Back
NEET PG Antimicrobial Pharmacology Glossary 2026: High-Yield Drug Classes, Mechanisms of Action and MCQ Mnemonics
Master antimicrobial pharmacology for NEET PG 2026 with this comprehensive glossary covering drug classes, mechanisms, resistance patterns, and clinical scenarios. Includes high-yield mnemonics and MCQ strategies.
NEET PG Antimicrobial Pharmacology Glossary 2026: High-Yield Drug Classes, Mechanisms of Action and MCQ Mnemonics
You are probably staring at another antimicrobial classification table, wondering how you'll memorize 47 different drug names, their mechanisms, and spectrum patterns before NEET PG. Here's the thing: antimicrobial pharmacology makes up 12-15% of the pharmacology section, and it's tested differently than you think.
NEET PG doesnt just ask "What's the mechanism of penicillin?" They ask scenario-based questions: "A 28-year-old presents with pneumonia. Sputum culture shows penicillin-resistant Streptococcus pneumoniae. Which drug would you choose?" The difference between scoring 65% and 85% in pharmacology lies in pattern recognition, not rote memorization.
This glossary organizes antimicrobials the way NEET PG tests them — by clinical scenarios, resistance patterns, and high-yield associations. Every entry includes the mechanism, spectrum, key adverse effects, and a memory hook thats actually useful during the exam.
Core Antimicrobial Mechanisms of Action
Before diving into specific drugs, master these 5 fundamental mechanisms. Every antimicrobial question ultimately traces back to one of these pathways.
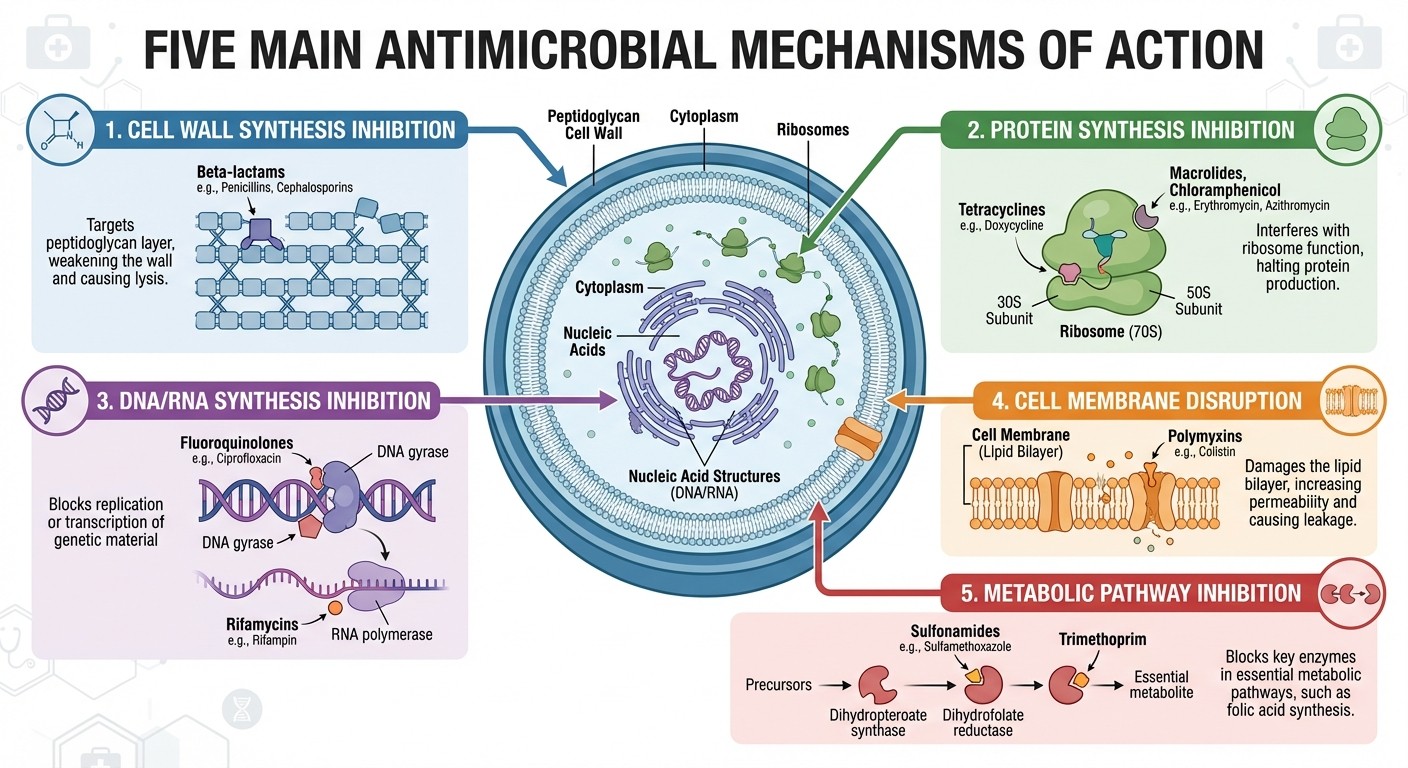
1. Cell Wall Synthesis Inhibition
Target: Peptidoglycan cross-linking in bacterial cell wall Drug Classes: Beta-lactams (penicillins, cephalosporins, carbapenems, monobactams), vancomycin, bacitracin Clinical Pearl: Most bactericidal against actively dividing organisms NEET PG Trap: Ineffective against mycoplasma (no cell wall) and in stationary-phase infections
2. Protein Synthesis Inhibition
30S Ribosomal Subunit: Aminoglycosides, tetracyclines 50S Ribosomal Subunit: Chloramphenicol, macrolides, lincosamides Clinical Pearl: 30S inhibitors are typically bactericidal; 50S inhibitors are bacteriostatic Memory Hook: "30 kills (cidal), 50 stops (static)" — though watch for exceptions like chloramphenicol
3. DNA/RNA Synthesis Inhibition
DNA Gyrase/Topoisomerase: Fluoroquinolones RNA Polymerase: Rifampin DNA Synthesis: Metronidazole (anaerobes) NEET PG Favorite: Fluoroquinolone resistance mechanisms and tendon rupture risk
4. Cell Membrane Disruption
Mechanism: Direct membrane damage Examples: Polymyxins (colistin), daptomycin, antifungals (amphotericin B, azoles) Clinical Use: Often reserved for multidrug-resistant organisms Key Point: Nephrotoxicity is common due to membrane effects on kidney cells
5. Metabolic Pathway Inhibition
Folate Synthesis: Sulfonamides, trimethoprim Mycolic Acid Synthesis: Isoniazid, ethambutol (mycobacteria) Ergosterol Synthesis: Antifungals (azoles, allylamines)
Beta-Lactam Antibiotics: The NEET PG Workhorses
Beta-lactams remain the most tested antimicrobial class. Master the resistance patterns and clinical applications for each subgroup.
Penicillins
Natural Penicillins (Penicillin G/V)
Spectrum: Gram-positive cocci, anaerobes (except Bacteroides)
Clinical Use: Streptococcal infections, syphilis, gas gangrene
Resistance: Beta-lactamase production
NEET PG Pearl: Still first-line for Group A strep pharyngitis
Anti-Staphylococcal Penicillins (Methicillin, Nafcillin)
Key Feature: Beta-lactamase resistant
Limitation: Ineffective against MRSA (altered PBP2a)
Clinical Use: MSSA infections
Memory Hook: "Naf kills Staph, but not MRSA"
Aminopenicillins (Ampicillin, Amoxicillin)
Extended Spectrum: Adds some gram-negatives (E. coli, Proteus)
Clinical Use: UTI, respiratory tract infections, Listeria meningitis
Side Effect: Ampicillin rash with EBV
Combination: Amoxicillin + clavulanate extends to beta-lactamase producers
Anti-Pseudomonal Penicillins (Piperacillin)
Spectrum: Broadest penicillin spectrum including Pseudomonas
Clinical Use: Hospital-acquired infections, neutropenic fever
Combination: Piperacillin-tazobactam (Zosyn) — covers most hospital pathogens
NEET PG High-Yield: Know when to use pip-tazo vs carbapenems
For deeper understanding of beta-lactam mechanisms and resistance patterns, explore our beta-lactam antibiotics lessons and practice with targeted MCQs.
Cephalosporins by Generation
First Generation (Cefazolin, Cephalexin)
Spectrum: Gram-positive cocci, some gram-negatives
Clinical Use: Skin/soft tissue infections, surgical prophylaxis
Memory: "First gen = First choice for skin"
Second Generation (Cefuroxime, Cefoxitin)
Added Coverage: H. influenzae, Moraxella, anaerobes (cefoxitin)
Clinical Use: Respiratory tract infections, mixed infections
NEET PG Favorite: Cefoxitin for Bacteroides coverage
Third Generation (Ceftriaxone, Ceftazidime)
Spectrum: Excellent gram-negative coverage, reduced gram-positive
Clinical Use: Meningitis (ceftriaxone), Pseudomonas (ceftazidime)
Key Difference: Ceftazidime covers Pseudomonas; ceftriaxone doesnt
Side Effects: Biliary sludge (ceftriaxone), bleeding (cefoperazone)
Fourth Generation (Cefepime)
Spectrum: Combines 3rd gen gram-negative with better gram-positive
Clinical Use: Hospital-acquired pneumonia, neutropenic fever
Advantage: More stable against beta-lactamases
Fifth Generation (Ceftaroline)
Unique Feature: MRSA coverage
Mechanism: Binds to PBP2a
Clinical Use: Complicated skin infections, pneumonia
When working through cephalosporin classification questions, try using Oncourse flashcards for spaced repetition of generation-specific spectrums.
Carbapenems and Monobactams
Carbapenems (Imipenem, Meropenem, Ertapenem)
Spectrum: Broadest beta-lactam spectrum
Clinical Use: Serious gram-negative infections, empiric therapy for sepsis
Resistance: Carbapenemases (NDM, KPC, OXA)
Side Effects: Seizures (imipenem > meropenem), C. diff
NEET PG Pearl: Ertapenem doesnt cover Pseudomonas or Acinetobacter
Monobactams (Aztreonam)
Unique Feature: Only beta-lactam safe in penicillin allergy
Spectrum: Gram-negative only (no gram-positive or anaerobic activity)
Clinical Use: Gram-negative infections in penicillin-allergic patients
Memory Hook: "AZT = Anti-Zero (gram-positive), Totally safe (in allergy)"
Protein Synthesis Inhibitors
Aminoglycosides
Examples: Gentamicin, amikacin, tobramycin, streptomycin Mechanism: 30S ribosomal subunit binding, bactericidal Spectrum: Gram-negative aerobes, synergy with cell wall inhibitors Clinical Uses:
Serious gram-negative infections
Endocarditis (with beta-lactams)
Tuberculosis (streptomycin)
Toxicities:
Nephrotoxicity: Dose-dependent, reversible
Ototoxicity: Vestibular (streptomycin) vs cochlear (gentamicin)
Neuromuscular blockade: Especially post-anesthesia
NEET PG High-Yield: Once-daily dosing reduces toxicity while maintaining efficacy due to concentration-dependent killing and post-antibiotic effect. Resistance Mechanisms:
1. Enzymatic modification (most common)
2. Altered ribosomal binding
3. Reduced uptake
Tetracyclines
Examples: Tetracycline, doxycycline, minocycline, tigecycline Mechanism: 30S ribosomal subunit, bacteriostatic Spectrum: Broad-spectrum including atypicals, rickettsiae, spirochetes Clinical Applications:
Doxycycline: Malaria prophylaxis, rickettsial diseases, atypical pneumonia
Minocycline: MRSA skin infections
Tigecycline: Multi-drug resistant gram-positives and gram-negatives
Contraindications: Pregnancy, children under 8 (teeth staining) Side Effects: GI upset, photosensitivity, pseudotumor cerebri
The mnemonic "VACUUM PACKED" helps remember tetracycline coverage: Vibrio, Atypicals, Chlamydia, Ureaplasma, Ureaplasma, Mycoplasma, Pasteurella, Anaerobes (some), Chlamydia, Klebsiella, Ehrlichia, Dermatophytes. Rezzy AI can help create personalized mnemonics based on your specific weak areas.
Macrolides and Related
Macrolides (Erythromycin, Clarithromycin, Azithromycin) Mechanism: 50S ribosomal subunit, bacteriostatic Spectrum: Gram-positives, atypicals, some gram-negatives Clinical Pearls:
Azithromycin: Longest half-life, better gram-negative coverage
Clarithromycin: H. pylori eradication, MAC prophylaxis
Erythromycin: GI motility agent, most GI side effects
Drug Interactions: CYP3A4 inhibition (except azithromycin) Cardiac Risk: QT prolongation, especially with azithromycin Clindamycin
Spectrum: Gram-positives, anaerobes (including Bacteroides)
Clinical Use: Anaerobic infections, severe strep/staph skin infections
Major Risk: C. difficile colitis
Mechanism: 50S ribosomal subunit (lincosamide)
DNA/RNA Synthesis Inhibitors
Fluoroquinolones
Mechanism: DNA gyrase (gram-negatives) and topoisomerase IV (gram-positives) inhibition Classification by Generations: Second Generation (Ciprofloxacin, Ofloxacin)
Spectrum: Excellent gram-negative including Pseudomonas
Clinical Use: UTI, GI infections, anthrax
Limitation: Poor gram-positive and anaerobic coverage
Third Generation (Levofloxacin)
Added Coverage: Streptococcus pneumoniae, atypicals
Clinical Use: Community-acquired pneumonia, sinusitis
Advantage: Once-daily dosing
Fourth Generation (Moxifloxacin)
Spectrum: Broadest including anaerobes
Clinical Use: Complicated pneumonia, complicated skin infections
Unique: No renal adjustment needed
Resistance Mechanisms:
1. Target enzyme mutations
2. Efflux pumps
3. Plasmid-mediated resistance
Toxicities:
Tendon rupture: Especially Achilles, increased with steroids/age
CNS effects: Seizures, psychosis
Cardiac: QT prolongation
Glucose: Hypo/hyperglycemia
NEET PG Pearl: Avoid in pregnancy, children, and myasthenia gravis.
Other DNA/RNA Inhibitors
Metronidazole
Mechanism: Reduction to toxic metabolites disrupting DNA
Spectrum: Anaerobes (including Bacteroides), protozoa
Clinical Use: C. difficile, anaerobic infections, H. pylori
Side Effects: Disulfiram-like reaction with alcohol, peripheral neuropathy
Rifampin
Mechanism: RNA polymerase inhibition
Clinical Use: Tuberculosis, MRSA (combination therapy), meningococcal prophylaxis
Drug Interactions: Powerful CYP inducer
Side Effect: Orange discoloration of body fluids
Glycopeptides and Lipopeptides
Vancomycin
Mechanism: Binds D-ala-D-ala peptide, preventing peptidoglycan cross-linking Spectrum: Gram-positive only, including MRSA Clinical Uses:
MRSA infections
C. difficile colitis (oral)
Endocarditis in penicillin-allergic patients
Toxicities:
Red man syndrome: Histamine release from rapid infusion
Nephrotoxicity: Especially with aminoglycosides
Ototoxicity: High trough levels
Resistance: VRE (vancomycin-resistant enterococci) — target becomes D-ala-D-lac
Newer Agents
Linezolid
Mechanism: 50S ribosomal subunit (unique binding site)
Spectrum: Gram-positives including VRE
Advantages: Oral bioavailability, tissue penetration
Toxicities: Thrombocytopenia, peripheral neuropathy, serotonin syndrome
Daptomycin
Mechanism: Cell membrane depolarization
Clinical Use: Complicated skin infections, bacteremia, endocarditis
Limitation: Inactivated by pulmonary surfactant
Side Effect: CK elevation, myopathy
Antimycobacterial Agents
Understanding TB drug combinations and resistance patterns is crucial for NEET PG internal medicine questions.
First-Line Anti-TB Drugs
Isoniazid (INH)
Mechanism: Inhibits mycolic acid synthesis
Resistance: KatG gene mutations
Toxicity: Peripheral neuropathy (give B6), hepatitis
Monitoring: LFTs, especially in elderly/alcoholics
Rifampin (RMP)
Mechanism: RNA polymerase inhibition
Resistance: rpoB gene mutations
Toxicity: Hepatitis, orange discoloration
Drug Interactions: CYP inducer
Ethambutol (EMB)
Mechanism: Inhibits arabinosyl transferase
Toxicity: Optic neuropathy (dose-related)
Monitoring: Monthly visual acuity and color discrimination
Contraindication: Children under 5 (cant assess vision)
Pyrazinamide (PZA)
Mechanism: Unknown (requires acidic pH)
Toxicity: Hepatitis, hyperuricemia
Limitation: Inactive against M. bovis
Standard Regimen: HRZE × 2 months, then HR × 4 months (total 6 months)
Second-Line Anti-TB Drugs
Used for drug-resistant TB or when first-line drugs cant be used:
Injectable Agents: Amikacin, capreomycin, streptomycin Fluoroquinolones: Levofloxacin, moxifloxacin Other Oral Agents: Ethionamide, cycloserine, PAS MDR-TB Definition: Resistance to at least INH + RMP XDR-TB Definition: MDR + resistance to fluoroquinolone + second-line injectable
Practice antimycobacterial scenarios with our antimycobacterial drugs MCQs to master resistance pattern recognition.
Antifungal Agents
Polyenes
Amphotericin B
Mechanism: Binds ergosterol, creates membrane pores
Spectrum: Broadest antifungal spectrum
Clinical Use: Serious systemic mycoses, empiric antifungal therapy
Formulations: Conventional (nephrotoxic) vs lipid formulations (less toxic)
Side Effects: Nephrotoxicity, infusion reactions, hypokalemia
Azoles
Mechanism: Inhibit 14α-demethylase (ergosterol synthesis) Fluconazole
Spectrum: Candida (except C. krusei), Cryptococcus
Clinical Use: Candidemia, cryptococcal meningitis, prophylaxis
Advantages: Oral/IV, good CNS penetration
Limitation: No activity against Aspergillus
Itraconazole
Spectrum: Broader including Aspergillus, endemic mycoses
Clinical Use: Histoplasmosis, blastomycosis, aspergillosis
Limitation: Poor oral absorption, drug interactions
Voriconazole
Spectrum: Aspergillus, Candida, emerging molds
Clinical Use: First-line for invasive aspergillosis
Side Effects: Visual disturbances, hepatitis, photosensitivity
Posaconazole
Spectrum: Broadest azole including mucormycosis
Clinical Use: Prophylaxis in high-risk patients, salvage therapy
Advantage: Once-daily dosing
Echinocandins
Examples: Caspofungin, micafungin, anidulafungin Mechanism: Inhibit β-glucan synthesis in fungal cell wall Spectrum: Candida, Aspergillus (not active against Cryptococcus) Clinical Use: Candidemia, invasive aspergillosis Advantage: Fungicidal against Candida, minimal drug interactions
Antiviral Agents
Anti-Herpes Drugs
Acyclovir
Mechanism: Guanosine analog, terminates DNA synthesis
Activation: Requires viral thymidine kinase
Clinical Use: HSV, VZV infections
Resistance: Thymidine kinase mutations
Valacyclovir: Oral prodrug of acyclovir, better bioavailability Ganciclovir
Spectrum: CMV, HSV, VZV
Clinical Use: CMV retinitis, CMV prophylaxis in transplant
Toxicity: Bone marrow suppression
Anti-Influenza Drugs
Oseltamivir (Tamiflu)
Mechanism: Neuraminidase inhibitor
Clinical Use: Influenza A and B treatment/prophylaxis
Timing: Most effective within 48 hours of symptom onset
Side Effects: Nausea, psychiatric symptoms (rare)
Zanamivir: Inhaled neuraminidase inhibitor
Anti-HIV Drugs
Nucleoside RTIs (NRTIs)
Examples: Zidovudine (AZT), lamivudine, emtricitabine
Mechanism: DNA chain termination
Side Effects: Mitochondrial toxicity, lactic acidosis
Non-Nucleoside RTIs (NNRTIs)
Examples: Efavirenz, rilpivirine
Mechanism: Allosteric RT inhibition
Resistance: Single point mutations
Protease Inhibitors
Examples: Atazanavir, darunavir
Mechanism: Prevent viral protein maturation
Side Effects: GI intolerance, metabolic syndrome
Integrase Inhibitors
Examples: Raltegravir, dolutegravir
Mechanism: Block viral DNA integration
Advantages: High barrier to resistance, fewer interactions
For comprehensive antiviral drug coverage, review our antiviral drugs lessons and test your knowledge with antiviral MCQs.
High-Yield Antimicrobial Resistance Patterns
MRSA (Methicillin-Resistant Staphylococcus aureus)
Mechanism: mecA gene produces altered PBP2a Treatment Options: Vancomycin, linezolid, daptomycin, ceftaroline Clinical Impact: Hospital-acquired vs community-acquired strains have different virulence
ESBL (Extended-Spectrum Beta-Lactamase)
Organisms: E. coli, Klebsiella Resistance Pattern: Resistant to penicillins, cephalosporins; sensitive to carbapenems Treatment: Carbapenems are preferred NEET PG Pearl: ESBL producers often co-resistant to fluoroquinolones
Carbapenem-Resistant Enterobacteriaceae (CRE)
Mechanisms: Carbapenemases (KPC, NDM, OXA), efflux pumps Treatment: Colistin, tigecycline (limited options) Clinical Significance: Pan-resistant organisms, high mortality
Vancomycin-Resistant Enterococci (VRE)
Mechanism: Van genes change target from D-ala-D-ala to D-ala-D-lac Treatment: Linezolid, daptomycin Prevention: Contact precautions, antimicrobial stewardship
Clinical Scenarios and Drug Selection
Community-Acquired Pneumonia
Typical Pathogens: S. pneumoniae, H. influenzae First Choice: Amoxicillin or macrolide (outpatient) Severe/Hospitalized: Beta-lactam + macrolide or respiratory fluoroquinolone
Healthcare-Associated Pneumonia
Risk Factors: Recent hospitalization, nursing home, immunocompromised Coverage Needed: MRSA, Pseudomonas Empiric Therapy: Pip-tazo or carbapenem + vancomycin or linezolid
Urinary Tract Infections
Uncomplicated Cystitis: Nitrofurantoin, trimethoprim-sulfamethoxazole, fosfomycin Complicated UTI: Fluoroquinolone or beta-lactam based on culture Catheter-Associated: Remove catheter + targeted therapy
Skin and Soft Tissue Infections
Cellulitis: Cephalexin (outpatient), clindamycin (MRSA risk) Necrotizing Fasciitis: Clindamycin + penicillin (Group A strep) Diabetic Foot: Broad-spectrum with anaerobic coverage
Understanding these clinical patterns helps answer NEET PG questions that test application rather than just memorization. The Synapses game can help reinforce these clinical associations through spaced repetition.
MCQ Memory Techniques
Drug Name Associations
Penicillin Generations:
"Natural Pen" = Natural penicillins (G, V)
"Amino Pen" = Aminopenicillins (ampicillin, amoxicillin)
"Anti-Staph" = Methicillin, nafcillin
"Anti-Pseudo" = Piperacillin
Cephalosporin Memory:
1st Gen: "CEF-azolin" (surgical prophylaxis)
2nd Gen: "CEF-uroxime" (respiratory)
3rd Gen: "CEF-triaxone" (meningitis), "CEF-tazidime" (Pseudomonas)
4th Gen: "CEF-epime" (hospital bugs)
Fluoroquinolone Spectrum:
"CIPRO" = Pseudomonas coverage
"LEVO" = Lung bugs (pneumonia)
"MOXI" = Most coverage (anaerobes too)
Toxicity Mnemonics
Aminoglycoside Toxicity: "Gentamicin Gives Nephritis, Ototoxicity, Neuromuscular blockade" Chloramphenicol: "Gray Baby Syndrome, Aplastic Anemia, Bone marrow suppression" Vancomycin: "Red Man syndrome, Renal toxicity, 'Ringing' (ototoxicity)"
Resistance Patterns
MRSA Treatment: "VLCD" = Vancomycin, Linezolid, Ceftaroline, Daptomycin Carbapenem Resistance: "KND-OXA" = KPC, NDM, OXA carbapenemases
Frequently Asked Questions
What percentage of NEET PG pharmacology questions focus on antimicrobials?
Antimicrobial pharmacology typically comprises 35-40% of the pharmacology section in NEET PG. This translates to approximately 12-15 questions out of the total pharmacology allocation. The questions often integrate clinical scenarios with drug selection, making pure memorization insufficient.
Which antimicrobial topics are most frequently tested in NEET PG 2026?
Based on recent patterns, beta-lactam antibiotics (especially cephalosporin generations and resistance mechanisms), fluoroquinolone classifications, antimycobacterial therapy for TB, and antifungal drug selection appear most frequently. Clinical scenario-based questions testing appropriate empiric therapy are increasingly common.
How should I approach antimicrobial resistance questions in NEET PG?
Focus on understanding mechanisms rather than memorizing organism lists. Key patterns include: MRSA (altered PBP), ESBL (extended beta-lactamase), carbapenem resistance (carbapenemases), and VRE (altered target). Know the first-line treatments for each resistant pattern.
What's the best way to remember antimicrobial drug classifications?
Use systematic approaches: group by mechanism first, then by spectrum. For beta-lactams, master the "generations" concept. For other classes, focus on unique features (aminoglycosides = ototoxicity, fluoroquinolones = tendon rupture). Clinical associations work better than pure lists.
Which antimicrobial side effects are high-yield for NEET PG MCQs?
Major toxicities tested include: aminoglycoside ototoxicity/nephrotoxicity, fluoroquinolone tendon rupture, vancomycin red man syndrome, chloramphenicol gray baby syndrome, and tetracycline teeth staining. Know both the toxicity and the population at risk.
How important are antiviral and antifungal drugs for NEET PG?
While bacterial antimicrobials dominate, antiviral therapy (especially anti-HIV combinations and oseltamivir) and antifungal drugs (azole mechanisms and amphotericin toxicity) appear regularly. They're often tested in clinical context - HIV treatment failure, candidemia management, or influenza prophylaxis scenarios.
Prepare smarter with Oncourse AI — adaptive MCQs, spaced repetition, and AI explanations built for NEET PG. Download free on Android and iOS.