation in 3 contexts: digestive enzymes (pepsinogen to pepsin), pancreatic proteases (the trypsin cascade), and coagulation factors (prothrombin to thrombin). Each follows the same principle but with different triggers. Master the pattern once, apply it everywhere.
This guide covers the high-yield zymogens, their activation mechanisms, and the MCQ patterns Step 1 actually tests. No fluff — just what gets you points.
What Are Zymogens? The Logic Behind Inactive Precursors
Zymogens are inactive enzyme precursors that require proteolytic cleavage to become functional. They are also called proenzymes — same concept, different name. Step 1 uses both terms interchangeably.
The key insight: zymogens exist where active enzymes would cause damage. Your stomach produces pepsinogen instead of pepsin because pepsin would digest the gastric mucosa before reaching food. Your pancreas stores trypsinogen because active trypsin would digest pancreatic proteins and cause acute pancreatitis.
Three main reasons cells use zymogens:
1. Autodigestion prevention: Proteases stored as inactive precursors cant damage the cells that make them
2. Spatial control: Activation happens where needed, not where produced
3. Signal amplification: One activated enzyme can activate many others in cascade fashion
This logic applies beyond digestion. Blood coagulation uses zymogens because you dont want spontaneous clotting in healthy vessels. Fibrinolysis uses plasminogen because active plasmin would dissolve clots indiscriminately.
When studying zymogen activation with Oncourse enzyme regulation lessons, you can ask Rezzy specific mechanism questions like "Why does enterokinase only activate trypsinogen and not other pancreatic zymogens?" The AI tutor explains the structural specificity without you having to dig through multiple textbook pages.
Mechanism of Zymogen Activation: Limited Proteolysis
Zymogen activation occurs through limited proteolysis — cleavage of specific peptide bonds that expose the active site. This is different from protein degradation, which breaks down the entire molecule.
The process follows these steps:
1. Recognition: Activating enzyme recognizes specific cleavage site on zymogen
2. Cleavage: Peptide bond is cut, often removing an inhibitory peptide
3. Conformational change: Protein refolds to expose active site
4. Activation: Enzyme becomes catalytically active
The Trypsin Cascade: Master Pattern for Step 1
Pancreatic enzyme activation follows a specific sequence that Step 1 tests repeatedly:
1. Enterokinase (brush border enzyme) activates trypsinogen to trypsin
2. Trypsin then activates other pancreatic zymogens:
- Chymotrypsinogen → chymotrypsin
- Proelastase → elastase
- Procarboxypeptidase A → carboxypeptidase A
- Procarboxypeptidase B → carboxypeptidase B
This cascade amplifies the signal. One enterokinase molecule activates multiple trypsin molecules, which then activate hundreds of other proteases. Its biochemical signal amplification.
For memorizing the complete zymogen activation sequence, the Oncourse mnemonic engine creates acronym-based recall chains that link all precursors in order. Instead of cramming individual enzyme pairs, you get the entire cascade in a single memorable phrase.
High-Yield Zymogens for USMLE Step 1
Digestive Enzyme Zymogens
Pepsinogen → Pepsin
Location: Stomach (chief cells)
Activator: Low pH (stomach acid)
Function: Protein digestion in acidic environment
Step 1 pearl: Pepsinogen can also be activated by pepsin itself (positive feedback)
Pancreatic Protease Zymogens
Trypsinogen → Trypsin (enterokinase activation)
Chymotrypsinogen → Chymotrypsin (trypsin activation)
Proelastase → Elastase (trypsin activation)
Procarboxypeptidase A/B → Carboxypeptidase A/B (trypsin activation)
Coagulation Cascade Zymogens
The coagulation cascade is essentially a zymogen activation sequence:
Intrinsic Pathway Factors
Factor XII (Hageman factor) → Factor XIIa
Factor XI → Factor XIa
Factor IX → Factor IXa
Factor VIII → Factor VIIIa
Extrinsic Pathway Factors
Factor VII → Factor VIIa
Factor X → Factor Xa
Common Pathway
Prothrombin (Factor II) → Thrombin (Factor IIa)
Fibrinogen → Fibrin
Fibrinolytic System
Plasminogen → Plasmin (activated by tissue plasminogen activator)
Each coagulation factor exists as an inactive precursor until activated by the preceding factor in the cascade. This prevents spontaneous coagulation while allowing rapid response when needed.
Review these zymogen facts with Oncourse biochemistry flashcards using spaced repetition. The algorithm schedules reviews based on your retrieval difficulty — if you keep missing proelastase activation, youll see it more frequently until it sticks.
Step 1 Testing Patterns: How Zymogens Appear on Exam
USMLE Step 1 tests zymogens in predictable patterns. Recognizing these helps you spot the correct answer faster.
Pattern 1: Activation Sequence Questions
"A patient has a genetic deficiency of enterokinase. Which of the following enzymes would be most directly affected?"
Answer: Trypsinogen activation. Without enterokinase, trypsinogen cant be activated, which blocks the entire pancreatic protease cascade.
Pattern 2: Hereditary Pancreatitis
"A mutation in the trypsinogen gene (PRSS1) causes hereditary pancreatitis. The mutation prevents normal regulation of trypsin activity. What is the most likely mechanism?"
Answer: Mutation creates trypsinogen that resists inactivation, leading to persistent trypsin activity and autodigestion of pancreatic tissue.
Pattern 3: Zymogen Granule Histology
"Electron microscopy of pancreatic acinar cells shows abundant electron-dense granules. These granules primarily contain:"
Answer: Inactive enzyme precursors (zymogens) including trypsinogen, chymotrypsinogen, and proelastase.
Pattern 4: Coagulation Factor Activation
"Factor X is activated by both intrinsic and extrinsic pathways. In its inactive form, Factor X is best described as:"
Answer: A zymogen that requires limited proteolysis for activation.
When practicing these question types, the Probe Game tests all 8 major zymogen pairs within 2 minutes. You get instant feedback on which enzyme activations you know cold versus which need more review.
Clinical Correlations: When Zymogen Regulation Fails
Understanding zymogen pathophysiology helps with Step 1 clinical vignettes.
Acute Pancreatitis
Premature trypsinogen activation within pancreatic acinar cells causes autodigestion. Common triggers include alcohol, gallstones, and certain drugs. The pancreas literally digests itself because zymogens activate in the wrong location.
Hereditary Pancreatitis
PRSS1 mutations affect trypsinogen regulation. Instead of normal inactivation, mutant trypsinogen remains persistently active, causing recurrent pancreatitis episodes.
Hemophilia
Coagulation factor deficiencies (VIII, IX, XI) are zymogens that cant be properly activated. Without functional activation cascades, patients have prolonged bleeding times.
Peptic Ulcer Disease
H. pylori infection disrupts the pepsinogen/pepsin balance in gastric mucosa. Increased pepsin activity combined with decreased protective factors leads to mucosal ulceration.
These clinical scenarios frequently appear in Step 1 vignettes testing zymogen knowledge.
High-Yield Practice MCQs
Question 1:
A 45-year-old man presents with severe epigastric pain radiating to his back. Laboratory studies show elevated serum amylase and lipase. If this condition involves premature activation of pancreatic zymogens, which enzyme would FIRST become active?
A) Chymotrypsin
B) Elastase
C) Trypsin
D) Carboxypeptidase A
E) Pepsin
Answer: C) Trypsin. Enterokinase normally activates trypsinogen to trypsin, which then activates other pancreatic zymogens. In acute pancreatitis, trypsin is the first protease activated. Question 2:
A patient has a hereditary deficiency of enterokinase. Which of the following would be most directly impaired?
A) Gastric protein digestion
B) Pancreatic protein digestion
C) Blood coagulation
D) Fibrinolysis
E) Fat digestion
Answer: B) Pancreatic protein digestion. Enterokinase activates trypsinogen to trypsin, which is required for activating other pancreatic proteases. Question 3:
In the coagulation cascade, prothrombin is converted to thrombin by:
A) Factor VIIa
B) Factor IXa
C) Factor Xa
D) Factor XIa
E) Plasmin
Answer: C) Factor Xa. Factor Xa (along with Factor Va as cofactor) converts prothrombin to thrombin in the common pathway. Question 4:
A research study examines pancreatic acinar cells under electron microscopy. The abundant electron-dense granules in these cells primarily contain:
A) Active digestive enzymes
B) Inactive enzyme precursors
C) Ribosomes and rough ER
D) Mitochondria
E) Lipid droplets
Answer: B) Inactive enzyme precursors. Zymogen granules contain inactive forms of digestive enzymes to prevent autodigestion. Question 5:
Which of the following best explains why pepsinogen is secreted instead of pepsin?
A) Pepsinogen is more stable at body temperature
B) Pepsin would digest gastric mucosa
C) Pepsinogen has higher catalytic activity
D) Pepsin cannot function at low pH
E) Pepsinogen requires less energy to produce
Answer: B) Pepsin would digest gastric mucosa. Pepsinogen prevents autodigestion until activated by stomach acid.
Practice more zymogen activation MCQs with detailed explanations for each answer choice.
Step 1 Study Strategy: Mastering Enzyme Regulation
Focus your zymogen review on these high-yield concepts:
1. Memorize the activation sequence: Enterokinase → trypsinogen → trypsin → other pancreatic zymogens
2. Understand the why: Zymogens prevent autodigestion and provide signal amplification
3. Connect to pathology: Acute pancreatitis, hereditary pancreatitis, coagulation disorders
4. Practice recognition: Identify zymogens in question stems and predict the activation mechanism
The clinical applications matter more than pure memorization. Step 1 wants you to apply zymogen concepts to solve problems, not just recall lists.
When using Oncourse vitamin K and coagulation lessons, connect the coagulation factor zymogens to vitamin K deficiency. Understanding how vitamin K affects zymogen activation helps with bleeding disorder questions.
Common Mistakes to Avoid
Mistake 1: Confusing activation triggers
Enterokinase activates trypsinogen. Trypsin activates other pancreatic zymogens. Dont mix up the sequence.
Mistake 2: Forgetting autodigestion prevention
Always remember WHY zymogens exist. Its not random — its protection against enzyme damage.
Mistake 3: Missing the cascade effect
One activated enzyme can activate many others. This amplification is crucial for both digestion and coagulation.
Mistake 4: Ignoring clinical correlations
Step 1 rarely asks pure biochemistry. Connect zymogens to pancreatitis, bleeding disorders, and ulcer disease.
Frequently Asked Questions
Why dont all enzymes exist as zymogens?
Only enzymes that could damage their host cells need zymogen forms. Metabolic enzymes like hexokinase or catalase dont cause tissue damage, so they can be produced in active form. Proteases are different — they can digest any protein, including the cell that made them.
What happens if trypsinogen activates inside pancreatic cells?
This causes acute pancreatitis. The pancreas literally digests itself because active trypsin breaks down pancreatic proteins. Its a medical emergency requiring immediate treatment.
How does enterokinase know to activate only trypsinogen?
Enterokinase recognizes a specific amino acid sequence in trypsinogen that acts like a molecular key. This sequence doesnt exist in other pancreatic zymogens, so enterokinase cant activate them directly.
Are there zymogens outside of digestion and coagulation?
Yes. Complement proteins (C3, C5), some hormones (angiotensinogen to angiotensin), and apoptosis enzymes (caspases) also exist as inactive precursors. The principle is the same — prevent unwanted activation until needed.
Why does pepsinogen activate at low pH while pancreatic zymogens need specific enzymes?
Different environments, different triggers. Stomach acid provides a reliable pH signal for pepsin activation. The small intestine doesnt have extreme pH, so it uses enzyme-based activation instead.
How do coagulation factor zymogens prevent spontaneous clotting?
Each factor requires activation by the previous factor in the cascade. Without an initial trigger (tissue damage), the cascade doesnt start. This multi-step requirement prevents random clot formation in healthy vessels.
Prepare smarter with Oncourse AI — adaptive MCQs, spaced repetition, and AI explanations built for USMLE Step 1. Download free on Android and iOS.